 Found 15 hits for monomerid = 50530251
Found 15 hits for monomerid = 50530251 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kcal/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
1,3-beta-glucan synthase component GLS2
(Saccharomyces cerevisiae) | BDBM50530251
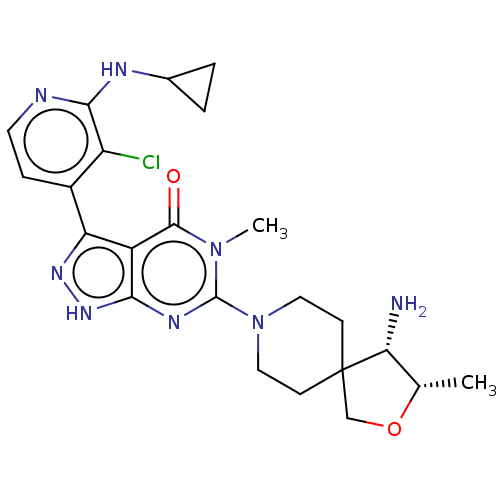
(CHEMBL4439006 | US10975080, Example 75)Show SMILES C[C@@H]1OCC2(CCN(CC2)c2nc3[nH]nc(-c4ccnc(NC5CC5)c4Cl)c3c(=O)n2C)[C@@H]1N |r| Show InChI InChI=1S/C23H29ClN8O2/c1-12-18(25)23(11-34-12)6-9-32(10-7-23)22-28-19-15(21(33)31(22)2)17(29-30-19)14-5-8-26-20(16(14)24)27-13-3-4-13/h5,8,12-13,18H,3-4,6-7,9-11,25H2,1-2H3,(H,26,27)(H,29,30)/t12-,18+/m0/s1 | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.70E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibition of human ERG |
J Med Chem 62: 1781-1792 (2019)
Article DOI: 10.1021/acs.jmedchem.8b01725 |
More data for this
Ligand-Target Pair | |
Protein-tyrosine phosphatase 2C
(Homo sapiens (Human)) | BDBM50530251

(CHEMBL4439006 | US10975080, Example 75)Show SMILES C[C@@H]1OCC2(CCN(CC2)c2nc3[nH]nc(-c4ccnc(NC5CC5)c4Cl)c3c(=O)n2C)[C@@H]1N |r| Show InChI InChI=1S/C23H29ClN8O2/c1-12-18(25)23(11-34-12)6-9-32(10-7-23)22-28-19-15(21(33)31(22)2)17(29-30-19)14-5-8-26-20(16(14)24)27-13-3-4-13/h5,8,12-13,18H,3-4,6-7,9-11,25H2,1-2H3,(H,26,27)(H,29,30)/t12-,18+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 36 | n/a | n/a | n/a | n/a | n/a | n/a |
NOVARTIS AG
US Patent
| Assay Description
SHP2 is allosterically activated through binding of bis-tyrosyl-phosphorylated peptides to its Src Homology 2 (SH2) domains. The latter activation st... |
US Patent US10975080 (2021)
|
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 11
(Homo sapiens) | BDBM50530251

(CHEMBL4439006 | US10975080, Example 75)Show SMILES C[C@@H]1OCC2(CCN(CC2)c2nc3[nH]nc(-c4ccnc(NC5CC5)c4Cl)c3c(=O)n2C)[C@@H]1N |r| Show InChI InChI=1S/C23H29ClN8O2/c1-12-18(25)23(11-34-12)6-9-32(10-7-23)22-28-19-15(21(33)31(22)2)17(29-30-19)14-5-8-26-20(16(14)24)27-13-3-4-13/h5,8,12-13,18H,3-4,6-7,9-11,25H2,1-2H3,(H,26,27)(H,29,30)/t12-,18+/m0/s1 | PDB
KEGG
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 36 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibition of of SHP2 in human KYSE520 cells assessed as suppression of ERK phosphorylation after 2 hrs by AlphaScreen SureFire Phospho-ERK1/2 assay |
J Med Chem 62: 1781-1792 (2019)
Article DOI: 10.1021/acs.jmedchem.8b01725 |
More data for this
Ligand-Target Pair | |
1,3-beta-glucan synthase component GLS2
(Saccharomyces cerevisiae) | BDBM50530251

