

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50458458 (CHEMBL4210729) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 1.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Defence Curated by ChEMBL | Assay Description Mixed type inhibition of human erythrocyte AChE assessed as enzyme-substrate-inhibitor complex using varying levels of acetylthiocholine iodide as su... | Eur J Med Chem 150: 292-306 (2018) Article DOI: 10.1016/j.ejmech.2018.02.083 BindingDB Entry DOI: 10.7270/Q2W95CT0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50458458 (CHEMBL4210729) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 3.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Defence Curated by ChEMBL | Assay Description Mixed type inhibition of human erythrocyte AChE assessed as enzyme-inhibitor complex using varying levels of acetylthiocholine iodide as substrate pr... | Eur J Med Chem 150: 292-306 (2018) Article DOI: 10.1016/j.ejmech.2018.02.083 BindingDB Entry DOI: 10.7270/Q2W95CT0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM8987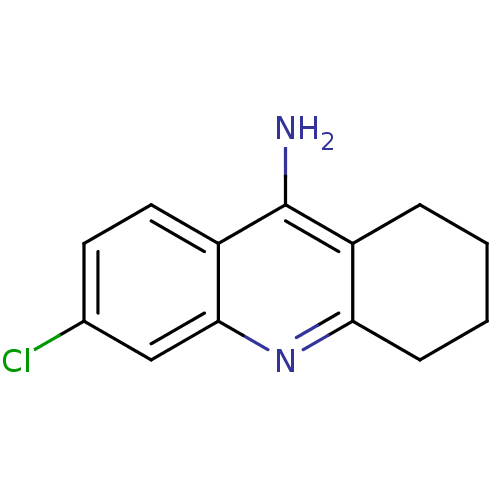 (6-chloro-1,2,3,4-tetrahydroacridin-9-amine | 6-chl...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 18 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Defence Curated by ChEMBL | Assay Description Inhibition of human erythrocyte AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured at 2... | Eur J Med Chem 150: 292-306 (2018) Article DOI: 10.1016/j.ejmech.2018.02.083 BindingDB Entry DOI: 10.7270/Q2W95CT0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50458458 (CHEMBL4210729) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 42 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Defence Curated by ChEMBL | Assay Description Inhibition of human erythrocyte AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured at 2... | Eur J Med Chem 150: 292-306 (2018) Article DOI: 10.1016/j.ejmech.2018.02.083 BindingDB Entry DOI: 10.7270/Q2W95CT0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50458457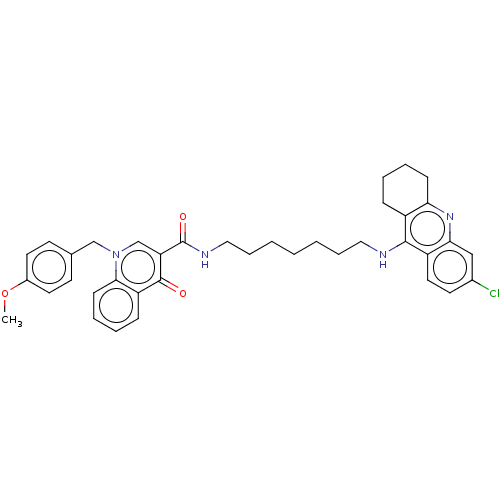 (CHEMBL4213591) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 52 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Defence Curated by ChEMBL | Assay Description Inhibition of human erythrocyte AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured at 2... | Eur J Med Chem 150: 292-306 (2018) Article DOI: 10.1016/j.ejmech.2018.02.083 BindingDB Entry DOI: 10.7270/Q2W95CT0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM50458451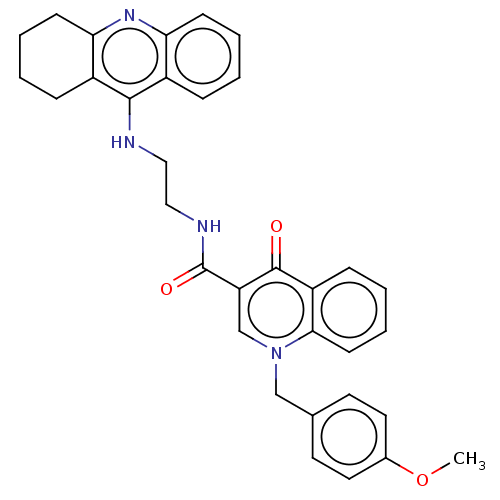 (CHEMBL4205374) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 59 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Defence Curated by ChEMBL | Assay Description Inhibition of human plasmatic BuChE using butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured at 2... | Eur J Med Chem 150: 292-306 (2018) Article DOI: 10.1016/j.ejmech.2018.02.083 BindingDB Entry DOI: 10.7270/Q2W95CT0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50458459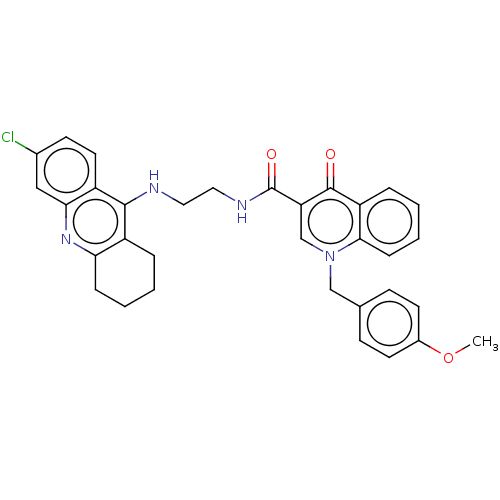 (CHEMBL4215217) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 