 Found 26 hits with Last Name = 'maestro' and Initial = 'm'
Found 26 hits with Last Name = 'maestro' and Initial = 'm' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Vitamin D3 receptor
(Homo sapiens (Human)) | BDBM50200182
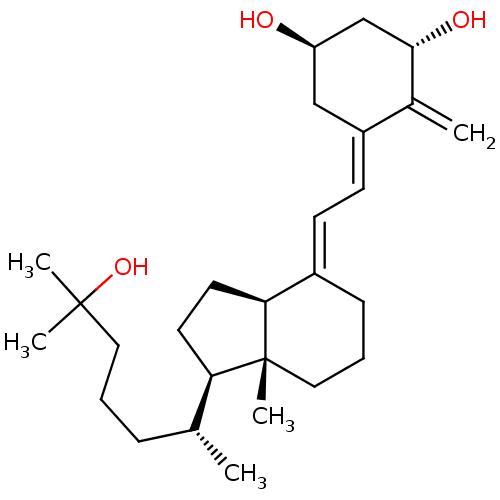
((1S,3R,5Z,7E)-9,10-secocholesta-5,7,10(19)-triene-...)Show SMILES C[C@H](CCCC(C)(C)O)[C@H]1CC[C@H]2\C(CCC[C@]12C)=C\C=C1\C[C@@H](O)C[C@H](O)C1=C |r| Show InChI InChI=1S/C27H44O3/c1-18(8-6-14-26(3,4)30)23-12-13-24-20(9-7-15-27(23,24)5)10-11-21-16-22(28)17-25(29)19(21)2/h10-11,18,22-25,28-30H,2,6-9,12-17H2,1,3-5H3/b20-10+,21-11-/t18-,22-,23-,24+,25+,27-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
Article
PubMed
| n/a | n/a | 1.20 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00900
BindingDB Entry DOI: 10.7270/Q27W6H5G |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Homo sapiens (Human)) | BDBM50200182

((1S,3R,5Z,7E)-9,10-secocholesta-5,7,10(19)-triene-...)Show SMILES C[C@H](CCCC(C)(C)O)[C@H]1CC[C@H]2\C(CCC[C@]12C)=C\C=C1\C[C@@H](O)C[C@H](O)C1=C |r| Show InChI InChI=1S/C27H44O3/c1-18(8-6-14-26(3,4)30)23-12-13-24-20(9-7-15-27(23,24)5)10-11-21-16-22(28)17-25(29)19(21)2/h10-11,18,22-25,28-30H,2,6-9,12-17H2,1,3-5H3/b20-10+,21-11-/t18-,22-,23-,24+,25+,27-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
Article
PubMed
| n/a | n/a | 1.24 | n/a | n/a | n/a | n/a | n/a | n/a |
Universidad de Santiago de Compostela
Curated by ChEMBL
| Assay Description
Binding affinity to VDR assessed as inhibition of fluorescent ligand by fluorescence polarization competition assay |
J Med Chem 55: 8642-56 (2012)
Article DOI: 10.1021/jm3008272
BindingDB Entry DOI: 10.7270/Q24Q7W49 |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Homo sapiens (Human)) | BDBM50200182

((1S,3R,5Z,7E)-9,10-secocholesta-5,7,10(19)-triene-...)Show SMILES C[C@H](CCCC(C)(C)O)[C@H]1CC[C@H]2\C(CCC[C@]12C)=C\C=C1\C[C@@H](O)C[C@H](O)C1=C |r| Show InChI InChI=1S/C27H44O3/c1-18(8-6-14-26(3,4)30)23-12-13-24-20(9-7-15-27(23,24)5)10-11-21-16-22(28)17-25(29)19(21)2/h10-11,18,22-25,28-30H,2,6-9,12-17H2,1,3-5H3/b20-10+,21-11-/t18-,22-,23-,24+,25+,27-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
Article
PubMed
