

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50294168 1-(2-(1H-indol-5-yloxy)ethyl)piperidine-4-carboxylic acid::1-[2-(1H-indol-5-yloxy)ethyl]piperidine-4-carboxylic acid::CHEMBL562164
SMILES: OC(=O)C1CCN(CCOc2ccc3[nH]ccc3c2)CC1
InChI Key: InChIKey=WEUOMMCSMCVWHD-UHFFFAOYSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Leukotriene A4 hydrolase (Homo sapiens (Human)) | BDBM50294168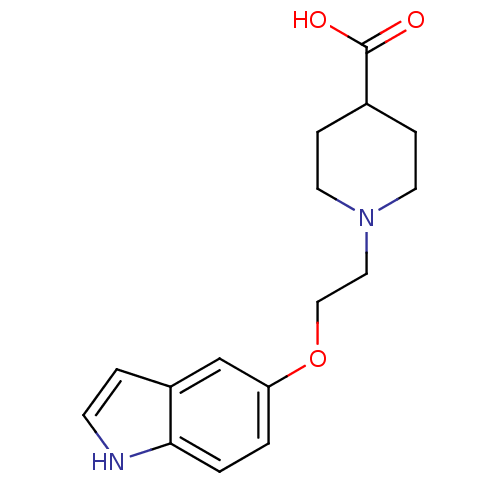 (1-(2-(1H-indol-5-yloxy)ethyl)piperidine-4-carboxyl...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem | MMDB PDB Article PubMed | n/a | n/a | 9.66E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
deCODE biostructures, Inc. Curated by ChEMBL | Assay Description Inhibition of hydrolase activity of human recombinant LTA4H expressed in Escherichia coli BL21-AI/pRARE assessed as LTB4 formation by tandem quadrupo... | J Med Chem 52: 4694-715 (2009) Article DOI: 10.1021/jm900259h BindingDB Entry DOI: 10.7270/Q2F47P6N | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Leukotriene A4 hydrolase (Homo sapiens (Human)) | BDBM50294168 (1-(2-(1H-indol-5-yloxy)ethyl)piperidine-4-carboxyl...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem | MMDB PDB Article PubMed | n/a | n/a | 1.49E+6 | n/a | n/a | n/a | n/a | n/a | n/a |
deCODE biostructures, Inc. Curated by ChEMBL | Assay Description Inhibition of peptidase activity of human recombinant LTA4H expressed in Escherichia coli BL21-AI/pRARE | J Med Chem 52: 4694-715 (2009) Article DOI: 10.1021/jm900259h BindingDB Entry DOI: 10.7270/Q2F47P6N | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||