75 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Defence Curated by ChEMBL | Assay Description Inhibition of human erythrocyte AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured at 2... | Eur J Med Chem 150: 292-306 (2018) Article DOI: 10.1016/j.ejmech.2018.02.083 BindingDB Entry DOI: 10.7270/Q2W95CT0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM50458459 (CHEMBL4215217) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 83 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Defence Curated by ChEMBL | Assay Description Inhibition of human plasmatic BuChE using butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured at 2... | Eur J Med Chem 150: 292-306 (2018) Article DOI: 10.1016/j.ejmech.2018.02.083 BindingDB Entry DOI: 10.7270/Q2W95CT0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM8961 (1,2,3,4-tetrahydro-9-acridinamine | 1,2,3,4-tetrah...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | n/a | n/a | 88 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Defence Curated by ChEMBL | Assay Description Inhibition of human plasmatic BuChE using butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured at 2... | Eur J Med Chem 150: 292-306 (2018) Article DOI: 10.1016/j.ejmech.2018.02.083 BindingDB Entry DOI: 10.7270/Q2W95CT0 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50458448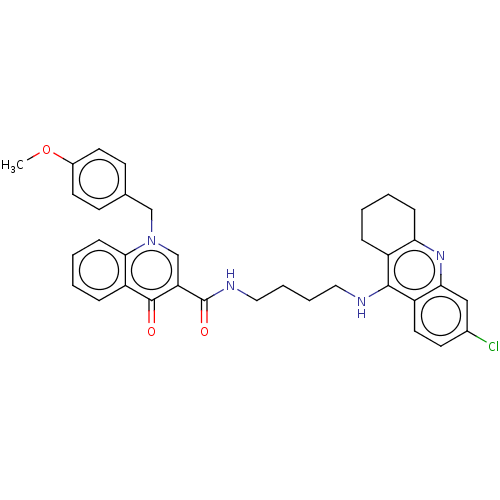 (CHEMBL4215154) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 90 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Defence Curated by ChEMBL | Assay Description Inhibition of human erythrocyte AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured at 2... | Eur J Med Chem 150: 292-306 (2018) Article DOI: 10.1016/j.ejmech.2018.02.083 BindingDB Entry DOI: 10.7270/Q2W95CT0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50458444 (CHEMBL4217346) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 94 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Defence Curated by ChEMBL | Assay Description Inhibition of human erythrocyte AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured at 2... | Eur J Med Chem 150: 292-306 (2018) Article DOI: 10.1016/j.ejmech.2018.02.083 BindingDB Entry DOI: 10.7270/Q2W95CT0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50458445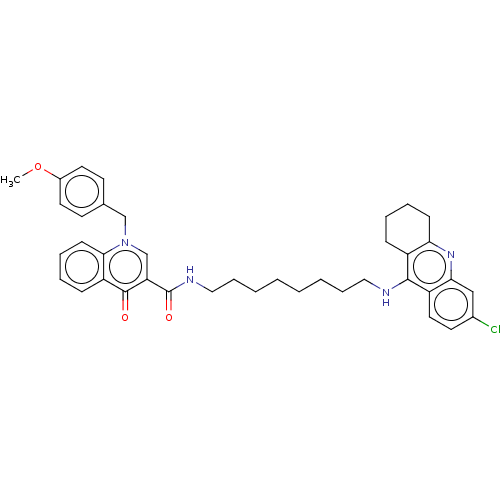 (CHEMBL4203672) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 119 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Defence Curated by ChEMBL | Assay Description Inhibition of human erythrocyte AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured at 2... | Eur J Med Chem 150: 292-306 (2018) Article DOI: 10.1016/j.ejmech.2018.02.083 BindingDB Entry DOI: 10.7270/Q2W95CT0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50458460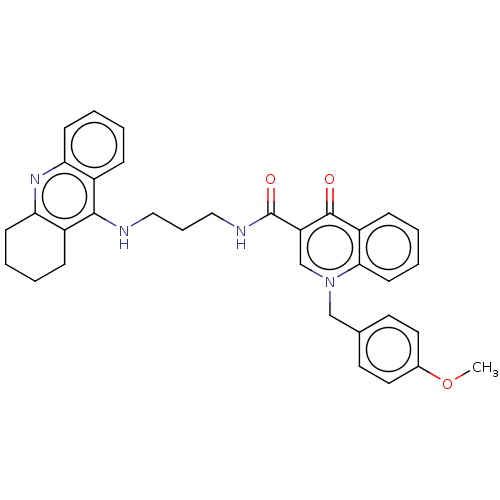 (CHEMBL4214668) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 129 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Defence Curated by ChEMBL | Assay Description Inhibition of human erythrocyte AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured at 2... | Eur J Med