

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50093808 (5-{1-[3-(Carboxymethyl-carbamoyl)-5-chloro-4-hydroxy-phenyl]-4-[(3S,10S,13R,17R)-17-(1,5-dimethyl-hexyl)-10,13-dimethyl-hexadecahydro-cyclopenta[a]phenanthren-3-yl]-but-1-enyl}-3-chloro-2-hydroxy-benzoylamino)-acetic acid::(5-{1-[3-(Carboxymethyl-carbamoyl)-5-chloro-4-hydroxy-phenyl]-4-[17-(1,5-dimethyl-hexyl)-10,13-dimethyl-hexadecahydro-cyclopenta[a]phenanthren-3-yl]-but-1-enyl}-3-chloro-2-hydroxy-benzoylamino)-acetic acid::5-{1-[3-carboxylato-4-(3-carboxy-2-methoxybenzyloxy)-5-chlorophenyl]-4-[1-(1,5-dimethylhexyl)-9a,11a-dimethylperhydrocyclopenta[a]phenanthren-7-yl]-1-butenyl}-2-(3-carboxy-2-methoxybenzyloxy)-3-chlorobenzoate::CHEMBL83315
SMILES: [#6]-[#6](-[#6])-[#6]-[#6]-[#6]-[#6](-[#6])-[#6@H]1-[#6]-[#6]-[#6]2-[#6]-3-[#6]-[#6]-[#6]4-[#6]-[#6@@H](-[#6]-[#6]\[#6]=[#6](\c5cc(Cl)c(-[#8])c(c5)-[#6](=O)-[#7]-[#6]-[#6](-[#8])=O)-c5cc(Cl)c(-[#8])c(c5)-[#6](=O)-[#7]-[#6]-[#6](-[#8])=O)-[#6]-[#6][C@]4([#6])[#6]-3-[#6]-[#6][C@]12[#6]
InChI Key: InChIKey=BIGCFRKTVFGWBA-QEMXKSKQSA-N
Data: 1 EC50
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| T-cell surface antigen CD4 (Homo sapiens (Human)) | BDBM50093808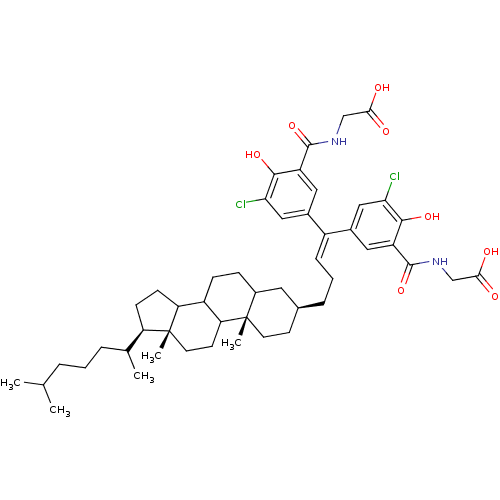 ((5-{1-[3-(Carboxymethyl-carbamoyl)-5-chloro-4-hydr...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | n/a | n/a | >1.25E+5 | n/a | n/a | n/a | n/a |
Purdue University Curated by ChEMBL | Assay Description In vitro inhibition of cytopathic effect was determined against HIV-2 ROD in MT-4 cells using MTS cytoprotection assay | J Med Chem 44: 703-14 (2001) BindingDB Entry DOI: 10.7270/Q2TX3GKX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||