

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50135842 Bicyclic Guanidinium Subunit::CHEMBL386994
SMILES: CC[C@H](C)[C@H](NC(=O)CNC(=O)CSCC1CCN2CCC(CO[Si](c3ccccc3)(c3ccccc3)C(C)(C)C)[NH+]=C2N1)C(=O)N[C@@H](CO)C(=O)N[C@@H](Cc1ccc(O)cc1)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CC(C)C)C(=O)OC
InChI Key: InChIKey=RVOHKMYQSGAMKR-NVQNAHMTSA-O
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Gag-Pol polyprotein [489-587] (Human immunodeficiency virus type 1) | BDBM50135842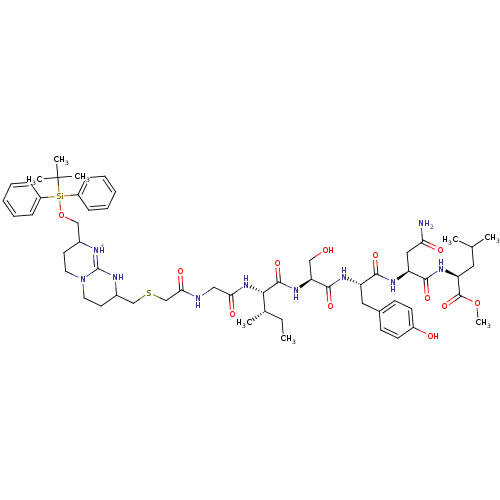 (Bicyclic Guanidinium Subunit | CHEMBL386994) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 6.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universidad Aut£noma de Madrid Curated by ChEMBL | Assay Description Competitive inhibition against HIV-1 Protease | J Med Chem 46: 5196-207 (2003) Article DOI: 10.1021/jm030871u BindingDB Entry DOI: 10.7270/Q2C53MKZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gag-Pol polyprotein [489-587] (Human immunodeficiency virus type 1) | BDBM50135842 (Bicyclic Guanidinium Subunit | CHEMBL386994) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 5.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Universidad Aut£noma de Madrid Curated by ChEMBL | Assay Description In vitro inhibitory activity against HIV-1 Protease | J Med Chem 46: 5196-207 (2003) Article DOI: 10.1021/jm030871u BindingDB Entry DOI: 10.7270/Q2C53MKZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||