Chem 150: 292-306 (2018) Article DOI: 10.1016/j.ejmech.2018.02.083 BindingDB Entry DOI: 10.7270/Q2W95CT0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50458447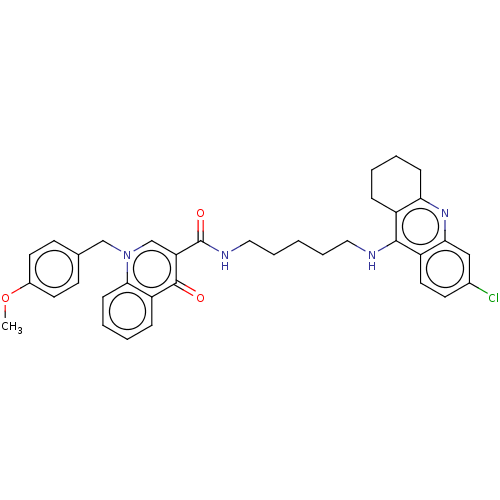 (CHEMBL4217660) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 180 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Defence Curated by ChEMBL | Assay Description Inhibition of human erythrocyte AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured at 2... | Eur J Med Chem 150: 292-306 (2018) Article DOI: 10.1016/j.ejmech.2018.02.083 BindingDB Entry DOI: 10.7270/Q2W95CT0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50458451 (CHEMBL4205374) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 187 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Defence Curated by ChEMBL | Assay Description Inhibition of human erythrocyte AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured at 2... | Eur J Med Chem 150: 292-306 (2018) Article DOI: 10.1016/j.ejmech.2018.02.083 BindingDB Entry DOI: 10.7270/Q2W95CT0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50458449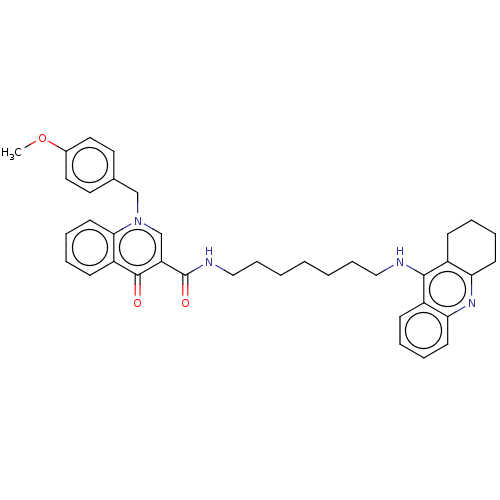 (CHEMBL4214900) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 194 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Defence Curated by ChEMBL | Assay Description Inhibition of human erythrocyte AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured at 2... | Eur J Med Chem 150: 292-306 (2018) Article DOI: 10.1016/j.ejmech.2018.02.083 BindingDB Entry DOI: 10.7270/Q2W95CT0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50458450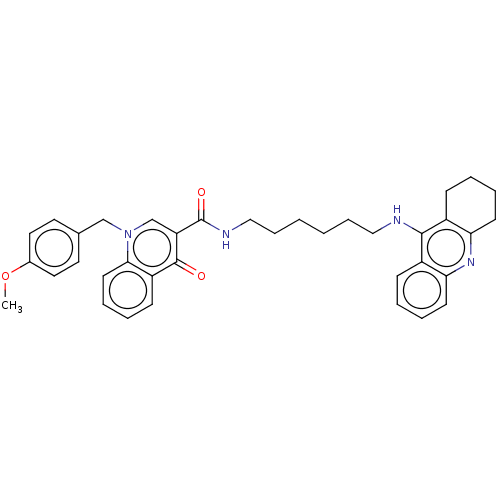 (CHEMBL4206484) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 194 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Defence Curated by ChEMBL | Assay Description Inhibition of human erythrocyte AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured at 2... | Eur J Med Chem 150: 292-306 (2018) Article DOI: 10.1016/j.ejmech.2018.02.083 BindingDB Entry DOI: 10.7270/Q2W95CT0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM50458450 (CHEMBL4206484) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 280 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Defence Curated by ChEMBL | Assay Description Inhibition of human plasmatic BuChE using butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured at 2... | Eur J Med Chem 150: 292-306 (2018) Article DOI: 10.1016/j.ejmech.2018.02.083 BindingDB Entry DOI: 10.7270/Q2W95CT0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50458461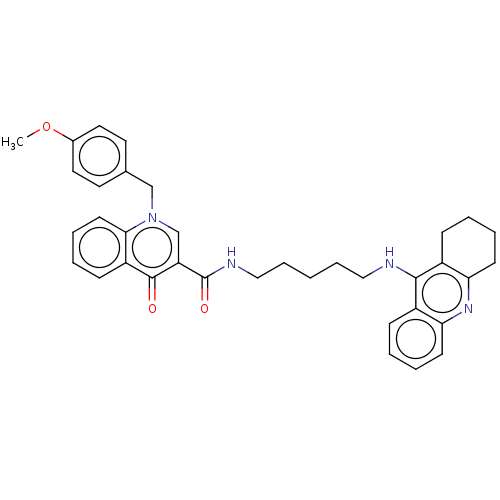 (CHEMBL4202784) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 293 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Defence Curated by ChEMBL | Assay Description Inhibition of human erythrocyte AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured at 2... | Eur J Med Chem 150: 292-306 (2018) Article DOI: 10.1016/j.ejmech.2018.02.083 BindingDB Entry DOI: 10.7270/Q2W95CT0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM8961 (1,2,3,4-tetrahydro-9-acridinamine | 1,2,3,4-tetrah...