

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Angiotensin-converting enzyme (Rattus norvegicus) | BDBM50367254 (ENALAPRILAT) | PDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase MCE KEGG MMDB PC cid PC sid PDB UniChem Similars | PubMed | n/a | n/a | 2.30 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibition of rat Angiotensin I converting enzyme (ACE), using Hip-Gly-Gly as synthetic substrate. | J Med Chem 30: 992-8 (1987) BindingDB Entry DOI: 10.7270/Q29C6Z02 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Rattus norvegicus) | BDBM50226811 (CHEMBL3349972) | PDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 2.40 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibition of rat Angiotensin I converting enzyme (ACE), using Hip-Gly-Gly as synthetic substrate. | J Med Chem 30: 992-8 (1987) BindingDB Entry DOI: 10.7270/Q29C6Z02 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Rattus norvegicus) | BDBM50405541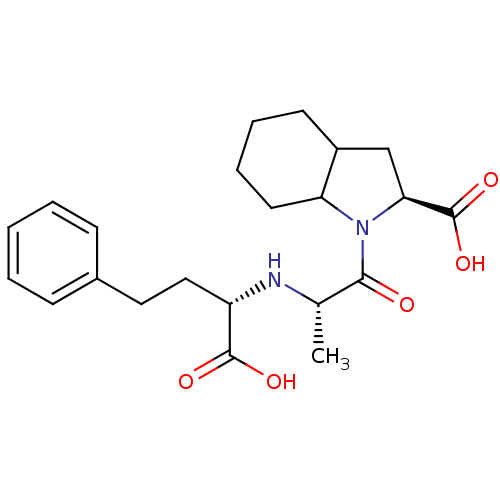 (CHEMBL2079670) | PDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 2.60 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibition of rat Angiotensin I converting enzyme (ACE), using Hip-Gly-Gly as synthetic substrate. | J Med Chem 30: 992-8 (1987) BindingDB Entry DOI: 10.7270/Q29C6Z02 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Rattus norvegicus) | BDBM50027141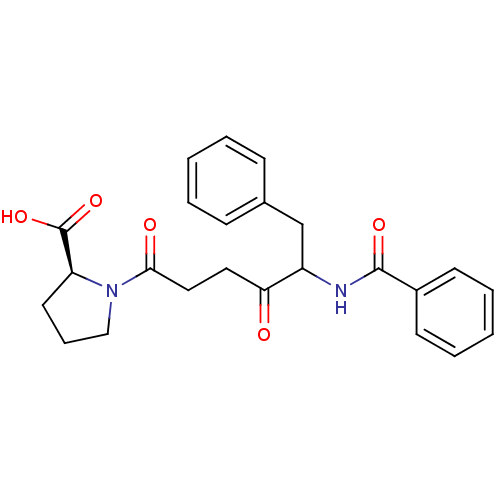 (1-(5-Benzoylamino-4-oxo-6-phenyl-hexanoyl)-pyrroli...) | PDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 3.20 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibition of rat Angiotensin I converting enzyme (ACE), using Hip-Gly-Gly as synthetic substrate. | J Med Chem 30: 992-8 (1987) BindingDB Entry DOI: 10.7270/Q29C6Z02 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Rattus norvegicus) | BDBM50421824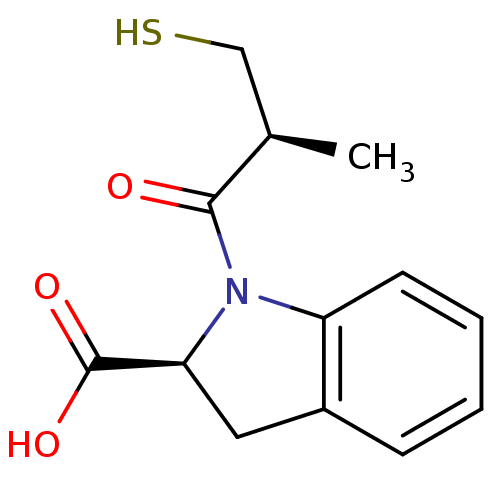 (CHEMBL75752) | PDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 3.70 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibition of rat Angiotensin I converting enzyme (ACE), using Hip-Gly-Gly as synthetic substrate. | J Med Chem 30: 992-8 (1987) BindingDB Entry DOI: 10.7270/Q29C6Z02 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Rattus norvegicus) | BDBM50021795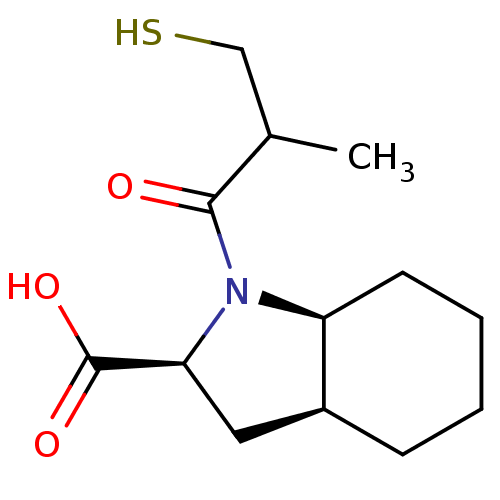 ((R,R+S,S) 1-(3-Mercapto-2-methyl-propionyl)-octahy...) | PDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 