 Found 8 hits for monomerid = 50112367
Found 8 hits for monomerid = 50112367 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kcal/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Nuclear receptor ROR-gamma (RORC)
(Homo sapiens (Human)) | BDBM50112367
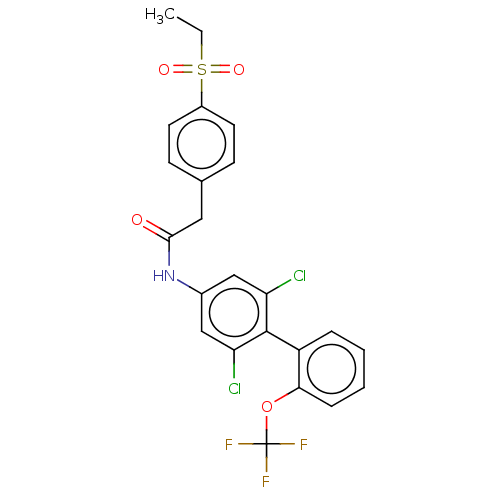
(CHEMBL3609407)Show SMILES CCS(=O)(=O)c1ccc(CC(=O)Nc2cc(Cl)c(c(Cl)c2)-c2ccccc2OC(F)(F)F)cc1 |(-3.74,3.7,;-2.67,3.08,;-2.67,1.54,;-3.73,.92,;-3.74,2.16,;-1.33,.77,;,1.54,;1.33,.77,;1.33,-.77,;2.67,-1.54,;2.66,-3.08,;1.6,-3.7,;4,-3.86,;3.99,-5.4,;5.33,-6.17,;5.32,-7.71,;6.39,-8.33,;3.98,-8.48,;2.65,-7.7,;1.58,-8.32,;2.66,-6.16,;3.98,-10.02,;2.64,-10.78,;2.63,-12.32,;3.96,-13.1,;5.3,-12.34,;5.31,-10.8,;6.65,-10.03,;7.98,-10.81,;9.05,-10.21,;7.97,-12.05,;9.04,-11.44,;,-1.54,;-1.33,-.77,)| Show InChI InChI=1S/C23H18Cl2F3NO4S/c1-2-34(31,32)16-9-7-14(8-10-16)11-21(30)29-15-12-18(24)22(19(25)13-15)17-5-3-4-6-20(17)33-23(26,27)28/h3-10,12-13H,2,11H2,1H3,(H,29,30) | PDB
UniProtKB/SwissProt
UniProtKB/TrEMBL
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
| PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Fudan University
Curated by ChEMBL
| Assay Description
Inhibition of RORgammat (unknown origin) assessed as inhibition of agonist-induced response by FRET assay |
ACS Med Chem Lett 6: 787-92 (2015)
BindingDB Entry DOI: 10.7270/Q2XG9SWD |
More data for this
Ligand-Target Pair | |
Nuclear receptor ROR-gamma (RORC)
(Homo sapiens (Human)) | BDBM50112367