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | n/a | n/a | 320 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Defence Curated by ChEMBL | Assay Description Inhibition of human erythrocyte AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured at 2... | Eur J Med Chem 150: 292-306 (2018) Article DOI: 10.1016/j.ejmech.2018.02.083 BindingDB Entry DOI: 10.7270/Q2W95CT0 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM50458443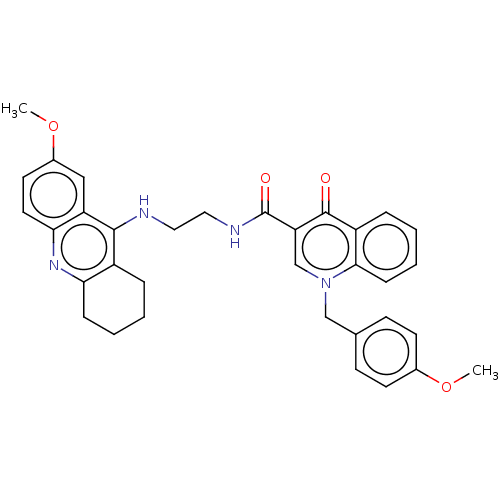 (CHEMBL4212151) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 408 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Defence Curated by ChEMBL | Assay Description Inhibition of human plasmatic BuChE using butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured at 2... | Eur J Med Chem 150: 292-306 (2018) Article DOI: 10.1016/j.ejmech.2018.02.083 BindingDB Entry DOI: 10.7270/Q2W95CT0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M1 (Homo sapiens (Human)) | BDBM50458460 (CHEMBL4214668) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 455 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Defence Curated by ChEMBL | Assay Description Antagonist activity at human M1 mAChR expressed in CHO cells assessed as inhibition of oxotremorine M-stimulated calcium influx preincubated for 10 m... | Eur J Med Chem 150: 292-306 (2018) Article DOI: 10.1016/j.ejmech.2018.02.083 BindingDB Entry DOI: 10.7270/Q2W95CT0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50458446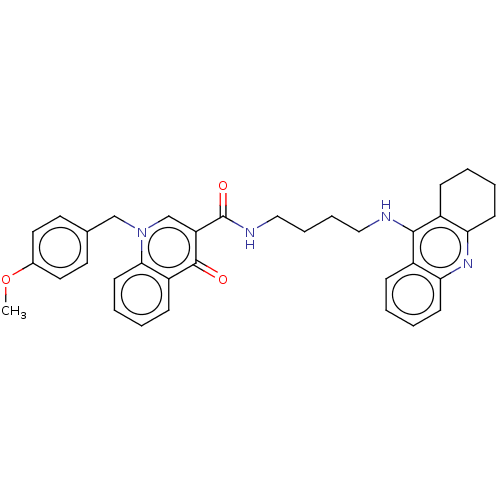 (CHEMBL4209755) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 491 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Defence Curated by ChEMBL | Assay Description Inhibition of human erythrocyte AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured at 2... | Eur J Med Chem 150: 292-306 (2018) Article DOI: 10.1016/j.ejmech.2018.02.083 BindingDB Entry DOI: 10.7270/Q2W95CT0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M1 (Homo sapiens (Human)) | BDBM50458451 (CHEMBL4205374) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 658 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Defence Curated by ChEMBL | Assay Description Antagonist activity at human M1 mAChR expressed in CHO cells assessed as inhibition of oxotremorine M-stimulated calcium influx preincubated for 10 m... | Eur J Med Chem 150: 292-306 (2018) Article DOI: 10.1016/j.ejmech.2018.02.083 BindingDB Entry DOI: 10.7270/Q2W95CT0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM50458460 (CHEMBL4214668) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 674 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Defence Curated by ChEMBL | Assay Description Inhibition of human plasmatic BuChE using butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured at 2... | Eur J Med Chem 150: 292-306 (2018) Article DOI: 10.1016/j.ejmech.2018.02.083 BindingDB Entry DOI: 10.7270/Q2W95CT0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50458442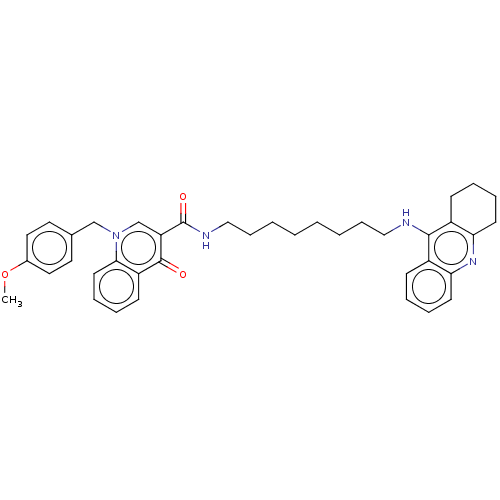 (CHEMBL4204901) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 768 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Defence Curated