5.20 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibition of rat Angiotensin I converting enzyme (ACE), using Hip-Gly-Gly as synthetic substrate. | J Med Chem 30: 992-8 (1987) BindingDB Entry DOI: 10.7270/Q29C6Z02 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Rattus norvegicus) | BDBM50021808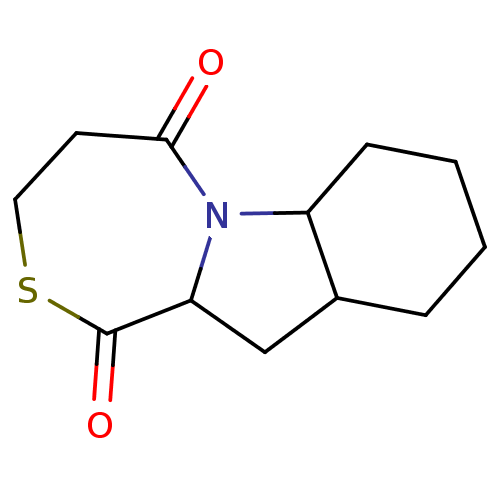 (CHEMBL368598 | Decahydro-8-thia-4b-aza-benzo[a]azu...) | PDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 7 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibition of rat Angiotensin I converting enzyme (ACE), using Hip-Gly-Gly as synthetic substrate. | J Med Chem 30: 992-8 (1987) BindingDB Entry DOI: 10.7270/Q29C6Z02 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Rattus norvegicus) | BDBM50021807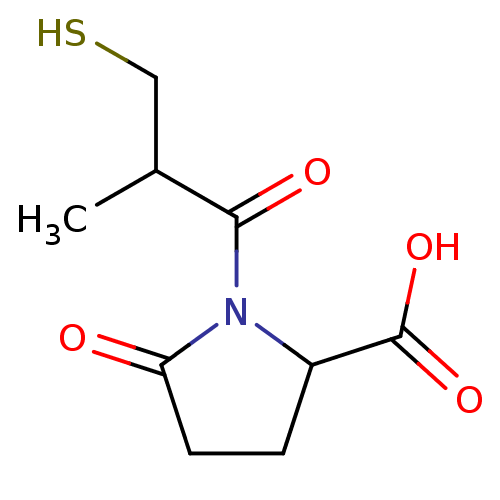 ((S,S)1-(3-Mercapto-2-methyl-propionyl)-5-oxo-pyrro...) | PDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 9.30 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibition of rat Angiotensin I converting enzyme (ACE), using Hip-Gly-Gly as synthetic substrate. | J Med Chem 30: 992-8 (1987) BindingDB Entry DOI: 10.7270/Q29C6Z02 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Rattus norvegicus) | BDBM50021784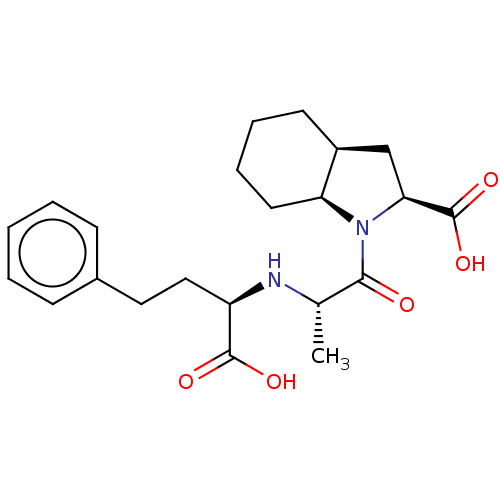 ((RSR)1-[2-(1-Carboxy-3-phenyl-propylamino)-propion...) | PDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 11 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibition of rat Angiotensin I converting enzyme (ACE), using Hip-Gly-Gly as synthetic substrate. | J Med Chem 30: 992-8 (1987) BindingDB Entry DOI: 10.7270/Q29C6Z02 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Rattus norvegicus) | BDBM50021801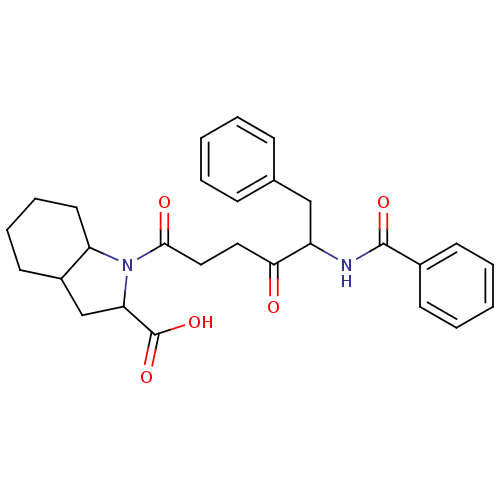 (1-(5-Benzoylamino-4-oxo-6-phenyl-hexanoyl)-octahyd...) | PDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 11 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibition of rat Angiotensin I converting enzyme (ACE), using Hip-Gly-Gly as synthetic substrate. | J Med Chem 30: 992-8 (1987) BindingDB Entry DOI: 10.7270/Q29C6Z02 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Rattus norvegicus) | BDBM50021784 ((RSR)1-[2-(1-Carboxy-3-phenyl-propylamino)-propion...) | PDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 11 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibition of rat Angiotensin I converting enzyme (ACE), using Hip-Gly-Gly as synthetic substrate. | J Med Chem 30: 992-8 (1987) BindingDB Entry DOI: 10.7270/Q29C6Z02 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Rattus norvegicus) | BDBM21642 ((2S)-1-[(2S)-2-methyl-3-sulfanylpropanoyl]pyrrolid...) | PDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE MMDB PC cid PC sid PDB UniChem Patents Similars | PubMed | n/a | n/a | 12 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibition of rat Angiotensin I converting enzyme (ACE), using Hip-Gly-Gly as synthetic substrate. | J Med Chem 30: 992-8 (1987) BindingDB Entry DOI: 10.7270/Q29C6Z02 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Rattus norvegicus) | BDBM50021792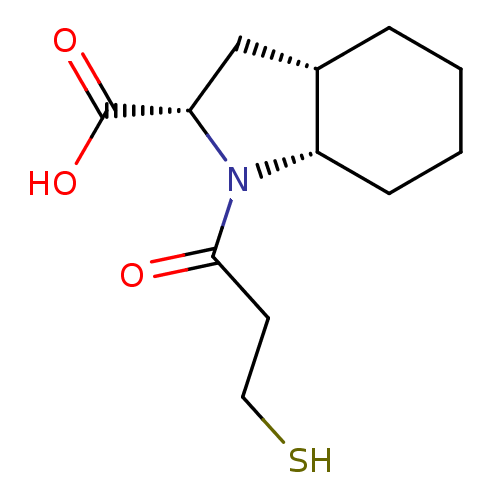 ((S)1-(3-Mercapto-propionyl)-octahydro-indole-2-car...) | PDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 13 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibition of rat Angiotensin I converting enzyme (ACE), using Hip-Gly-Gly as synthetic substrate. | J Med Chem 30: 992-8 (1987) BindingDB Entry DOI: 10.7270/Q29C6Z02 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Rattus norvegicus) | BDBM50367514 (CHEMBL1744311) | PDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 13 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibition of rat Angiotensin I converting enzyme (ACE), using Hip-Gly-Gly as synthetic substrate. | J Med Chem 30: 992-8 (1987) BindingDB Entry DOI: 10.7270/Q29C6Z02 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Rattus norvegicus) | BDBM50021792 ((S)1-(3-Mercapto-propionyl)-octahydro-indole-2-car...) | PDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 13 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibition of rat Angiotensin I converting enzyme (ACE), using Hip-Gly-Gly as synthetic substrate. | J Med Chem 30: 992-8 (1987) BindingDB Entry DOI: 10.7270/Q29C6Z02 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Rattus norvegicus) | BDBM50367513 (CHEMBL1744310) | PDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 14 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibition of rat Angiotensin I converting enzyme (ACE), using Hip-Gly-Gly as synthetic substrate. | J Med Chem 30: 992-8 (1987) BindingDB Entry DOI: 10.7270/Q29C6Z02 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Rattus