

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM17350 (4S)-4-[(2S)-2-{[(2S,3S)-3-{[(tert-butoxy)carbonyl]amino}-2-hydroxy-4-phenylbutyl]amino}-3-phenylpropanamido]-4-{[(1S)-1-carbamoyl-2-phenylethyl]carbamoyl}butanoic acid::CHEMBL386563
SMILES: CC(C)(C)OC(=O)N[C@@H](Cc1ccccc1)[C@@H](O)CN[C@@H](Cc1ccccc1)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](Cc1ccccc1)C(N)=O
InChI Key: InChIKey=MPMUDVMRFYJRLP-XDIGFQIYSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| HIV-1 Protease (Human immunodeficiency virus type 1) | BDBM17350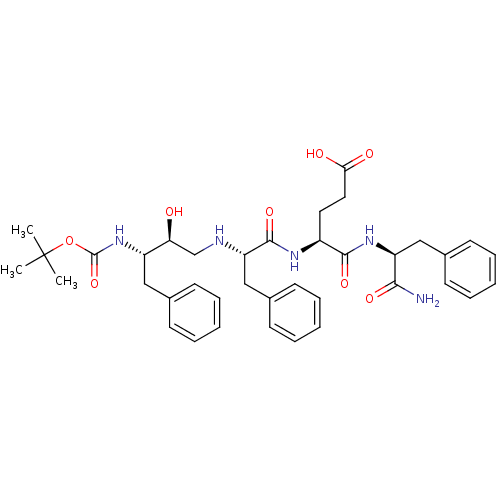 ((4S)-4-[(2S)-2-{[(2S,3S)-3-{[(tert-butoxy)carbonyl...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Similars | PDB Article PubMed | 0.150 | n/a | n/a | n/a | n/a | n/a | n/a | 4.7 | n/a |
Academy of Sciences of the Czech Republic | Assay Description Inhibition constants were determined by a spectrophotometric assay with the chromogenic peptide substrate. | J Med Chem 46: 1636-44 (2003) Article DOI: 10.1021/jm021079g BindingDB Entry DOI: 10.7270/Q29Z932M | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Human immunodeficiency virus type 1 protease (Human immunodeficiency virus type 1) | BDBM17350 ((4S)-4-[(2S)-2-{[(2S,3S)-3-{[(tert-butoxy)carbonyl...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Similars | PDB Article PubMed | 0.150 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Academy of Sciences of the Czech Republic Curated by ChEMBL | Assay Description Inhibition of wild type HIV1 protease | J Med Chem 49: 5777-84 (2006) Article DOI: 10.1021/jm0605583 BindingDB Entry DOI: 10.7270/Q2X63MK8 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Human immunodeficiency virus type 1 protease (Human immunodeficiency virus type 1) | BDBM17350 ((4S)-4-[(2S)-2-{[(2S,3S)-3-{[(tert-butoxy)carbonyl...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Similars | PDB Article PubMed | 86 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Academy of Sciences of the Czech Republic Curated by ChEMBL | Assay Description Inhibition of HIV1 protease I8 mutant | J Med Chem 49: 5777-84 (2006) Article DOI: 10.1021/jm0605583 BindingDB Entry DOI: 10.7270/Q2X63MK8 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||