 Found 3 hits for monomerid = 50198309
Found 3 hits for monomerid = 50198309 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kcal/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
kinesin spindle protein
(Homo sapiens (Human)) | BDBM50198309
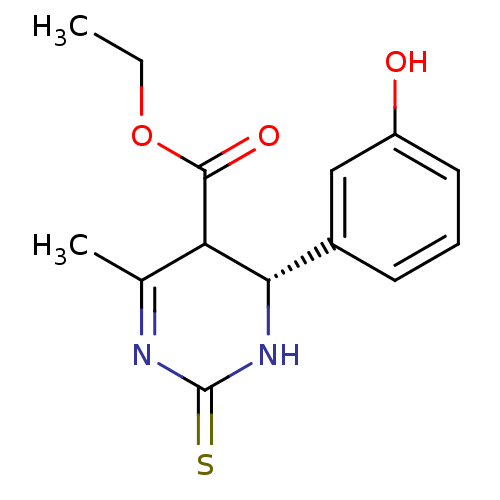
((S)-4-(3-Hydroxy-phenyl)-6-methyl-2-thioxo-1,2,3,4...)Show SMILES CCOC(=O)C1[C@@H](NC(=S)N=C1C)c1cccc(O)c1 |r,c:10| Show InChI InChI=1S/C14H16N2O3S/c1-3-19-13(18)11-8(2)15-14(20)16-12(11)9-5-4-6-10(17)7-9/h4-7,11-12,17H,3H2,1-2H3,(H,16,20)/t11?,12-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| n/a | n/a | 2.07E+4 | n/a | n/a | n/a | n/a | 6.8 | 25 |
University of Leipzig
| Assay Description
The ATPase activity of the Eg5 motor domain was measured by using the malachite green assay. |
Chembiochem 6: 2005-13 (2005)
Article DOI: 10.1002/cbic.200500168
BindingDB Entry DOI: 10.7270/Q2XW4HB5 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Kinesin-like protein 1
(Homo sapiens (Human)) | BDBM50198309

((S)-4-(3-Hydroxy-phenyl)-6-methyl-2-thioxo-1,2,3,4...)Show SMILES CCOC(=O)C1[C@@H](NC(=S)N=C1C)c1cccc(O)c1 |r,c:10| Show InChI InChI=1S/C14H16N2O3S/c1-3-19-13(18)11-8(2)15-14(20)16-12(11)9-5-4-6-10(17)7-9/h4-7,11-12,17H,3H2,1-2H3,(H,16,20)/t11?,12-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| n/a | n/a | 1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University
Curated by ChEMBL
| Assay Description
Inhibition of KSP |
Bioorg Med Chem Lett 17: 722-6 (2007)
Article DOI: 10.1016/j.bmcl.2006.10.083
BindingDB Entry DOI: 10.7270/Q2HT2Q4S |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Kinesin-like protein 1
(Homo sapiens (Human)) | BDBM50198309

((S)-4-(3-Hydroxy-phenyl)-6-methyl-2-thioxo-1,2,3,4...)Show SMILES CCOC(=O)C1[C@@H](NC(=S)N=C1C)c1cccc(O)c1 |r,c:10| Show InChI InChI=1S/C14H16N2O3S/c1-3-19-13(18)11-8(2)15-14(20)16-12(11)9-5-4-6-10(17)7-9/h4-7,11-12,17H,3H2,1-2H3,(H,16,20)/t11?,12-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| n/a | n/a | 1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
In vitro inhibitory concentration towards kinesin spindle protein activity of ATP hydrolysis in the presence of microtubules measured by ATPase assay... |
Bioorg Med Chem Lett 15: 2041-5 (2005)
Article DOI: 10.1016/j.bmcl.2005.02.055
BindingDB Entry DOI: 10.7270/Q2ST7PCW |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data