

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50135933 3-(6-Amino-pyridin-3-yl)-2-(1H-imidazol-4-yl)-propionic acid::CHEMBL302067
SMILES: Nc1ccc(CC(C(O)=O)c2cnc[nH]2)cn1
InChI Key: InChIKey=IFHWJTOFROTZBD-UHFFFAOYSA-N
Data: 9 IC50
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Carboxypeptidase B2 (Homo sapiens (Human)) | BDBM50135933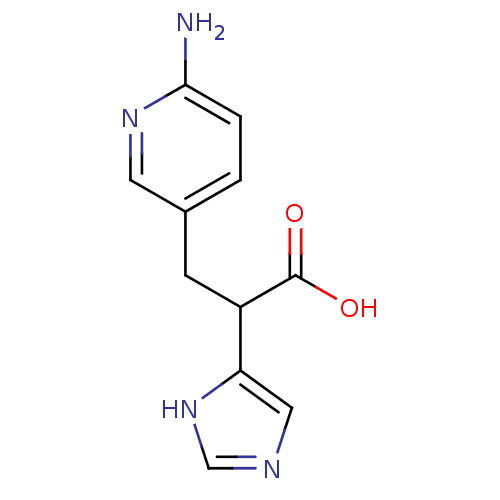 (3-(6-Amino-pyridin-3-yl)-2-(1H-imidazol-4-yl)-prop...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 80 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories Curated by ChEMBL | Assay Description Inhibitory potency against human TAFIa (thrombin-activatable fibrinolysis inhibitor) | Bioorg Med Chem Lett 14: 2141-5 (2004) Article DOI: 10.1016/j.bmcl.2004.02.033 BindingDB Entry DOI: 10.7270/Q2N8797S | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carboxypeptidase B2 (Homo sapiens (Human)) | BDBM50135933 (3-(6-Amino-pyridin-3-yl)-2-(1H-imidazol-4-yl)-prop...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca Curated by ChEMBL | Assay Description Inhibition of human TAF1a using hippuryl-L-arginine/hippuryl-L-lysine as substrate by liquid chromatographic analysis | Bioorg Med Chem 22: 2261-8 (2014) Article DOI: 10.1016/j.bmc.2014.02.010 BindingDB Entry DOI: 10.7270/Q2DZ09TG | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carboxypeptidase B2 (Homo sapiens (Human)) | BDBM50135933 (3-(6-Amino-pyridin-3-yl)-2-(1H-imidazol-4-yl)-prop...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 3.10E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca Curated by ChEMBL | Assay Description Inhibition of TAF1a in human plasma assessed as clot lysis | Bioorg Med Chem 22: 2261-8 (2014) Article DOI: 10.1016/j.bmc.2014.02.010 BindingDB Entry DOI: 10.7270/Q2DZ09TG | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carboxypeptidase B2 (Homo sapiens (Human)) | BDBM50135933 (3-(6-Amino-pyridin-3-yl)-2-(1H-imidazol-4-yl)-prop...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 67 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories Curated by ChEMBL | Assay Description In vitro inhibition of purified Carboxypeptidase B (CPB) by clot lysis assay in human plasma | J Med Chem 46: 5294-7 (2003) Article DOI: 10.1021/jm034141y BindingDB Entry DOI: 10.7270/Q27H1J0Z | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carboxypeptidase B (Homo sapiens (Human)) | BDBM50135933 (3-(6-Amino-pyridin-3-yl)-2-(1H-imidazol-4-yl)-prop...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 70 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories Curated by ChEMBL | Assay Description Inhibition of human carboxypeptidase B (CPB) | Bioorg Med Chem Lett 14: 2141-5 (2004) Article DOI: 10.1016/j.bmcl.2004.02.033 BindingDB Entry DOI: 10.7270/Q2N8797S | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carboxypeptidase M (Homo sapiens (Human)) | BDBM50135933 (3-(6-Amino-pyridin-3-yl)-2-(1H-imidazol-4-yl)-prop...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories Curated by ChEMBL | Assay Description In vitro inhibition of purified Carboxypeptidase M (CPM) by clot lysis assay in human plasma | J Med Chem 46: 5294-7 (2003) Article DOI: 10.1021/jm034141y BindingDB Entry DOI: 10.7270/Q27H1J0Z | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carboxypeptidase A1 (Homo sapiens (Human)) | BDBM50135933 (3-(6-Amino-pyridin-3-yl)-2-(1H-imidazol-4-yl)-prop...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories Curated by ChEMBL | Assay Description In vitro inhibition of Carboxypeptidase A (CPA) was determined by clot lysis assay using human plasma | J Med Chem 46: 5294-7 (2003) Article DOI: 10.1021/jm034141y BindingDB Entry DOI: 10.7270/Q27H1J0Z | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carboxypeptidase M (Homo sapiens (Human)) | BDBM50135933 (3-(6-Amino-pyridin-3-yl)-2-(1H-imidazol-4-yl)-prop...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories Curated by ChEMBL | Assay Description In vitro inhibition of purified Carboxypeptidase M (CPM) by clot lysis assay in human plasma | J Med Chem 46: 5294-7 (2003) Article DOI: 10.1021/jm034141y BindingDB Entry DOI: 10.7270/Q27H1J0Z | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carboxypeptidase A1 (Homo sapiens (Human)) | BDBM50135933 (3-(6-Amino-pyridin-3-yl)-2-(1H-imidazol-4-yl)-prop...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories Curated by ChEMBL | Assay Description In vitro inhibition of Carboxypeptidase A (CPA) was determined by clot lysis assay using human plasma | J Med Chem 46: 5294-7 (2003) Article DOI: 10.1021/jm034141y BindingDB Entry DOI: 10.7270/Q27H1J0Z | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||