

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50436358 CHEMBL2396718
SMILES: CCCNC(=O)COc1ccc(cc1)-c1oc2cc(O)c(cc2c1C#Cc1cccc(Cl)c1)C(O)=O
InChI Key: InChIKey=ZAKVHDWXAFAMTH-UHFFFAOYSA-N
Data: 19 IC50
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Low molecular weight phosphotyrosine protein phosphatase (Homo sapiens (Human)) | BDBM50436358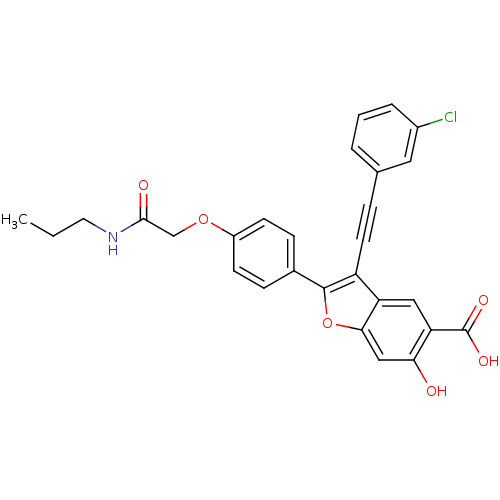 (CHEMBL2396718) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 340 | n/a | n/a | n/a | n/a | n/a | n/a |
Indiana University School of Medicine Curated by ChEMBL | Assay Description Inhibition of recombinant LMWPTP (unknown origin) using pNPP as substrate by spectrophotometric analysis | J Med Chem 56: 4990-5008 (2013) Article DOI: 10.1021/jm400248c BindingDB Entry DOI: 10.7270/Q2N017XW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Hematopoietic cell protein-tyrosine phosphatase 70Z-PEP (Homo sapiens (Human)) | BDBM50436358 (CHEMBL2396718) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 171 | n/a | n/a | n/a | n/a | n/a | n/a |
Indiana University School of Medicine Curated by ChEMBL | Assay Description Inhibition of N-terminal 6xHis-tagged LYP catalytic domain (1 to 303) (unknown origin) expressed in Escherichia coli BL21(DE3) using pNPP as substrat... | J Med Chem 56: 4990-5008 (2013) Article DOI: 10.1021/jm400248c BindingDB Entry DOI: 10.7270/Q2N017XW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Dual specificity protein phosphatase 15 (Homo sapiens (Human)) | BDBM50436358 (CHEMBL2396718) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.90E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Indiana University School of Medicine Curated by ChEMBL | Assay Description Inhibition of recombinant VHX (unknown origin) using pNPP as substrate by spectrophotometric analysis | J Med Chem 56: 4990-5008 (2013) Article DOI: 10.1021/jm400248c BindingDB Entry DOI: 10.7270/Q2N017XW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Dual specificity protein phosphatase (VHR) (Homo sapiens (Human)) | BDBM50436358 (CHEMBL2396718) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 5.90E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Indiana University School of Medicine Curated by ChEMBL | Assay Description Inhibition of recombinant VHR (unknown origin) using pNPP as substrate by spectrophotometric analysis | J Med Chem 56: 4990-5008 (2013) Article DOI: 10.1021/jm400248c BindingDB Entry DOI: 10.7270/Q2N017XW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Receptor-type tyrosine-protein phosphatase mu (PTPμ) (Homo sapiens (Human)) | BDBM50436358 (CHEMBL2396718) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 7.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Indiana University School of Medicine Curated by ChEMBL | Assay Description Inhibition of recombinant PTPmu (unknown origin) using pNPP as substrate by spectrophotometric analysis | J Med Chem 56: 4990-5008 (2013) Article DOI: 10.1021/jm400248c BindingDB Entry DOI: 10.7270/Q2N017XW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Receptor-type tyrosine-protein phosphatase gamma (Homo sapiens (Human)) | BDBM50436358 (CHEMBL2396718) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 4.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Indiana University School of Medicine Curated by ChEMBL | Assay Description Inhibition of recombinant PTPgamma (unknown origin) using pNPP as substrate by spectrophotometric analysis | J Med Chem 56: 4990-5008 (2013) Article DOI: 10.1021/jm400248c BindingDB Entry DOI: 10.7270/Q2N017XW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Receptor-type tyrosine-protein phosphatase epsilon (PTPε) (Homo sapiens (Human)) | BDBM50436358 (CHEMBL2396718) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >2.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Indiana University School of Medicine Curated by ChEMBL | Assay Description Inhibition of recombinant PTPepsilon (unknown origin) using pNPP as substrate by spectrophotometric analysis | J Med Chem 56: 4990-5008 (2013) Article DOI: 10.1021/jm400248c BindingDB Entry DOI: 10.7270/Q2N017XW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Receptor-type tyrosine-protein phosphatase beta (PTPβ) (Homo sapiens (Human)) | BDBM50436358 (CHEMBL2396718) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 470 | n/a | n/a | n/a | n/a | n/a | n/a |
Indiana University School of Medicine Curated by ChEMBL | Assay Description Inhibition of recombinant PTPbeta (unknown origin) using pNPP as substrate by spectrophotometric analysis | J Med Chem 56: 4990-5008 (2013) Article DOI: 10.1021/jm400248c BindingDB Entry DOI: 10.7270/Q2N017XW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Leukocyte common antigen (Homo sapiens (Human)) | BDBM50436358 (CHEMBL2396718) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 3.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Indiana University School of Medicine Curated by ChEMBL | Assay Description Inhibition of recombinant CD45 (unknown origin) using pNPP as substrate by spectrophotometric analysis | J Med Chem 56: 4990-5008 (2013) Article DOI: 10.1021/jm400248c BindingDB Entry DOI: 10.7270/Q2N017XW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Tyrosine-protein phosphatase non-receptor type 3 (PTPH1) (Homo sapiens (Human)) | BDBM50436358 (CHEMBL2396718) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Indiana University School of Medicine Curated by ChEMBL | Assay Description Inhibition of recombinant PTPH1 (unknown origin) using pNPP as substrate by spectrophotometric analysis | J Med Chem 56: 4990-5008 (2013) Article DOI: 10.1021/jm400248c BindingDB Entry DOI: 10.7270/Q2N017XW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Tyrosine-protein phosphatase non-receptor type 12 (PTP-PEST) (Homo sapiens (Human)) | BDBM50436358 (CHEMBL2396718) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 800 | n/a | n/a | n/a | n/a | n/a | n/a |