norvegicus) | BDBM50405539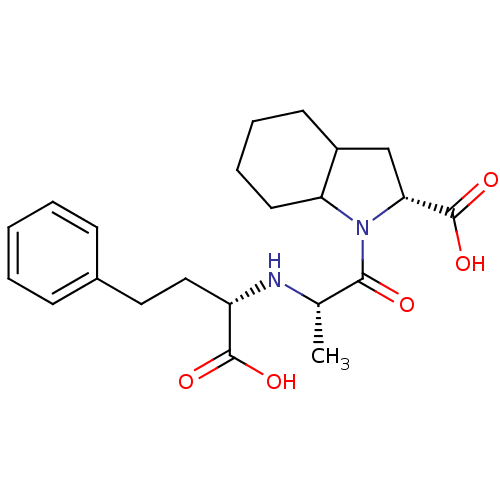 (CHEMBL2052024) | PDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 18 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibition of rat Angiotensin I converting enzyme (ACE), using Hip-Gly-Gly as synthetic substrate. | J Med Chem 30: 992-8 (1987) BindingDB Entry DOI: 10.7270/Q29C6Z02 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Rattus norvegicus) | BDBM50367515 (CHEMBL1744316) | PDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 24 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibition of rat Angiotensin I converting enzyme (ACE), using Hip-Gly-Gly as synthetic substrate. | J Med Chem 30: 992-8 (1987) BindingDB Entry DOI: 10.7270/Q29C6Z02 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Rattus norvegicus) | BDBM50021804 (2-(3-Mercapto-2-methyl-propionyl)-octahydro-isoind...) | PDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 28 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibition of rat Angiotensin I converting enzyme (ACE), using Hip-Gly-Gly as synthetic substrate. | J Med Chem 30: 992-8 (1987) BindingDB Entry DOI: 10.7270/Q29C6Z02 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Rattus norvegicus) | BDBM50021786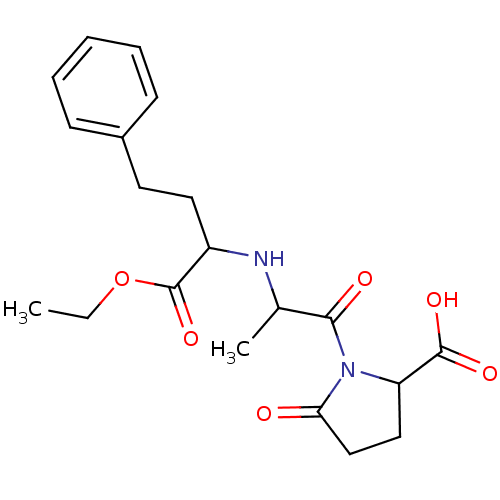 ((SSS)1-[2-(1-Ethoxycarbonyl-3-phenyl-propylamino)-...) | PDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 63 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibition of rat Angiotensin I converting enzyme (ACE), using Hip-Gly-Gly as synthetic substrate. | J Med Chem 30: 992-8 (1987) BindingDB Entry DOI: 10.7270/Q29C6Z02 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Rattus norvegicus) | BDBM50017129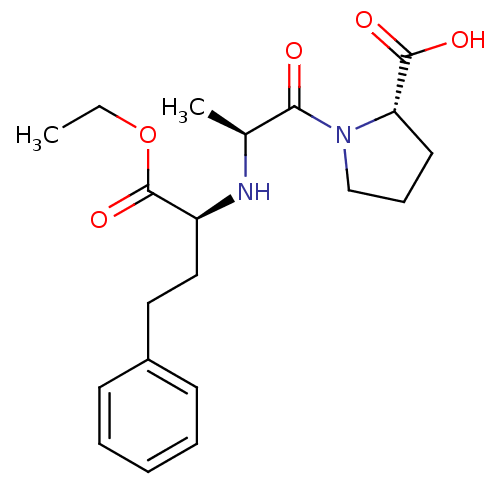 ((S)-1-((S)-2-((R)-1-ethoxy-1-oxo-4-phenylbutan-2-y...) | PDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 140 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibition of rat Angiotensin I converting enzyme (ACE), using Hip-Gly-Gly as synthetic substrate. | J Med Chem 30: 992-8 (1987) BindingDB Entry DOI: 10.7270/Q29C6Z02 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Rattus norvegicus) | BDBM50021783 (1-(3-Mercapto-propionyl)-2,3-dihydro-1H-indole-2-c...) | PDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 140 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibition of rat Angiotensin I converting enzyme (ACE), using