

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50084905 CHEMBL147964::{1-[2-Dibenzofuran-3-yl-1-(1H-tetrazol-5-yl)-ethylamino]-3-naphthalen-1-yl-propyl}-phosphonic acid
SMILES: OP(O)(=O)C(CCc1cccc2ccccc12)N[C@@H](Cc1ccc2c(c1)oc1ccccc21)c1nnn[nH]1
InChI Key: InChIKey=TUPJIGTXAYPCTF-BXXZMZEQSA-N
Data: 4 IC50
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Endothelin-converting enzyme 1 (ECE1) (Homo sapiens (Human)) | BDBM50084905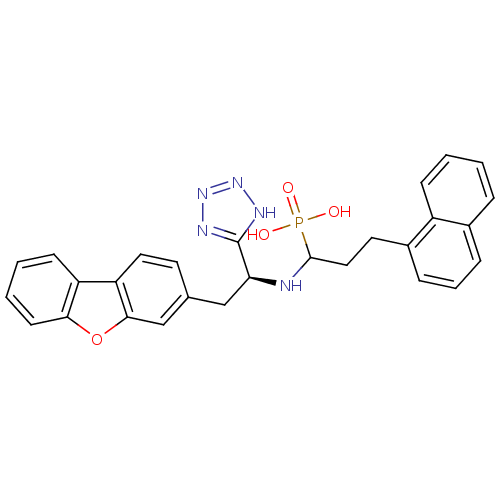 (CHEMBL147964 | {1-[2-Dibenzofuran-3-yl-1-(1H-tetra...) | PDB UniProtKB/SwissProt UniProtKB/TrEMBL antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | PubMed | n/a | n/a | 2.60 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institute for Biomedical Research Curated by ChEMBL | Assay Description In Vitro inhibition of recombinant human endothelin converting enzyme-1 | J Med Chem 43: 488-504 (2000) BindingDB Entry DOI: 10.7270/Q21G0MZR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neutral Endopeptidase (NEP) (Rattus norvegicus (Rat)) | BDBM50084905 (CHEMBL147964 | {1-[2-Dibenzofuran-3-yl-1-(1H-tetra...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | PubMed | n/a | n/a | 1.30E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institute for Biomedical Research Curated by ChEMBL | Assay Description In vitro inhibition of rat neutral endopeptidase | J Med Chem 43: 488-504 (2000) BindingDB Entry DOI: 10.7270/Q21G0MZR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Endothelin-converting enzyme 1 (ECE1) (Homo sapiens (Human)) | BDBM50084905 (CHEMBL147964 | {1-[2-Dibenzofuran-3-yl-1-(1H-tetra...) | PDB UniProtKB/SwissProt UniProtKB/TrEMBL antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | PubMed | n/a | n/a | 440 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institute for Biomedical Research Curated by ChEMBL | Assay Description In Vitro inhibition of recombinant human endothelin converting enzyme-1 | J Med Chem 43: 488-504 (2000) BindingDB Entry DOI: 10.7270/Q21G0MZR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neutral Endopeptidase (NEP) (Rattus norvegicus (Rat)) | BDBM50084905 (CHEMBL147964 | {1-[2-Dibenzofuran-3-yl-1-(1H-tetra...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | PubMed | n/a | n/a | 210 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institute for Biomedical Research Curated by ChEMBL | Assay Description In vitro inhibition of rat neutral endopeptidase | J Med Chem 43: 488-504 (2000) BindingDB Entry DOI: 10.7270/Q21G0MZR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||