| n/a | n/a | 1.30 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Santiago de Compostela
Curated by ChEMBL
| Assay Description
Displacement of fluormone from human full length VDR after 2 hrs by fluorescence polarization assay |
J Med Chem 61: 4928-4937 (2018)
Article DOI: 10.1021/acs.jmedchem.8b00337
BindingDB Entry DOI: 10.7270/Q22R3V8W |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Homo sapiens (Human)) | BDBM50455852
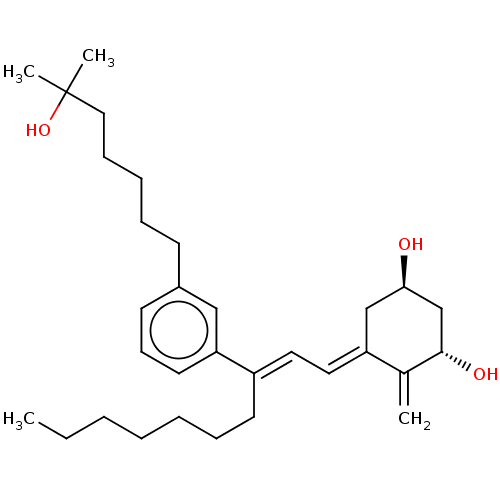
(CHEMBL4209452)Show SMILES CCCCCCC\C(=C/C=C1/C[C@@H](O)C[C@H](O)C1=C)c1cccc(CCCCCC(C)(C)O)c1 |r| Show InChI InChI=1S/C31H48O3/c1-5-6-7-8-11-16-26(18-19-27-22-29(32)23-30(33)24(27)2)28-17-13-15-25(21-28)14-10-9-12-20-31(3,4)34/h13,15,17-19,21,29-30,32-34H,2,5-12,14,16,20,22-23H2,1,3-4H3/b26-18+,27-19-/t29-,30+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | 3.5 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Santiago de Compostela
Curated by ChEMBL
| Assay Description
Displacement of fluormone from human full length VDR after 2 hrs by fluorescence polarization assay |
J Med Chem 61: 4928-4937 (2018)
Article DOI: 10.1021/acs.jmedchem.8b00337
BindingDB Entry DOI: 10.7270/Q22R3V8W |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Homo sapiens (Human)) | BDBM50455853
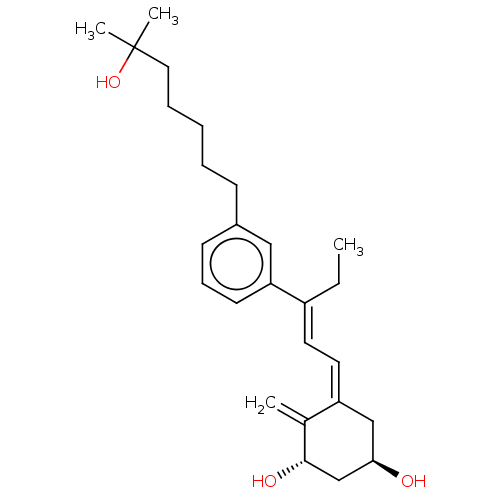
(CHEMBL4202828)Show SMILES CC\C(=C/C=C1/C[C@@H](O)C[C@H](O)C1=C)c1cccc(CCCCCC(C)(C)O)c1 |r| Show InChI InChI=1S/C26H38O3/c1-5-21(13-14-22-17-24(27)18-25(28)19(22)2)23-12-9-11-20(16-23)10-7-6-8-15-26(3,4)29/h9,11-14,16,24-25,27-29H,2,5-8,10,15,17-18H2,1,3-4H3/b21-13+,22-14-/t24-,25+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | 4.5 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Santiago de Compostela
Curated by ChEMBL
| Assay Description
Displacement of fluormone from human full length VDR after 2 hrs by fluorescence polarization assay |
J Med Chem 61: 4928-4937 (2018)
Article DOI: 10.1021/acs.jmedchem.8b00337
BindingDB Entry DOI: 10.7270/Q22R3V8W |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Homo sapiens (Human)) | BDBM50455855
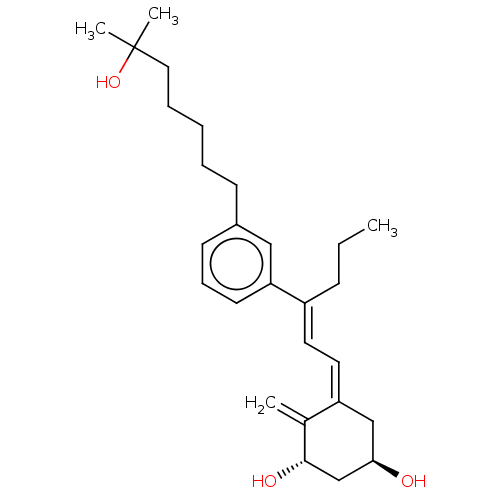