Hip-Gly-Gly as synthetic substrate. | J Med Chem 30: 992-8 (1987) BindingDB Entry DOI: 10.7270/Q29C6Z02 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Rattus norvegicus) | BDBM50021782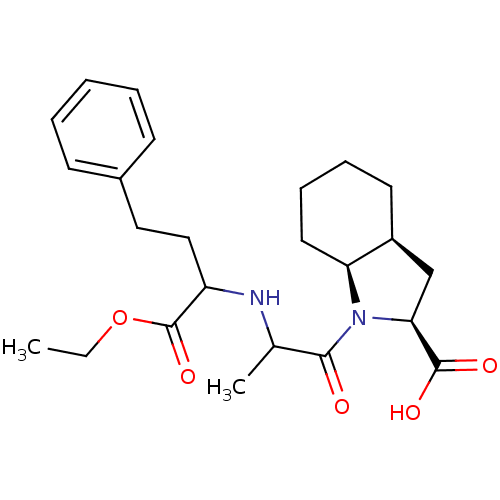 ((RSR)1-[2-(1-Ethoxycarbonyl-3-phenyl-propylamino)-...) | PDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 170 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibition of rat Angiotensin I converting enzyme (ACE), using Hip-Gly-Gly as synthetic substrate. | J Med Chem 30: 992-8 (1987) BindingDB Entry DOI: 10.7270/Q29C6Z02 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Rattus norvegicus) | BDBM50021782 ((RSR)1-[2-(1-Ethoxycarbonyl-3-phenyl-propylamino)-...) | PDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 170 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibition of rat Angiotensin I converting enzyme (ACE), using Hip-Gly-Gly as synthetic substrate. | J Med Chem 30: 992-8 (1987) BindingDB Entry DOI: 10.7270/Q29C6Z02 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Rattus norvegicus) | BDBM50367511 (CHEMBL1744309) | PDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 300 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibition of rat Angiotensin I converting enzyme (ACE), using Hip-Gly-Gly as synthetic substrate. | J Med Chem 30: 992-8 (1987) BindingDB Entry DOI: 10.7270/Q29C6Z02 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Rattus norvegicus) | BDBM50021803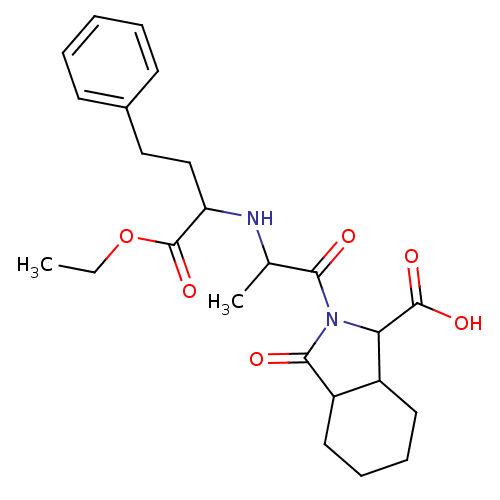 ((SSS+SSR)2-[2-(1-Ethoxycarbonyl-3-phenyl-propylami...) | PDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 340 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibition of rat Angiotensin I converting enzyme (ACE), using Hip-Gly-Gly as synthetic substrate. | J Med Chem 30: 992-8 (1987) BindingDB Entry DOI: 10.7270/Q29C6Z02 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Rattus norvegicus) | BDBM50367510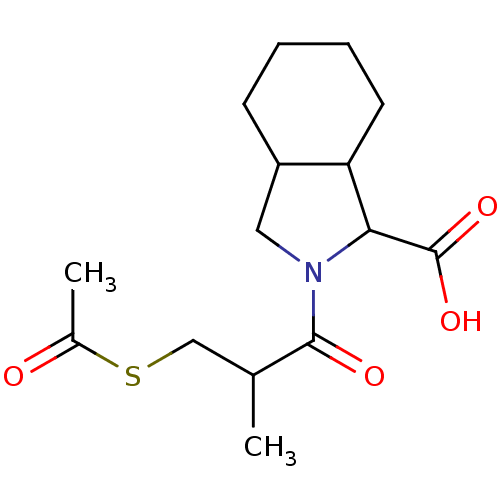 (CHEMBL1794914) | PDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 420 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibition of rat Angiotensin I converting enzyme (ACE), using Hip-Gly-Gly as synthetic substrate. | J Med Chem 30: 992-8 (1987) BindingDB Entry DOI: 10.7270/Q29C6Z02 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Rattus norvegicus) | BDBM50021799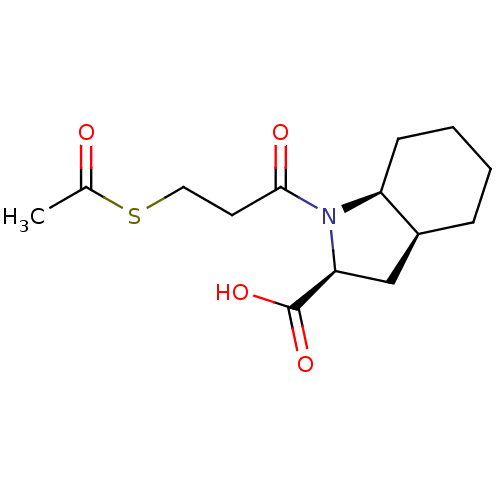 ((S)1-(3-Acetylsulfanyl-propionyl)-octahydro-indole...) | PDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 480 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibition of rat Angiotensin I converting enzyme (ACE), using Hip-Gly-Gly as synthetic substrate. | J Med Chem 30: 992-8 (1987) BindingDB Entry DOI: 10.7270/Q29C6Z02 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Rattus norvegicus) | BDBM50021799 ((S)1-(3-Acetylsulfanyl-propionyl)-octahydro-indole...) | PDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 480 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibition of rat Angiotensin I converting enzyme (ACE), using Hip-Gly-Gly as synthetic substrate. | J Med Chem 30: 992-8 (1987) BindingDB Entry DOI: 10.7270/Q29C6Z02 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Rattus norvegicus) | BDBM50021793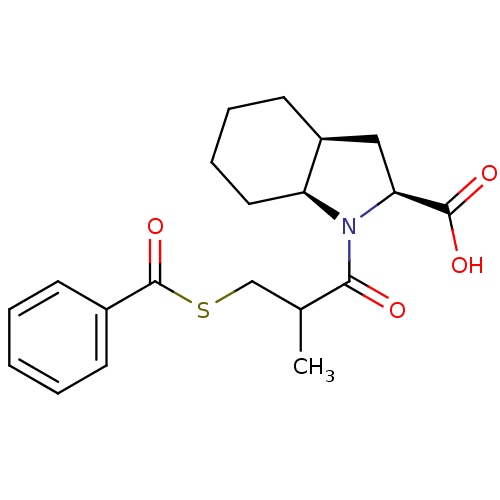 ((S,S) 1-(3-Benzoylsulfanyl-2-methyl-propionyl)-oct...) | PDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 540 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibition of rat Angiotensin I converting enzyme (ACE), using Hip-Gly-Gly as synthetic substrate. | J Med Chem 30: 992-8 (1987) BindingDB Entry DOI: 10.7270/Q29C6Z02 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Rattus norvegicus) | BDBM50367512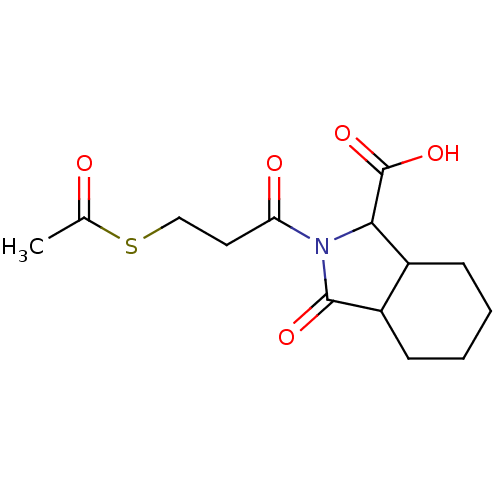 (CHEMBL1744321) | PDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 660 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibition of rat Angiotensin I converting enzyme (ACE), using Hip-Gly-Gly as synthetic substrate. | J Med Chem 30: 992-8 (1987) BindingDB Entry DOI: 10.7270/Q29C6Z02 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Rattus norvegicus) | BDBM50018792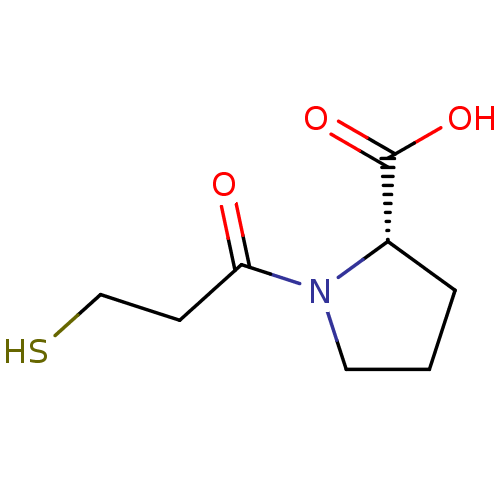 ((L)-1-(3-Mercapto-propionyl)-pyrrolidine-2-carboxy...) | PDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 820 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibition of rat Angiotensin I converting enzyme (ACE), using Hip-Gly-Gly as synthetic substrate. | J Med Chem 30: 992-8 (1987) BindingDB Entry DOI: 10.7270/Q29C6Z02 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Rattus norvegicus) | BDBM50021787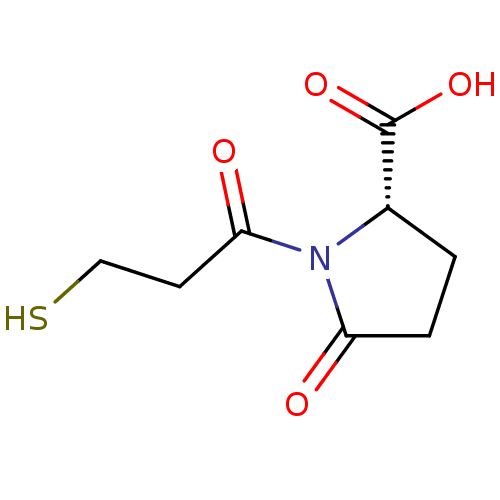 ((S)1-(3-Mercapto-propionyl)-5-oxo-pyrrolidine-2-ca...) | PDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 960 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibition of rat Angiotensin I converting enzyme (ACE), using Hip-Gly-Gly as synthetic substrate. | J Med Chem 30: 992-8 (1987) BindingDB Entry DOI: 10.7270/Q29C6Z02 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Rattus norvegicus) | BDBM50021795 ((R,R+S,S) 1-(3-Mercapto-2-methyl-propionyl)-octahy...) | PDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 1.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibition of rat Angiotensin I converting enzyme (ACE), using Hip-Gly-Gly as synthetic substrate. | J Med Chem 30: 992-8 (1987) BindingDB Entry DOI: 10.7270/Q29C6Z02 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Rattus norvegicus) | BDBM50021795 ((R,R+S,S) 1-(3-Mercapto-2-methyl-propionyl)-octahy...) | PDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 1.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibition of rat Angiotensin I converting enzyme (ACE), using Hip-Gly-Gly as synthetic substrate. | J Med Chem 30: 992-8 (1987) BindingDB Entry DOI: 10.7270/Q29C6Z02 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Rattus norvegicus) | BDBM50021794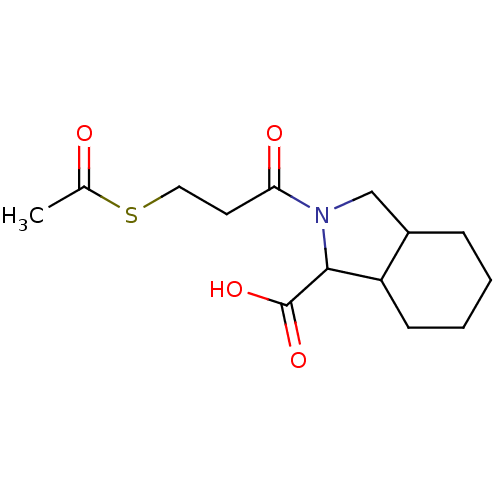 (2-(3-Acetylsulfanyl-propionyl)-octahydro-isoindole...) | PDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 1.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibition of rat Angiotensin I converting enzyme (ACE), using Hip-Gly-Gly as synthetic substrate. | J Med Chem 30: 992-8 (1987) BindingDB Entry DOI: 10.7270/Q29C6Z02 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Rattus norvegicus) | BDBM50021784 ((RSR)1-[2-(1-Carboxy-3-phenyl-propylamino)-propion...) | PDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 2.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibition of rat Angiotensin I converting enzyme (ACE), using Hip-Gly-Gly as synthetic substrate. | J Med Chem 30: 992-8 (1987) BindingDB Entry DOI: 10.7270/Q29C6Z02 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Rattus norvegicus) | BDBM50405540 (CHEMBL2093952) | PDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 7.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibition of rat Angiotensin I converting enzyme (ACE), using Hip-Gly-Gly as synthetic substrate. | J Med Chem 30: 992-8 (1987) BindingDB Entry DOI: 10.7270/Q29C6Z02 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Rattus norvegicus) | BDBM50021797 ((R,R+S,S) 1-(3-Acetylsulfanyl-2-methyl-propionyl)-...) | PDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 3.