

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
null
SMILES: Cc1ccn(n1)-c1cc(Cl)ccc1[C@@H](Oc1cc(nc(N)n1)C1=CCC(C[C@H](N)C(O)=O)CC1)C(F)(F)F
InChI Key:
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Tryptophan 5-hydroxylase 1 (Homo sapiens (Human)) | BDBM563495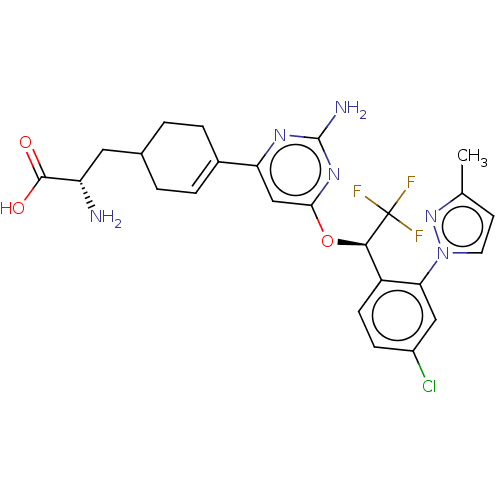 ((2S)-2-amino-3-(4-(2- amino-6-((R)-1-(4- chloro-...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 98 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description In order to confirm the inhibitory effect of the compound represented by formula 1 according to the present invention on the tryptophan hydroxylase a... | Citation and Details BindingDB Entry DOI: 10.7270/Q2Q52STW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Tryptophan 5-hydroxylase 1 (Homo sapiens (Human)) | BDBM563495 ((2S)-2-amino-3-(4-(2- amino-6-((R)-1-(4- chloro-...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 114 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description In order to confirm the inhibitory effect of the compound represented by formula 1 according to the present invention on the tryptophan hydroxylase a... | Citation and Details BindingDB Entry DOI: 10.7270/Q2Q52STW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||