(CHEMBL4202601)Show SMILES CCC\C(=C/C=C1/C[C@@H](O)C[C@H](O)C1=C)c1cccc(CCCCCC(C)(C)O)c1 |r| Show InChI InChI=1S/C27H40O3/c1-5-10-22(14-15-23-18-25(28)19-26(29)20(23)2)24-13-9-12-21(17-24)11-7-6-8-16-27(3,4)30/h9,12-15,17,25-26,28-30H,2,5-8,10-11,16,18-19H2,1,3-4H3/b22-14+,23-15-/t25-,26+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | 5.60 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Santiago de Compostela
Curated by ChEMBL
| Assay Description
Displacement of fluormone from human full length VDR after 2 hrs by fluorescence polarization assay |
J Med Chem 61: 4928-4937 (2018)
Article DOI: 10.1021/acs.jmedchem.8b00337
BindingDB Entry DOI: 10.7270/Q22R3V8W |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Homo sapiens (Human)) | BDBM50455851
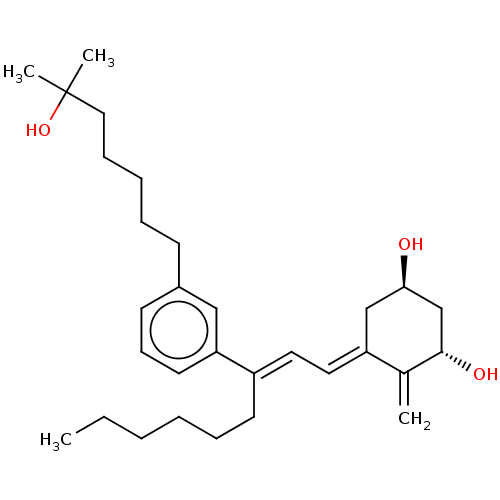
(CHEMBL4203540)Show SMILES CCCCCC\C(=C/C=C1/C[C@@H](O)C[C@H](O)C1=C)c1cccc(CCCCCC(C)(C)O)c1 |r| Show InChI InChI=1S/C30H46O3/c1-5-6-7-10-15-25(17-18-26-21-28(31)22-29(32)23(26)2)27-16-12-14-24(20-27)13-9-8-11-19-30(3,4)33/h12,14,16-18,20,28-29,31-33H,2,5-11,13,15,19,21-22H2,1,3-4H3/b25-17+,26-18-/t28-,29+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | 8.30 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Santiago de Compostela
Curated by ChEMBL
| Assay Description
Displacement of fluormone from human full length VDR after 2 hrs by fluorescence polarization assay |
J Med Chem 61: 4928-4937 (2018)
Article DOI: 10.1021/acs.jmedchem.8b00337
BindingDB Entry DOI: 10.7270/Q22R3V8W |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Homo sapiens (Human)) | BDBM50455854
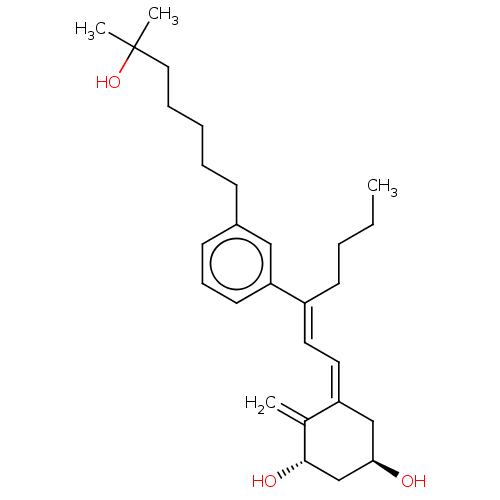
(CHEMBL4203464)Show SMILES CCCC\C(=C/C=C1/C[C@@H](O)C[C@H](O)C1=C)c1cccc(CCCCCC(C)(C)O)c1 |r| Show InChI InChI=1S/C28H42O3/c1-5-6-13-23(15-16-24-19-26(29)20-27(30)21(24)2)25-14-10-12-22(18-25)11-8-7-9-17-28(3,4)31/h10,12,14-16,18,26-27,29-31H,2,5-9,11,13,17,19-20H2,1,3-4H3/b23-15+,24-16-/t26-,27+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Santiago de Compostela
Curated by ChEMBL
| Assay Description
Displacement of fluormone from human full length VDR after 2 hrs by fluorescence polarization assay |
J Med Chem 61: 4928-4937 (2018)
Article DOI: 10.1021/acs.jmedchem.8b00337