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibition of rat Angiotensin I converting enzyme (ACE), using Hip-Gly-Gly as synthetic substrate. | J Med Chem 30: 992-8 (1987) BindingDB Entry DOI: 10.7270/Q29C6Z02 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Rattus norvegicus) | BDBM50021797 ((R,R+S,S) 1-(3-Acetylsulfanyl-2-methyl-propionyl)-...) | PDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 3.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibition of rat Angiotensin I converting enzyme (ACE), using Hip-Gly-Gly as synthetic substrate. | J Med Chem 30: 992-8 (1987) BindingDB Entry DOI: 10.7270/Q29C6Z02 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Rattus norvegicus) | BDBM50021782 ((RSR)1-[2-(1-Ethoxycarbonyl-3-phenyl-propylamino)-...) | PDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 5.70E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibition of rat Angiotensin I converting enzyme (ACE), using Hip-Gly-Gly as synthetic substrate. | J Med Chem 30: 992-8 (1987) BindingDB Entry DOI: 10.7270/Q29C6Z02 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Rattus norvegicus) | BDBM50021782 ((RSR)1-[2-(1-Ethoxycarbonyl-3-phenyl-propylamino)-...) | PDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 5.70E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibition of rat Angiotensin I converting enzyme (ACE), using Hip-Gly-Gly as synthetic substrate. | J Med Chem 30: 992-8 (1987) BindingDB Entry DOI: 10.7270/Q29C6Z02 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Rattus norvegicus) | BDBM50021798 ((SSS)2-(3-Methyl-1,4-dioxo-decahydro-pyrazino[1,2-...) | PDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibition of rat Angiotensin I converting enzyme (ACE), using Hip-Gly-Gly as synthetic substrate. | J Med Chem 30: 992-8 (1987) BindingDB Entry DOI: 10.7270/Q29C6Z02 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Rattus norvegicus) | BDBM50021782 ((RSR)1-[2-(1-Ethoxycarbonyl-3-phenyl-propylamino)-...) | PDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibition of rat Angiotensin I converting enzyme (ACE), using Hip-Gly-Gly as synthetic substrate. | J Med Chem 30: 992-8 (1987) BindingDB Entry DOI: 10.7270/Q29C6Z02 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||