(CHEMBL3609407)Show SMILES CCS(=O)(=O)c1ccc(CC(=O)Nc2cc(Cl)c(c(Cl)c2)-c2ccccc2OC(F)(F)F)cc1 |(-3.74,3.7,;-2.67,3.08,;-2.67,1.54,;-3.73,.92,;-3.74,2.16,;-1.33,.77,;,1.54,;1.33,.77,;1.33,-.77,;2.67,-1.54,;2.66,-3.08,;1.6,-3.7,;4,-3.86,;3.99,-5.4,;5.33,-6.17,;5.32,-7.71,;6.39,-8.33,;3.98,-8.48,;2.65,-7.7,;1.58,-8.32,;2.66,-6.16,;3.98,-10.02,;2.64,-10.78,;2.63,-12.32,;3.96,-13.1,;5.3,-12.34,;5.31,-10.8,;6.65,-10.03,;7.98,-10.81,;9.05,-10.21,;7.97,-12.05,;9.04,-11.44,;,-1.54,;-1.33,-.77,)| Show InChI InChI=1S/C23H18Cl2F3NO4S/c1-2-34(31,32)16-9-7-14(8-10-16)11-21(30)29-15-12-18(24)22(19(25)13-15)17-5-3-4-6-20(17)33-23(26,27)28/h3-10,12-13H,2,11H2,1H3,(H,29,30) | PDB
UniProtKB/SwissProt
UniProtKB/TrEMBL
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 34 | n/a | n/a | n/a | n/a | n/a | n/a |
Fudan University
Curated by ChEMBL
| Assay Description
Inverse agonist activity at APC-labeled RORgammat LBD (unknown origin) assessed as inhibition of N-(2-chloro-6-fluorobenzyl)-N-((20-methoxy-[1,10-bip... |
Eur J Med Chem 148: 465-476 (2018)
Article DOI: 10.1016/j.ejmech.2018.02.050
BindingDB Entry DOI: 10.7270/Q26Q20VP |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C19
(Homo sapiens (Human)) | BDBM50112367

(CHEMBL3609407)Show SMILES CCS(=O)(=O)c1ccc(CC(=O)Nc2cc(Cl)c(c(Cl)c2)-c2ccccc2OC(F)(F)F)cc1 |(-3.74,3.7,;-2.67,3.08,;-2.67,1.54,;-3.73,.92,;-3.74,2.16,;-1.33,.77,;,1.54,;1.33,.77,;1.33,-.77,;2.67,-1.54,;2.66,-3.08,;1.6,-3.7,;4,-3.86,;3.99,-5.4,;5.33,-6.17,;5.32,-7.71,;6.39,-8.33,;3.98,-8.48,;2.65,-7.7,;1.58,-8.32,;2.66,-6.16,;3.98,-10.02,;2.64,-10.78,;2.63,-12.32,;3.96,-13.1,;5.3,-12.34,;5.31,-10.8,;6.65,-10.03,;7.98,-10.81,;9.05,-10.21,;7.97,-12.05,;9.04,-11.44,;,-1.54,;-1.33,-.77,)| Show InChI InChI=1S/C23H18Cl2F3NO4S/c1-2-34(31,32)16-9-7-14(8-10-16)11-21(30)29-15-12-18(24)22(19(25)13-15)17-5-3-4-6-20(17)33-23(26,27)28/h3-10,12-13H,2,11H2,1H3,(H,29,30) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 9.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Advinus Therapeutics Limited
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C19 in human liver microsomes using (S)-mephenytoin as substrate preincubated for 5 mins followed by NADPH addition measured after ... |
Bioorg Med Chem Lett 29: 2208-2217 (2019)
Article DOI: 10.1016/j.bmcl.2019.06.044 |
More data for this
Ligand-Target Pair | |
Nuclear receptor ROR-gamma (RORC)
(Homo sapiens (Human)) | BDBM50112367

(CHEMBL3609407)Show SMILES CCS(=O)(=O)c1ccc(CC(=O)Nc2cc(Cl)c(c(Cl)c2)-c2ccccc2OC(F)(F)F)cc1 |(-3.74,3.7,;-2.67,3.08,;-2.67,1.54,;-3.73,.92,;-3.74,2.16,;-1.33,.77,;,1.54,;1.33,.77,;1.33,-.77,;2.67,-1.54,;2.66,-3.08,;1.6,-3.7,;4,-3.86,;3.99,-5.4,;5.33,-6.17,;5.32,-7.71,;6.39,-8.33,;3.98,-8.48,;2.65,-7.7,;1.58,-8.32,;2.66,-6.16,;3.98,-10.02,;2.64,-10.78,;2.63,-12.32,;3.96,-13.1,;5.3,-12.34,;5.31,-10.8,;6.65,-10.03,;7.98,-10.81,;9.05,-10.21,;7.97,-12.05,;9.04,-11.44,;,-1.54,;-1.33,-.77,)| Show InChI InChI=1S/C23H18Cl2F3NO4S/c1-2-34(31,32)16-9-7-14(8-10-16)11-21(30)29-15-12-18(24)22(19(25)13-15)17-5-3-4-6-20(17)33-23(26,27)28/h3-10,12-13H,2,11H2,1H3,(H,29,30) | PDB
UniProtKB/SwissProt
UniProtKB/TrEMBL
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
| | n/a | n/a | 35 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Nuclear receptor ROR-gamma (RORC)
(Homo sapiens (Human)) | BDBM50112367