BindingDB Entry DOI: 10.7270/Q22R3V8W |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Homo sapiens (Human)) | BDBM50589996
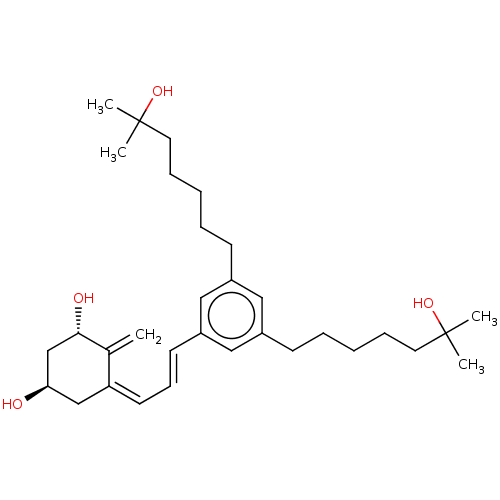
(CHEMBL5182683)Show SMILES CC(C)(O)CCCCCc1cc(CCCCCC(C)(C)O)cc(\C=C\C=C2\C[C@@H](O)C[C@H](O)C2=C)c1 |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | 15.7 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00900
BindingDB Entry DOI: 10.7270/Q27W6H5G |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Homo sapiens (Human)) | BDBM50397221
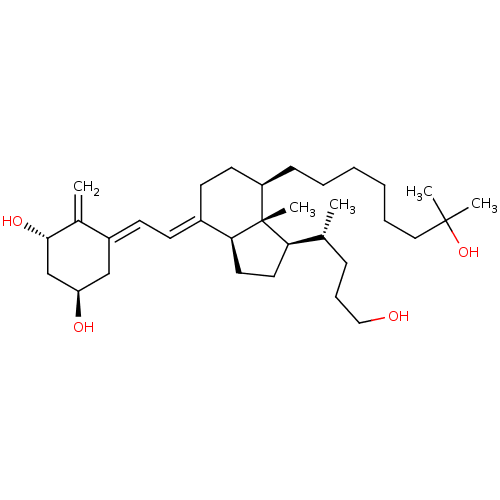
(CHEMBL2172537)Show SMILES C[C@H](CCCO)[C@H]1CC[C@H]2\C(CC[C@@H](CCCCCCC(C)(C)O)[C@]12C)=C\C=C1\C[C@@H](O)C[C@H](O)C1=C |r| Show InChI InChI=1S/C33H56O4/c1-23(11-10-20-34)29-17-18-30-25(13-14-26-21-28(35)22-31(36)24(26)2)15-16-27(33(29,30)5)12-8-6-7-9-19-32(3,4)37/h13-14,23,27-31,34-37H,2,6-12,15-22H2,1,3-5H3/b25-13+,26-14-/t23-,27-,28-,29-,30+,31+,33-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 15.7 | n/a | n/a | n/a | n/a | n/a | n/a |
Universidad de Santiago de Compostela
Curated by ChEMBL
| Assay Description
Binding affinity to VDR assessed as inhibition of fluorescent ligand by fluorescence polarization competition assay |
J Med Chem 55: 8642-56 (2012)
Article DOI: 10.1021/jm3008272
BindingDB Entry DOI: 10.7270/Q24Q7W49 |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Homo sapiens (Human)) | BDBM50397223
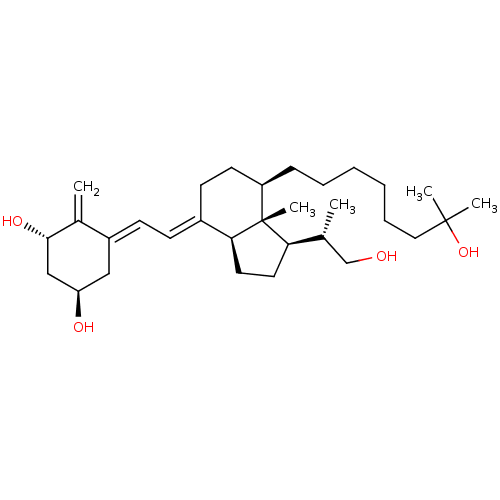
(CHEMBL2172539)Show SMILES C[C@H](CO)[C@H]1CC[C@H]2\C(CC[C@@H](CCCCCCC(C)(C)O)[C@]12C)=C\C=C1\C[C@@H](O)C[C@H](O)C1=C |r| Show InChI InChI=1S/C31H52O4/c1-21(20-32)27-15-16-28-23(11-12-24-18-26(33)19-29(34)22(24)2)13-14-25(31(27,28)5)10-8-6-7-9-17-30(3,4)35/h11-12,21,25-29,32-35H,2,6-10,13-20H2,1,3-5H3/b23-11+,24-12-/t21-,25-,26-,27-,28+,29+,31-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 20.9 | n/a | n/a | n/a | n/a | n/a | n/a |
Universidad de Santiago de Compostela
Curated by ChEMBL
| Assay Description
Binding affinity to VDR assessed as inhibition of fluorescent ligand by fluorescence polarization competition assay |
J Med Chem 55: 8642-56 (2012)
Article DOI: 10.1021/jm3008272
BindingDB Entry DOI: 10.7270/Q24Q7W49 |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Homo sapiens (Human)) | BDBM50397222
