

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50182153 4-[7-bromo-3-(3-methyl-but-2-enyl)-3H-benzoimidazole-5-sulfonyl]-5-methylsulfanyl-thiophene-2-carboxamidine::CHEMBL380315
SMILES: [#6]-[#16]-c1sc(cc1S(=O)(=O)c1cc(Br)c2ncn(-[#6]\[#6]=[#6](\[#6])-[#6])c2c1)-[#6](-[#7])=[#7]
InChI Key: InChIKey=QWUKQOSMVRSUGE-UHFFFAOYSA-N
Data: 1 KI
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Complement C1s (Homo sapiens (Human)) | BDBM50182153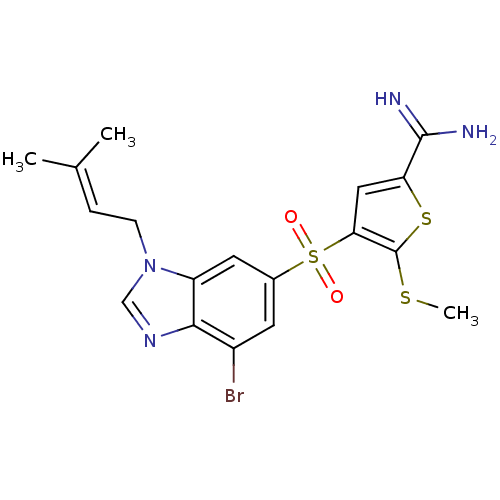 (4-[7-bromo-3-(3-methyl-but-2-enyl)-3H-benzoimidazo...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson & Johnson Pharmaceutical Research and Development Curated by ChEMBL | Assay Description Inhibition of C1S | Bioorg Med Chem Lett 16: 2200-4 (2006) Article DOI: 10.1016/j.bmcl.2006.01.036 BindingDB Entry DOI: 10.7270/Q2251HSV | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||