(CHEMBL3609407)Show SMILES CCS(=O)(=O)c1ccc(CC(=O)Nc2cc(Cl)c(c(Cl)c2)-c2ccccc2OC(F)(F)F)cc1 |(-3.74,3.7,;-2.67,3.08,;-2.67,1.54,;-3.73,.92,;-3.74,2.16,;-1.33,.77,;,1.54,;1.33,.77,;1.33,-.77,;2.67,-1.54,;2.66,-3.08,;1.6,-3.7,;4,-3.86,;3.99,-5.4,;5.33,-6.17,;5.32,-7.71,;6.39,-8.33,;3.98,-8.48,;2.65,-7.7,;1.58,-8.32,;2.66,-6.16,;3.98,-10.02,;2.64,-10.78,;2.63,-12.32,;3.96,-13.1,;5.3,-12.34,;5.31,-10.8,;6.65,-10.03,;7.98,-10.81,;9.05,-10.21,;7.97,-12.05,;9.04,-11.44,;,-1.54,;-1.33,-.77,)| Show InChI InChI=1S/C23H18Cl2F3NO4S/c1-2-34(31,32)16-9-7-14(8-10-16)11-21(30)29-15-12-18(24)22(19(25)13-15)17-5-3-4-6-20(17)33-23(26,27)28/h3-10,12-13H,2,11H2,1H3,(H,29,30) | PDB
UniProtKB/SwissProt
UniProtKB/TrEMBL
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 112 | n/a | n/a | n/a | n/a | n/a | n/a |
Advinus Therapeutics Limited
Curated by ChEMBL
| Assay Description
Transactivation of recombinant GAL4-DBD fused RORgammat LBD (unknown origin) expressed in HEK293T cells incubated for 18 to 24 hrs by dual-Glo lucife... |
Bioorg Med Chem Lett 29: 2208-2217 (2019)
Article DOI: 10.1016/j.bmcl.2019.06.044 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C9
(Homo sapiens (Human)) | BDBM50112367

(CHEMBL3609407)Show SMILES CCS(=O)(=O)c1ccc(CC(=O)Nc2cc(Cl)c(c(Cl)c2)-c2ccccc2OC(F)(F)F)cc1 |(-3.74,3.7,;-2.67,3.08,;-2.67,1.54,;-3.73,.92,;-3.74,2.16,;-1.33,.77,;,1.54,;1.33,.77,;1.33,-.77,;2.67,-1.54,;2.66,-3.08,;1.6,-3.7,;4,-3.86,;3.99,-5.4,;5.33,-6.17,;5.32,-7.71,;6.39,-8.33,;3.98,-8.48,;2.65,-7.7,;1.58,-8.32,;2.66,-6.16,;3.98,-10.02,;2.64,-10.78,;2.63,-12.32,;3.96,-13.1,;5.3,-12.34,;5.31,-10.8,;6.65,-10.03,;7.98,-10.81,;9.05,-10.21,;7.97,-12.05,;9.04,-11.44,;,-1.54,;-1.33,-.77,)| Show InChI InChI=1S/C23H18Cl2F3NO4S/c1-2-34(31,32)16-9-7-14(8-10-16)11-21(30)29-15-12-18(24)22(19(25)13-15)17-5-3-4-6-20(17)33-23(26,27)28/h3-10,12-13H,2,11H2,1H3,(H,29,30) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 2.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Advinus Therapeutics Limited
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C9 in human liver microsomes using diclofenac as substrate preincubated for 5 mins followed by NADPH addition measured after 10 min... |
Bioorg Med Chem Lett 29: 2208-2217 (2019)
Article DOI: 10.1016/j.bmcl.2019.06.044 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM50112367

