 Found 13 hits for monomerid = 50113332
Found 13 hits for monomerid = 50113332 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kcal/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
5-hydroxytryptamine receptor 2A
(Rattus norvegicus (rat)) | BDBM50113332
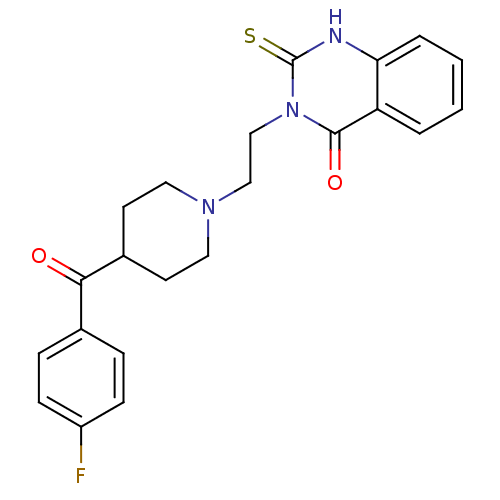
(3-(2-(4-(4-fluorobenzoyl)piperidin-1-yl)ethyl)-2-t...)Show SMILES Fc1ccc(cc1)C(=O)C1CCN(CCn2c(=S)[nH]c3ccccc3c2=O)CC1 Show InChI InChI=1S/C22H22FN3O2S/c23-17-7-5-15(6-8-17)20(27)16-9-11-25(12-10-16)13-14-26-21(28)18-3-1-2-4-19(18)24-22(26)29/h1-8,16H,9-14H2,(H,24,29) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.130 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute of Nuclear Chemistry Johannes Gutenberg-University Mainz
Curated by ChEMBL
| Assay Description
Displacement of [3H]altanserine from rat cortical membrane 5HT2A receptor |
Bioorg Med Chem 17: 2989-3002 (2009)
Article DOI: 10.1016/j.bmc.2009.03.021
BindingDB Entry DOI: 10.7270/Q21V5DW2 |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 2A
(Homo sapiens (Human)) | BDBM50113332

(3-(2-(4-(4-fluorobenzoyl)piperidin-1-yl)ethyl)-2-t...)Show SMILES Fc1ccc(cc1)C(=O)C1CCN(CCn2c(=S)[nH]c3ccccc3c2=O)CC1 Show InChI InChI=1S/C22H22FN3O2S/c23-17-7-5-15(6-8-17)20(27)16-9-11-25(12-10-16)13-14-26-21(28)18-3-1-2-4-19(18)24-22(26)29/h1-8,16H,9-14H2,(H,24,29) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.130 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Mainz
Curated by ChEMBL
| Assay Description
Binding affinity to 5HT2A receptor |
Bioorg Med Chem Lett 18: 1515-9 (2008)
Article DOI: 10.1016/j.bmcl.2007.12.054
BindingDB Entry DOI: 10.7270/Q23F4QHZ |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 2A
(Homo sapiens (Human)) | BDBM50113332

(3-(2-(4-(4-fluorobenzoyl)piperidin-1-yl)ethyl)-2-t...)Show SMILES Fc1ccc(cc1)C(=O)C1CCN(CCn2c(=S)[nH]c3ccccc3c2=O)CC1 Show InChI InChI=1S/C22H22FN3O2S/c23-17-7-5-15(6-8-17)20(27)16-9-11-25(12-10-16)13-14-26-21(28)18-3-1-2-4-19(18)24-22(26)29/h1-8,16H,9-14H2,(H,24,29) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yale University
Curated by ChEMBL
| Assay Description
Binding affinity against 5-hydroxytryptamine 2A receptor in humans |
J Med Chem 45: 2319-24 (2002)
BindingDB Entry DOI: 10.7270/Q2B27TM2 |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 2A
(Rattus norvegicus (rat)) | BDBM50113332

(3-(2-(4-(4-fluorobenzoyl)piperidin-1-yl)ethyl)-2-t...)Show SMILES Fc1ccc(cc1)C(=O)C1CCN(CCn2c(=S)[nH]c3ccccc3c2=O)CC1 Show InChI InChI=1S/C22H22FN3O2S/c23-17-7-5-15(6-8-17)20(27)16-9-11-25(12-10-16)13-14-26-21(28)18-3-1-2-4-19(18)24-22(26)29/h1-8,16H,9-14H2,(H,24,29) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.720 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute of Nuclear Chemistry Johannes Gutenberg-University Mainz
Curated by ChEMBL
| Assay Description
Displacement of [3H]MDL from rat 5HT2A receptor expressed in GF62 cells by liquid scintillation analyser |
Bioorg Med Chem 17: 2989-3002 (2009)
Article DOI: 10.1016/j.bmc.2009.03.021
BindingDB Entry DOI: 10.7270/Q21V5DW2 |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 2A
(Rattus norvegicus (rat)) | BDBM50113332