(CHEMBL4439006 | US10975080, Example 75)Show SMILES C[C@@H]1OCC2(CCN(CC2)c2nc3[nH]nc(-c4ccnc(NC5CC5)c4Cl)c3c(=O)n2C)[C@@H]1N |r| Show InChI InChI=1S/C23H29ClN8O2/c1-12-18(25)23(11-34-12)6-9-32(10-7-23)22-28-19-15(21(33)31(22)2)17(29-30-19)14-5-8-26-20(16(14)24)27-13-3-4-13/h5,8,12-13,18H,3-4,6-7,9-11,25H2,1-2H3,(H,26,27)(H,29,30)/t12-,18+/m0/s1 | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.70E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibition of human ERG by Q-patch assay |
J Med Chem 62: 1781-1792 (2019)
Article DOI: 10.1021/acs.jmedchem.8b01725 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50530251

(CHEMBL4439006 | US10975080, Example 75)Show SMILES C[C@@H]1OCC2(CCN(CC2)c2nc3[nH]nc(-c4ccnc(NC5CC5)c4Cl)c3c(=O)n2C)[C@@H]1N |r| Show InChI InChI=1S/C23H29ClN8O2/c1-12-18(25)23(11-34-12)6-9-32(10-7-23)22-28-19-15(21(33)31(22)2)17(29-30-19)14-5-8-26-20(16(14)24)27-13-3-4-13/h5,8,12-13,18H,3-4,6-7,9-11,25H2,1-2H3,(H,26,27)(H,29,30)/t12-,18+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 (unknown origin) |
J Med Chem 62: 1781-1792 (2019)
Article DOI: 10.1021/acs.jmedchem.8b01725 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C9
(Homo sapiens (Human)) | BDBM50530251

(CHEMBL4439006 | US10975080, Example 75)Show SMILES C[C@@H]1OCC2(CCN(CC2)c2nc3[nH]nc(-c4ccnc(NC5CC5)c4Cl)c3c(=O)n2C)[C@@H]1N |r| Show InChI InChI=1S/C23H29ClN8O2/c1-12-18(25)23(11-34-12)6-9-32(10-7-23)22-28-19-15(21(33)31(22)2)17(29-30-19)14-5-8-26-20(16(14)24)27-13-3-4-13/h5,8,12-13,18H,3-4,6-7,9-11,25H2,1-2H3,(H,26,27)(H,29,30)/t12-,18+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C9 (unknown origin) |
J Med Chem 62: 1781-1792 (2019)
Article DOI: 10.1021/acs.jmedchem.8b01725 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 11
(Homo sapiens) | BDBM50530251

(CHEMBL4439006 | US10975080, Example 75)Show SMILES C[C@@H]1OCC2(CCN(CC2)c2nc3[nH]nc(-c4ccnc(NC5CC5)c4Cl)c3c(=O)n2C)[C@@H]1N |r| Show InChI InChI=1S/C23H29ClN8O2/c1-12-18(25)23(11-34-12)6-9-32(10-7-23)22-28-19-15(21(33)31(22)2)17(29-30-19)14-5-8-26-20(16(14)24)27-13-3-4-13/h5,8,12-13,18H,3-4,6-7,9-11,25H2,1-2H3,(H,26,27)(H,29,30)/t12-,18+/m0/s1 | PDB
KEGG
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 36 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibition of of SHP2 in human KYSE520 cells assessed as suppression of ERK phosphorylation after 2 hrs by AlphaScreen SureFire Phospho-ERK1/2 assay |
J Med Chem 62: 1781-1792 (2019)
Article DOI: 10.1021/acs.jmedchem.8b01725 |
More data for this
Ligand-Target Pair | |
1,3-beta-glucan synthase component GLS2
(Saccharomyces cerevisiae) | BDBM50530251