by ChEMBL | Assay Description Inhibition of human erythrocyte AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured at 2... | Eur J Med Chem 150: 292-306 (2018) Article DOI: 10.1016/j.ejmech.2018.02.083 BindingDB Entry DOI: 10.7270/Q2W95CT0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM50458461 (CHEMBL4202784) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 823 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Defence Curated by ChEMBL | Assay Description Inhibition of human plasmatic BuChE using butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured at 2... | Eur J Med Chem 150: 292-306 (2018) Article DOI: 10.1016/j.ejmech.2018.02.083 BindingDB Entry DOI: 10.7270/Q2W95CT0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM50458455 (CHEMBL4217126) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 1.03E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Defence Curated by ChEMBL | Assay Description Inhibition of human plasmatic BuChE using butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured at 2... | Eur J Med Chem 150: 292-306 (2018) Article DOI: 10.1016/j.ejmech.2018.02.083 BindingDB Entry DOI: 10.7270/Q2W95CT0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M1 (Homo sapiens (Human)) | BDBM50458454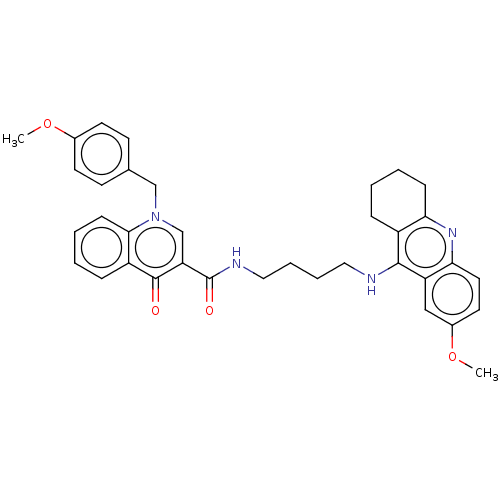 (CHEMBL4216601) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 1.07E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Defence Curated by ChEMBL | Assay Description Antagonist activity at human M1 mAChR expressed in CHO cells assessed as inhibition of oxotremorine M-stimulated calcium influx preincubated for 10 m... | Eur J Med Chem 150: 292-306 (2018) Article DOI: 10.1016/j.ejmech.2018.02.083 BindingDB Entry DOI: 10.7270/Q2W95CT0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM50458454 (CHEMBL4216601) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 1.08E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Defence Curated by ChEMBL | Assay Description Inhibition of human plasmatic BuChE using butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured at 2... | Eur J Med Chem 150: 292-306 (2018) Article DOI: 10.1016/j.ejmech.2018.02.083 BindingDB Entry DOI: 10.7270/Q2W95CT0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM50458449 (CHEMBL4214900) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 1.19E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Defence Curated by ChEMBL | Assay Description Inhibition of human plasmatic BuChE using butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured at 2... | Eur J Med Chem 150: 292-306 (2018) Article DOI: 10.1016/j.ejmech.2018.02.083 BindingDB Entry DOI: 10.7270/Q2W95CT0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM50458442 (CHEMBL4204901) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 1.28E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Defence Curated by ChEMBL | Assay Description Inhibition of human plasmatic BuChE using butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured at 2... | Eur J Med Chem 150: 292-306 (2018) Article DOI: 10.1016/j.ejmech.2018.02.083 BindingDB Entry DOI: 