Indiana University School of Medicine Curated by ChEMBL | Assay Description Inhibition of recombinant PTP-PEST (unknown origin) using pNPP as substrate by spectrophotometric analysis | J Med Chem 56: 4990-5008 (2013) Article DOI: 10.1021/jm400248c BindingDB Entry DOI: 10.7270/Q2N017XW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Fas-associated protein-tyrosine phosphatase 1 (FAP1) (Homo sapiens (Human)) | BDBM50436358 (CHEMBL2396718) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 390 | n/a | n/a | n/a | n/a | n/a | n/a |
Indiana University School of Medicine Curated by ChEMBL | Assay Description Inhibition of recombinant FAP-1 (unknown origin) using pNPP as substrate by spectrophotometric analysis | J Med Chem 56: 4990-5008 (2013) Article DOI: 10.1021/jm400248c BindingDB Entry DOI: 10.7270/Q2N017XW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Protein-tyrosine phosphatase MEG2 (PTP-Meg2) (Homo sapiens (Human)) | BDBM50436358 (CHEMBL2396718) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 590 | n/a | n/a | n/a | n/a | n/a | n/a |
Indiana University School of Medicine Curated by ChEMBL | Assay Description Inhibition of recombinant PTP-MEG2 (unknown origin) using pNPP as substrate by spectrophotometric analysis | J Med Chem 56: 4990-5008 (2013) Article DOI: 10.1021/jm400248c BindingDB Entry DOI: 10.7270/Q2N017XW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Hematopoietic protein-tyrosine phosphatase (HEPTP) (Homo sapiens (Human)) | BDBM50436358 (CHEMBL2396718) | PDB KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 3.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Indiana University School of Medicine Curated by ChEMBL | Assay Description Inhibition of recombinant HEPTP (unknown origin) using pNPP as substrate by spectrophotometric analysis | J Med Chem 56: 4990-5008 (2013) Article DOI: 10.1021/jm400248c BindingDB Entry DOI: 10.7270/Q2N017XW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Tyrosine-protein phosphatase non-receptor type 2 (Homo sapiens (Human)) | BDBM50436358 (CHEMBL2396718) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.13E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Indiana University School of Medicine Curated by ChEMBL | Assay Description Inhibition of recombinant TCPTP (unknown origin) using pNPP as substrate by spectrophotometric analysis | J Med Chem 56: 4990-5008 (2013) Article DOI: 10.1021/jm400248c BindingDB Entry DOI: 10.7270/Q2N017XW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Tyrosine-protein phosphatase non-receptor type 11 (Homo sapiens (Human)) | BDBM50436358 (CHEMBL2396718) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 560 | n/a | n/a | n/a | n/a | n/a | n/a |
Indiana University School of Medicine Curated by ChEMBL | Assay Description Inhibition of recombinant SHP-2 (unknown origin) using pNPP as substrate by spectrophotometric analysis | J Med Chem 56: 4990-5008 (2013) Article DOI: 10.1021/jm400248c BindingDB Entry DOI: 10.7270/Q2N017XW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Protein-tyrosine phosphatase 1C (Homo sapiens (Human)) | BDBM50436358 (CHEMBL2396718) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 910 | n/a | n/a | n/a | n/a | n/a | n/a |
Indiana University School of Medicine Curated by ChEMBL | Assay Description Inhibition of recombinant SHP-1 (unknown origin) using pNPP as substrate by spectrophotometric analysis | J Med Chem 56: 4990-5008 (2013) Article DOI: 10.1021/jm400248c BindingDB Entry DOI: 10.7270/Q2N017XW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Protein-tyrosine phosphatase 1B (Homo sapiens (Human)) | BDBM50436358 (CHEMBL2396718) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Indiana University School of Medicine Curated by ChEMBL | Assay Description Inhibition of recombinant PTP1B (unknown origin) using pNPP as substrate by spectrophotometric analysis | J Med Chem 56: 4990-5008 (2013) Article DOI: 10.1021/jm400248c BindingDB Entry DOI: 10.7270/Q2N017XW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Dual specificity protein phosphatase 23 (VHZ) (Homo sapiens (Human)) | BDBM50436358 (CHEMBL2396718) | PDB UniProtKB/SwissProt UniProtKB/TrEMBL antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 3.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Indiana University School of Medicine Curated by ChEMBL | Assay Description Inhibition of recombinant VHZ (unknown origin) using pNPP as substrate by spectrophotometric analysis | J Med Chem 56: 4990-5008 (2013) Article DOI: 10.1021/jm400248c BindingDB Entry DOI: 10.7270/Q2N017XW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||