(3-(2-(4-(4-fluorobenzoyl)piperidin-1-yl)ethyl)-2-t...)Show SMILES Fc1ccc(cc1)C(=O)C1CCN(CCn2c(=S)[nH]c3ccccc3c2=O)CC1 Show InChI InChI=1S/C22H22FN3O2S/c23-17-7-5-15(6-8-17)20(27)16-9-11-25(12-10-16)13-14-26-21(28)18-3-1-2-4-19(18)24-22(26)29/h1-8,16H,9-14H2,(H,24,29) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.740 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Mainz
Curated by ChEMBL
| Assay Description
Displacement of [3H]MDL from rat 5HT2A receptor expressed in GF62 cells |
Bioorg Med Chem Lett 18: 1515-9 (2008)
Article DOI: 10.1016/j.bmcl.2007.12.054
BindingDB Entry DOI: 10.7270/Q23F4QHZ |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 2C
(Homo sapiens (Human)) | BDBM50113332

(3-(2-(4-(4-fluorobenzoyl)piperidin-1-yl)ethyl)-2-t...)Show SMILES Fc1ccc(cc1)C(=O)C1CCN(CCn2c(=S)[nH]c3ccccc3c2=O)CC1 Show InChI InChI=1S/C22H22FN3O2S/c23-17-7-5-15(6-8-17)20(27)16-9-11-25(12-10-16)13-14-26-21(28)18-3-1-2-4-19(18)24-22(26)29/h1-8,16H,9-14H2,(H,24,29) | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute of Nuclear Chemistry Johannes Gutenberg-University Mainz
Curated by ChEMBL
| Assay Description
Binding affinity to 5HT2C receptor (unknown origin) by competitive binding experiment |
Bioorg Med Chem 17: 2989-3002 (2009)
Article DOI: 10.1016/j.bmc.2009.03.021
BindingDB Entry DOI: 10.7270/Q21V5DW2 |
More data for this
Ligand-Target Pair | |
D(2) dopamine receptor
(Homo sapiens (Human)) | BDBM50113332

(3-(2-(4-(4-fluorobenzoyl)piperidin-1-yl)ethyl)-2-t...)Show SMILES Fc1ccc(cc1)C(=O)C1CCN(CCn2c(=S)[nH]c3ccccc3c2=O)CC1 Show InChI InChI=1S/C22H22FN3O2S/c23-17-7-5-15(6-8-17)20(27)16-9-11-25(12-10-16)13-14-26-21(28)18-3-1-2-4-19(18)24-22(26)29/h1-8,16H,9-14H2,(H,24,29) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 62 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute of Nuclear Chemistry Johannes Gutenberg-University Mainz
Curated by ChEMBL
| Assay Description
Binding affinity to dopamine D2 receptor (unknown origin) by competitive binding experiment |
Bioorg Med Chem 17: 2989-3002 (2009)
Article DOI: 10.1016/j.bmc.2009.03.021
BindingDB Entry DOI: 10.7270/Q21V5DW2 |
More data for this
Ligand-Target Pair | |
adrenergic Alpha2
(RAT) | BDBM50113332

(3-(2-(4-(4-fluorobenzoyl)piperidin-1-yl)ethyl)-2-t...)Show SMILES Fc1ccc(cc1)C(=O)C1CCN(CCn2c(=S)[nH]c3ccccc3c2=O)CC1 Show InChI InChI=1S/C22H22FN3O2S/c23-17-7-5-15(6-8-17)20(27)16-9-11-25(12-10-16)13-14-26-21(28)18-3-1-2-4-19(18)24-22(26)29/h1-8,16H,9-14H2,(H,24,29) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 71 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Janssen Pharmaceutica
Curated by PDSP Ki Database
| |
Eur J Pharmacol 129: 49-55 (1986)
Article DOI: 10.1016/0014-2999(86)90335-3
BindingDB Entry DOI: 10.7270/Q27S7M8D |
More data for this
Ligand-Target Pair | |
adrenergic Alpha2
(RAT) | BDBM50113332