10.7270/Q2W95CT0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM50458446 (CHEMBL4209755) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 1.32E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Defence Curated by ChEMBL | Assay Description Inhibition of human plasmatic BuChE using butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured at 2... | Eur J Med Chem 150: 292-306 (2018) Article DOI: 10.1016/j.ejmech.2018.02.083 BindingDB Entry DOI: 10.7270/Q2W95CT0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M1 (Homo sapiens (Human)) | BDBM50458441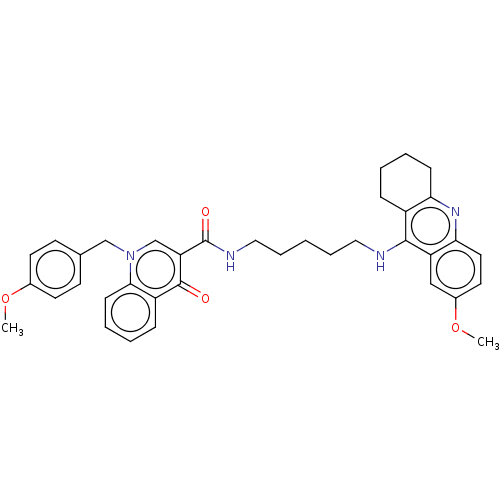 (CHEMBL4204913) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 1.37E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Defence Curated by ChEMBL | Assay Description Antagonist activity at human M1 mAChR expressed in CHO cells assessed as inhibition of oxotremorine M-stimulated calcium influx preincubated for 10 m... | Eur J Med Chem 150: 292-306 (2018) Article DOI: 10.1016/j.ejmech.2018.02.083 BindingDB Entry DOI: 10.7270/Q2W95CT0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M1 (Homo sapiens (Human)) | BDBM50458461 (CHEMBL4202784) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 1.48E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Defence Curated by ChEMBL | Assay Description Antagonist activity at human M1 mAChR expressed in CHO cells assessed as inhibition of oxotremorine M-stimulated calcium influx preincubated for 10 m... | Eur J Med Chem 150: 292-306 (2018) Article DOI: 10.1016/j.ejmech.2018.02.083 BindingDB Entry DOI: 10.7270/Q2W95CT0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M1 (Homo sapiens (Human)) | BDBM50458450 (CHEMBL4206484) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 1.49E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Defence Curated by ChEMBL | Assay Description Antagonist activity at human M1 mAChR expressed in CHO cells assessed as inhibition of oxotremorine M-stimulated calcium influx preincubated for 10 m... | Eur J Med Chem 150: 292-306 (2018) Article DOI: 10.1016/j.ejmech.2018.02.083 BindingDB Entry DOI: 10.7270/Q2W95CT0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50458456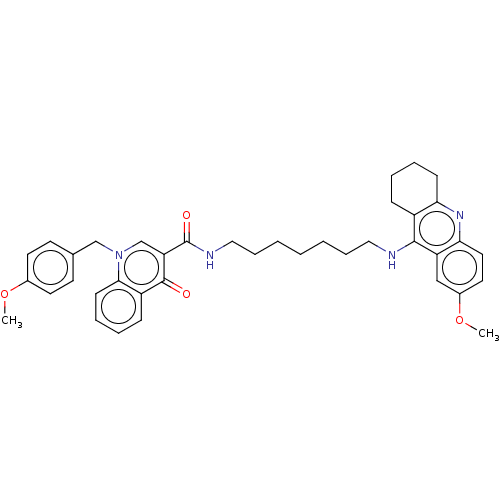 (CHEMBL4217617) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 1.53E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Defence Curated by ChEMBL | Assay Description Inhibition of human erythrocyte AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured at 2... | Eur J Med Chem 150: 292-306 (2018) Article DOI: 10.1016/j.ejmech.2018.02.083 BindingDB Entry DOI: 10.7270/Q2W95CT0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M1 (Homo sapiens (Human)) | BDBM50458446 (CHEMBL4209755) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 1.59E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Defence Curated by ChEMBL | Assay Description Antagonist activity at human M1 mAChR expressed in CHO cells assessed as inhibition of oxotremorine M-stimulated calcium influx preincubated for 10 m... | Eur J Med Chem 150: 292-306 (2018) Article DOI: 10.1016/j.ejmech.2018.02.083 BindingDB Entry DOI: 10.7270/Q2W95CT0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM8987 (6-chloro-1,2,3,4-tetrahydroacridin-9-amine | 6-chl...