(CHEMBL4439006 | US10975080, Example 75)Show SMILES C[C@@H]1OCC2(CCN(CC2)c2nc3[nH]nc(-c4ccnc(NC5CC5)c4Cl)c3c(=O)n2C)[C@@H]1N |r| Show InChI InChI=1S/C23H29ClN8O2/c1-12-18(25)23(11-34-12)6-9-32(10-7-23)22-28-19-15(21(33)31(22)2)17(29-30-19)14-5-8-26-20(16(14)24)27-13-3-4-13/h5,8,12-13,18H,3-4,6-7,9-11,25H2,1-2H3,(H,26,27)(H,29,30)/t12-,18+/m0/s1 | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.70E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibition of human ERG by Q-patch assay |
J Med Chem 62: 1781-1792 (2019)
Article DOI: 10.1021/acs.jmedchem.8b01725 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50530251

(CHEMBL4439006 | US10975080, Example 75)Show SMILES C[C@@H]1OCC2(CCN(CC2)c2nc3[nH]nc(-c4ccnc(NC5CC5)c4Cl)c3c(=O)n2C)[C@@H]1N |r| Show InChI InChI=1S/C23H29ClN8O2/c1-12-18(25)23(11-34-12)6-9-32(10-7-23)22-28-19-15(21(33)31(22)2)17(29-30-19)14-5-8-26-20(16(14)24)27-13-3-4-13/h5,8,12-13,18H,3-4,6-7,9-11,25H2,1-2H3,(H,26,27)(H,29,30)/t12-,18+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 (unknown origin) |
J Med Chem 62: 1781-1792 (2019)
Article DOI: 10.1021/acs.jmedchem.8b01725 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C9
(Homo sapiens (Human)) | BDBM50530251

(CHEMBL4439006 | US10975080, Example 75)Show SMILES C[C@@H]1OCC2(CCN(CC2)c2nc3[nH]nc(-c4ccnc(NC5CC5)c4Cl)c3c(=O)n2C)[C@@H]1N |r| Show InChI InChI=1S/C23H29ClN8O2/c1-12-18(25)23(11-34-12)6-9-32(10-7-23)22-28-19-15(21(33)31(22)2)17(29-30-19)14-5-8-26-20(16(14)24)27-13-3-4-13/h5,8,12-13,18H,3-4,6-7,9-11,25H2,1-2H3,(H,26,27)(H,29,30)/t12-,18+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C9 (unknown origin) |
J Med Chem 62: 1781-1792 (2019)
Article DOI: 10.1021/acs.jmedchem.8b01725 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM50530251

(CHEMBL4439006 | US10975080, Example 75)Show SMILES C[C@@H]1OCC2(CCN(CC2)c2nc3[nH]nc(-c4ccnc(NC5CC5)c4Cl)c3c(=O)n2C)[C@@H]1N |r| Show InChI InChI=1S/C23H29ClN8O2/c1-12-18(25)23(11-34-12)6-9-32(10-7-23)22-28-19-15(21(33)31(22)2)17(29-30-19)14-5-8-26-20(16(14)24)27-13-3-4-13/h5,8,12-13,18H,3-4,6-7,9-11,25H2,1-2H3,(H,26,27)(H,29,30)/t12-,18+/m0/s1 | PDB
UniProtKB/SwissProt
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibition of CYP2D6 (unknown origin) |
J Med Chem 62: 1781-1792 (2019)
Article DOI: 10.1021/acs.jmedchem.8b01725 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM50530251