(3-(2-(4-(4-fluorobenzoyl)piperidin-1-yl)ethyl)-2-t...)Show SMILES Fc1ccc(cc1)C(=O)C1CCN(CCn2c(=S)[nH]c3ccccc3c2=O)CC1 Show InChI InChI=1S/C22H22FN3O2S/c23-17-7-5-15(6-8-17)20(27)16-9-11-25(12-10-16)13-14-26-21(28)18-3-1-2-4-19(18)24-22(26)29/h1-8,16H,9-14H2,(H,24,29) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Janssen Pharmaceutica
Curated by PDSP Ki Database
| |
Eur J Pharmacol 129: 49-55 (1986)
Article DOI: 10.1016/0014-2999(86)90335-3
BindingDB Entry DOI: 10.7270/Q27S7M8D |
More data for this
Ligand-Target Pair | |
adrenergic Alpha2
(RAT) | BDBM50113332

(3-(2-(4-(4-fluorobenzoyl)piperidin-1-yl)ethyl)-2-t...)Show SMILES Fc1ccc(cc1)C(=O)C1CCN(CCn2c(=S)[nH]c3ccccc3c2=O)CC1 Show InChI InChI=1S/C22H22FN3O2S/c23-17-7-5-15(6-8-17)20(27)16-9-11-25(12-10-16)13-14-26-21(28)18-3-1-2-4-19(18)24-22(26)29/h1-8,16H,9-14H2,(H,24,29) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 280 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Janssen Pharmaceutica
Curated by PDSP Ki Database
| |
Eur J Pharmacol 129: 49-55 (1986)
Article DOI: 10.1016/0014-2999(86)90335-3
BindingDB Entry DOI: 10.7270/Q27S7M8D |
More data for this
Ligand-Target Pair | |
adrenergic Alpha2
(RAT) | BDBM50113332

(3-(2-(4-(4-fluorobenzoyl)piperidin-1-yl)ethyl)-2-t...)Show SMILES Fc1ccc(cc1)C(=O)C1CCN(CCn2c(=S)[nH]c3ccccc3c2=O)CC1 Show InChI InChI=1S/C22H22FN3O2S/c23-17-7-5-15(6-8-17)20(27)16-9-11-25(12-10-16)13-14-26-21(28)18-3-1-2-4-19(18)24-22(26)29/h1-8,16H,9-14H2,(H,24,29) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 290 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Janssen Pharmaceutica
Curated by PDSP Ki Database
| |
Eur J Pharmacol 129: 49-55 (1986)
Article DOI: 10.1016/0014-2999(86)90335-3
BindingDB Entry DOI: 10.7270/Q27S7M8D |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1A
(Homo sapiens (Human)) | BDBM50113332

(3-(2-(4-(4-fluorobenzoyl)piperidin-1-yl)ethyl)-2-t...)Show SMILES Fc1ccc(cc1)C(=O)C1CCN(CCn2c(=S)[nH]c3ccccc3c2=O)CC1 Show InChI InChI=1S/C22H22FN3O2S/c23-17-7-5-15(6-8-17)20(27)16-9-11-25(12-10-16)13-14-26-21(28)18-3-1-2-4-19(18)24-22(26)29/h1-8,16H,9-14H2,(H,24,29) | PDB
UniProtKB/SwissProt
UniProtKB/TrEMBL
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.57E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute of Nuclear Chemistry Johannes Gutenberg-University Mainz
Curated by ChEMBL
| Assay Description
Binding affinity to 5-HT1A receptor (unknown origin) by competitive binding experiment |
Bioorg Med Chem 17: 2989-3002 (2009)
Article DOI: 10.1016/j.bmc.2009.03.021
BindingDB Entry DOI: 10.7270/Q21V5DW2 |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 2A
(Homo sapiens (Human)) | BDBM50113332

(3-(2-(4-(4-fluorobenzoyl)piperidin-1-yl)ethyl)-2-t...)Show SMILES Fc1ccc(cc1)C(=O)C1CCN(CCn2c(=S)[nH]c3ccccc3c2=O)CC1 Show InChI InChI=1S/C22H22FN3O2S/c23-17-7-5-15(6-8-17)20(27)16-9-11-25(12-10-16)13-14-26-21(28)18-3-1-2-4-19(18)24-22(26)29/h1-8,16H,9-14H2,(H,24,29) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 1.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Dominican College
Curated by ChEMBL
| Assay Description
Antagonist activity at recombinant human 5-HT2A expressed in human U2OS cells by pathhunter beta-arrestin assay |
Bioorg Med Chem Lett 30: (2020)
|
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data