

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM10027 5-[2-(1H-imidazol-1-yl)ethyl]-7,8-dihydroquinoline::5-[2-(Imidazol-1-yl)ethyl]-7,8-dihydroquinoline::CHEMBL293122::dihydroquinoline 31
SMILES: C(Cn1ccnc1)C1=CCCc2ncccc12
InChI Key: InChIKey=DJMSMHXCSIBVSA-UHFFFAOYSA-N
Data: 4 IC50
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Cytochrome P450 19A1 (Homo sapiens (Human)) | BDBM10027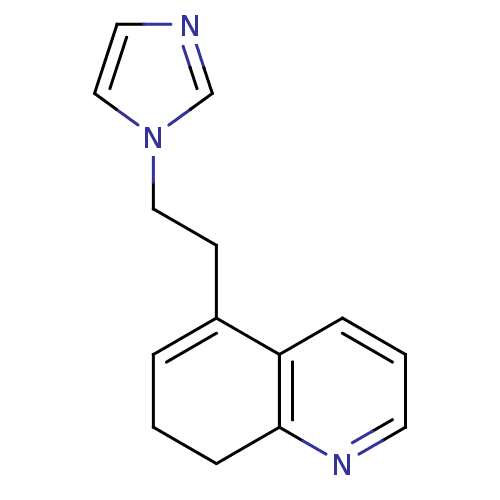 (5-[2-(1H-imidazol-1-yl)ethyl]-7,8-dihydroquinoline...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 500 | n/a | n/a | n/a | n/a | 7.4 | 30 |
Universitat des Saarlandes | Assay Description The enzyme activity was assayed by measuring the 3H-labeled H2O formed from [1beta,2beta-3H]testosterone during aromatization. After incubation, the ... | J Med Chem 43: 1841-51 (2000) Article DOI: 10.1021/jm991180u BindingDB Entry DOI: 10.7270/Q2D21VTH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cytochrome P450 19A1 (Homo sapiens (Human)) | BDBM10027 (5-[2-(1H-imidazol-1-yl)ethyl]-7,8-dihydroquinoline...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 500 | n/a | n/a | n/a | n/a | n/a | n/a |
Universidade de Coimbra Curated by ChEMBL | Assay Description Inhibition of human aromatase-mediated conversion of [1beta3H]androstenedione to estrone by liquid scintillation counting in presence of NADPH | J Med Chem 52: 143-50 (2009) Article DOI: 10.1021/jm800945c BindingDB Entry DOI: 10.7270/Q2SQ9080 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cytochrome P450 19A1 (Homo sapiens (Human)) | BDBM10027 (5-[2-(1H-imidazol-1-yl)ethyl]-7,8-dihydroquinoline...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 500 | n/a | n/a | n/a | n/a | n/a | n/a |
American University of Ras Al Khaimah Curated by ChEMBL | Assay Description Inhibition of aromatase (unknown origin) | Eur J Med Chem 102: 375-86 (2015) BindingDB Entry DOI: 10.7270/Q2ZC84Q6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Thromboxane A2 Synthase (P450 TxA2) (Homo sapiens (Human)) | BDBM10027 (5-[2-(1H-imidazol-1-yl)ethyl]-7,8-dihydroquinoline...) | KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 290 | n/a | n/a | n/a | n/a | n/a | n/a |
Universitat des Saarlandes | Assay Description Thromboxane A2 synthase activity was assayed by measuring the fragmentation of prostaglandin H2 to form 12-hydroxy-5, 8,10-heptadecatrienoic acid and... | J Med Chem 43: 1841-51 (2000) Article DOI: 10.1021/jm991180u BindingDB Entry DOI: 10.7270/Q2D21VTH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||