(CHEMBL4439006 | US10975080, Example 75)Show SMILES C[C@@H]1OCC2(CCN(CC2)c2nc3[nH]nc(-c4ccnc(NC5CC5)c4Cl)c3c(=O)n2C)[C@@H]1N |r| Show InChI InChI=1S/C23H29ClN8O2/c1-12-18(25)23(11-34-12)6-9-32(10-7-23)22-28-19-15(21(33)31(22)2)17(29-30-19)14-5-8-26-20(16(14)24)27-13-3-4-13/h5,8,12-13,18H,3-4,6-7,9-11,25H2,1-2H3,(H,26,27)(H,29,30)/t12-,18+/m0/s1 | PDB
UniProtKB/SwissProt
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibition of CYP2D6 (unknown origin) |
J Med Chem 62: 1781-1792 (2019)
Article DOI: 10.1021/acs.jmedchem.8b01725 |
More data for this
Ligand-Target Pair | |
1,3-beta-glucan synthase component GLS2
(Saccharomyces cerevisiae) | BDBM50530251

(CHEMBL4439006 | US10975080, Example 75)Show SMILES C[C@@H]1OCC2(CCN(CC2)c2nc3[nH]nc(-c4ccnc(NC5CC5)c4Cl)c3c(=O)n2C)[C@@H]1N |r| Show InChI InChI=1S/C23H29ClN8O2/c1-12-18(25)23(11-34-12)6-9-32(10-7-23)22-28-19-15(21(33)31(22)2)17(29-30-19)14-5-8-26-20(16(14)24)27-13-3-4-13/h5,8,12-13,18H,3-4,6-7,9-11,25H2,1-2H3,(H,26,27)(H,29,30)/t12-,18+/m0/s1 | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.70E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibition of human ERG |
J Med Chem 62: 1781-1792 (2019)
Article DOI: 10.1021/acs.jmedchem.8b01725 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 11
(Homo sapiens) | BDBM50530251

(CHEMBL4439006 | US10975080, Example 75)Show SMILES C[C@@H]1OCC2(CCN(CC2)c2nc3[nH]nc(-c4ccnc(NC5CC5)c4Cl)c3c(=O)n2C)[C@@H]1N |r| Show InChI InChI=1S/C23H29ClN8O2/c1-12-18(25)23(11-34-12)6-9-32(10-7-23)22-28-19-15(21(33)31(22)2)17(29-30-19)14-5-8-26-20(16(14)24)27-13-3-4-13/h5,8,12-13,18H,3-4,6-7,9-11,25H2,1-2H3,(H,26,27)(H,29,30)/t12-,18+/m0/s1 | PDB
KEGG
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 36 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibition of human 6His-tagged SHP2 (1 to 525 residues) expressed in Escherichia coli BL21 Star (DE3) using DiFMUP as substrate preincubated for 30 ... |
J Med Chem 62: 1781-1792 (2019)
Article DOI: 10.1021/acs.jmedchem.8b01725 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 11
(Homo sapiens) | BDBM50530251

(CHEMBL4439006 | US10975080, Example 75)Show SMILES C[C@@H]1OCC2(CCN(CC2)c2nc3[nH]nc(-c4ccnc(NC5CC5)c4Cl)c3c(=O)n2C)[C@@H]1N |r| Show InChI InChI=1S/C23H29ClN8O2/c1-12-18(25)23(11-34-12)6-9-32(10-7-23)22-28-19-15(21(33)31(22)2)17(29-30-19)14-5-8-26-20(16(14)24)27-13-3-4-13/h5,8,12-13,18H,3-4,6-7,9-11,25H2,1-2H3,(H,26,27)(H,29,30)/t12-,18+/m0/s1 | PDB
KEGG
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 36 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibition of human 6His-tagged SHP2 (1 to 525 residues) expressed in Escherichia coli BL21 Star (DE3) using DiFMUP as substrate preincubated for 30 ... |
J Med Chem 62: 1781-1792 (2019)
Article DOI: 10.1021/acs.jmedchem.8b01725 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data