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 1.73E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Defence Curated by ChEMBL | Assay Description Inhibition of human plasmatic BuChE using butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured at 2... | Eur J Med Chem 150: 292-306 (2018) Article DOI: 10.1016/j.ejmech.2018.02.083 BindingDB Entry DOI: 10.7270/Q2W95CT0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M1 (Homo sapiens (Human)) | BDBM50458442 (CHEMBL4204901) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 2.05E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Defence Curated by ChEMBL | Assay Description Antagonist activity at human M1 mAChR expressed in CHO cells assessed as inhibition of oxotremorine M-stimulated calcium influx preincubated for 10 m... | Eur J Med Chem 150: 292-306 (2018) Article DOI: 10.1016/j.ejmech.2018.02.083 BindingDB Entry DOI: 10.7270/Q2W95CT0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M1 (Homo sapiens (Human)) | BDBM50458449 (CHEMBL4214900) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 2.14E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Defence Curated by ChEMBL | Assay Description Antagonist activity at human M1 mAChR expressed in CHO cells assessed as inhibition of oxotremorine M-stimulated calcium influx preincubated for 10 m... | Eur J Med Chem 150: 292-306 (2018) Article DOI: 10.1016/j.ejmech.2018.02.083 BindingDB Entry DOI: 10.7270/Q2W95CT0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50458455 (CHEMBL4217126) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 2.24E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Defence Curated by ChEMBL | Assay Description Inhibition of human erythrocyte AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured at 2... | Eur J Med Chem 150: 292-306 (2018) Article DOI: 10.1016/j.ejmech.2018.02.083 BindingDB Entry DOI: 10.7270/Q2W95CT0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M1 (Homo sapiens (Human)) | BDBM50458455 (CHEMBL4217126) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 2.48E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Defence Curated by ChEMBL | Assay Description Antagonist activity at human M1 mAChR expressed in CHO cells assessed as inhibition of oxotremorine M-stimulated calcium influx preincubated for 10 m... | Eur J Med Chem 150: 292-306 (2018) Article DOI: 10.1016/j.ejmech.2018.02.083 BindingDB Entry DOI: 10.7270/Q2W95CT0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50458443 (CHEMBL4212151) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 2.64E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Defence Curated by ChEMBL | Assay Description Inhibition of human erythrocyte AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured at 2... | Eur J Med Chem 150: 292-306 (2018) Article DOI: 10.1016/j.ejmech.2018.02.083 BindingDB Entry DOI: 10.7270/Q2W95CT0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M1 (Homo sapiens (Human)) | BDBM50458444 (CHEMBL4217346) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 2.78E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Defence Curated by ChEMBL | Assay Description Antagonist activity at human M1 mAChR expressed in CHO cells assessed as inhibition of oxotremorine M-stimulated calcium influx preincubated for 10 m... | Eur J Med Chem 150: 292-306 (2018) Article DOI: 10.1016/j.ejmech.2018.02.083 BindingDB Entry DOI: 10.7270/Q2W95CT0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM50458448 (CHEMBL4215154) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 3.12E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Defence Curated by ChEMBL | Assay Description Inhibition of human plasmatic BuChE using butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured at 2... | Eur J Med Chem 150: 292-306 (2018) Article DOI: 10.1016/j.ejmech.2018.02.083 BindingDB Entry DOI: 10.7270/Q2W95CT0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M1 (Homo sapiens (Human)) | BDBM50458447 (CHEMBL4217660) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 3.27E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Defence Curated by ChEMBL | Assay Description Antagonist activity at human M1 mAChR expressed in CHO cells assessed as inhibition of oxotremorine M-stimulated calcium influx preincubated for 10 m... | Eur J Med Chem 150: 292-306 (2018) Article DOI: 10.1016/j.ejmech.2018.02.083 BindingDB Entry DOI: 10.7270/Q2W95CT0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM50458457 (CHEMBL4213591) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 3.34E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Defence Curated by ChEMBL | Assay Description Inhibition of human plasmatic BuChE using butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured at 2... | Eur J Med Chem 150: 292-306 (2018) Article DOI: 10.1016/j.ejmech.2018.02.083 BindingDB Entry DOI: 10.7270/Q2W95CT0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M1 (Homo sapiens (Human)) | BDBM50458443 (CHEMBL4212151) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 3.71E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Defence Curated by ChEMBL | Assay Description Antagonist activity at human M1 mAChR expressed in CHO cells assessed as inhibition of oxotremorine M-stimulated calcium influx preincubated for 10 m... | Eur J Med Chem 150: 292-306 (2018) Article DOI: 10.1016/j.ejmech.2018.02.083 BindingDB Entry DOI: 10.7270/Q2W95CT0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M1 (Homo sapiens (Human)) | BDBM50327939 (7-methoxytacrine | CHEMBL1256415) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 3.71E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Defence Curated by ChEMBL | Assay Description Antagonist activity at human M1 mAChR expressed in CHO cells assessed as inhibition of oxotremorine M-stimulated calcium influx preincubated for 10 m... | Eur J Med Chem 150: 292-306 (2018) Article DOI: 10.1016/j.ejmech.2018.02.083 BindingDB Entry DOI: 10.7270/Q2W95CT0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Displayed 1 to 50 (of 76 total ) | Next | Last >> |