(CHEMBL3609407)Show SMILES CCS(=O)(=O)c1ccc(CC(=O)Nc2cc(Cl)c(c(Cl)c2)-c2ccccc2OC(F)(F)F)cc1 |(-3.74,3.7,;-2.67,3.08,;-2.67,1.54,;-3.73,.92,;-3.74,2.16,;-1.33,.77,;,1.54,;1.33,.77,;1.33,-.77,;2.67,-1.54,;2.66,-3.08,;1.6,-3.7,;4,-3.86,;3.99,-5.4,;5.33,-6.17,;5.32,-7.71,;6.39,-8.33,;3.98,-8.48,;2.65,-7.7,;1.58,-8.32,;2.66,-6.16,;3.98,-10.02,;2.64,-10.78,;2.63,-12.32,;3.96,-13.1,;5.3,-12.34,;5.31,-10.8,;6.65,-10.03,;7.98,-10.81,;9.05,-10.21,;7.97,-12.05,;9.04,-11.44,;,-1.54,;-1.33,-.77,)| Show InChI InChI=1S/C23H18Cl2F3NO4S/c1-2-34(31,32)16-9-7-14(8-10-16)11-21(30)29-15-12-18(24)22(19(25)13-15)17-5-3-4-6-20(17)33-23(26,27)28/h3-10,12-13H,2,11H2,1H3,(H,29,30) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Advinus Therapeutics Limited
Curated by ChEMBL
| Assay Description
Inhibition of CYP1A2 in human liver microsomes using phenacetin as substrate preincubated for 5 mins followed by NADPH addition measured after 10 min... |
Bioorg Med Chem Lett 29: 2208-2217 (2019)
Article DOI: 10.1016/j.bmcl.2019.06.044 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50112367

(CHEMBL3609407)Show SMILES CCS(=O)(=O)c1ccc(CC(=O)Nc2cc(Cl)c(c(Cl)c2)-c2ccccc2OC(F)(F)F)cc1 |(-3.74,3.7,;-2.67,3.08,;-2.67,1.54,;-3.73,.92,;-3.74,2.16,;-1.33,.77,;,1.54,;1.33,.77,;1.33,-.77,;2.67,-1.54,;2.66,-3.08,;1.6,-3.7,;4,-3.86,;3.99,-5.4,;5.33,-6.17,;5.32,-7.71,;6.39,-8.33,;3.98,-8.48,;2.65,-7.7,;1.58,-8.32,;2.66,-6.16,;3.98,-10.02,;2.64,-10.78,;2.63,-12.32,;3.96,-13.1,;5.3,-12.34,;5.31,-10.8,;6.65,-10.03,;7.98,-10.81,;9.05,-10.21,;7.97,-12.05,;9.04,-11.44,;,-1.54,;-1.33,-.77,)| Show InChI InChI=1S/C23H18Cl2F3NO4S/c1-2-34(31,32)16-9-7-14(8-10-16)11-21(30)29-15-12-18(24)22(19(25)13-15)17-5-3-4-6-20(17)33-23(26,27)28/h3-10,12-13H,2,11H2,1H3,(H,29,30) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Advinus Therapeutics Limited
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 in human liver microsomes using testosterone as substrate preincubated for 5 mins followed by NADPH addition measured after 10 m... |
Bioorg Med Chem Lett 29: 2208-2217 (2019)
Article DOI: 10.1016/j.bmcl.2019.06.044 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data