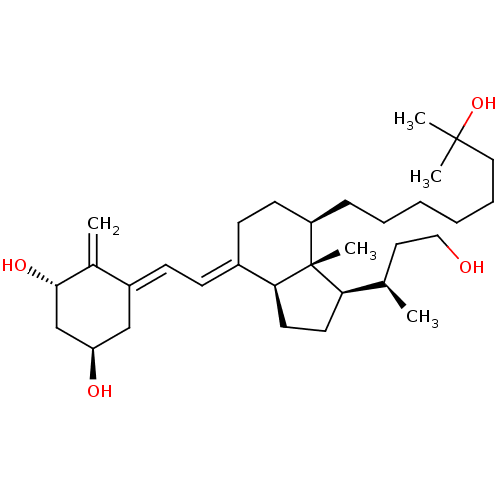
(CHEMBL2172538)Show SMILES C[C@H](CCO)[C@H]1CC[C@H]2\C(CC[C@@H](CCCCCCC(C)(C)O)[C@]12C)=C\C=C1\C[C@@H](O)C[C@H](O)C1=C |r| Show InChI InChI=1S/C32H54O4/c1-22(17-19-33)28-15-16-29-24(11-12-25-20-27(34)21-30(35)23(25)2)13-14-26(32(28,29)5)10-8-6-7-9-18-31(3,4)36/h11-12,22,26-30,33-36H,2,6-10,13-21H2,1,3-5H3/b24-11+,25-12-/t22-,26-,27-,28-,29+,30+,32-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 27.3 | n/a | n/a | n/a | n/a | n/a | n/a |
Universidad de Santiago de Compostela
Curated by ChEMBL
| Assay Description
Binding affinity to VDR assessed as inhibition of fluorescent ligand by fluorescence polarization competition assay |
J Med Chem 55: 8642-56 (2012)
Article DOI: 10.1021/jm3008272
BindingDB Entry DOI: 10.7270/Q24Q7W49 |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Homo sapiens (Human)) | BDBM50589995
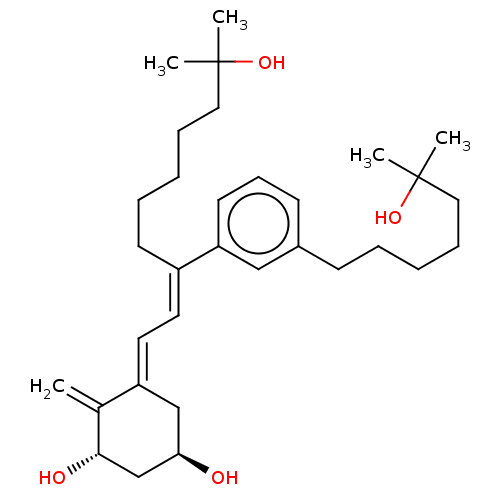
(CHEMBL5175578)Show SMILES CC(C)(O)CCCCC\C(=C/C=C1/C[C@@H](O)C[C@H](O)C1=C)c1cccc(CCCCCC(C)(C)O)c1 |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | 27.3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00900
BindingDB Entry DOI: 10.7270/Q27W6H5G |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Homo sapiens (Human)) | BDBM50455853

(CHEMBL4202828)Show SMILES CC\C(=C/C=C1/C[C@@H](O)C[C@H](O)C1=C)c1cccc(CCCCCC(C)(C)O)c1 |r| Show InChI InChI=1S/C26H38O3/c1-5-21(13-14-22-17-24(27)18-25(28)19(22)2)23-12-9-11-20(16-23)10-7-6-8-15-26(3,4)29/h9,11-14,16,24-25,27-29H,2,5-8,10,15,17-18H2,1,3-4H3/b21-13+,22-14-/t24-,25+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | n/a | n/a | 5.60 | n/a | n/a | n/a | n/a |
University of Santiago de Compostela
Curated by ChEMBL
| Assay Description
Agonist activity at VDR in human MCF7 cells assessed as increase in transcription of CYP24A1 gene after 24 hrs by luciferase reporter gene assay |
J Med Chem 61: 4928-4937 (2018)
Article DOI: 10.1021/acs.jmedchem.8b00337
BindingDB Entry DOI: 10.7270/Q22R3V8W |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Homo sapiens (Human)) | BDBM50455854

(CHEMBL4203464)Show SMILES CCCC\C(=C/C=C1/C[C@@H](O)C[C@H](O)C1=C)c1cccc(CCCCCC(C)(C)O)c1 |r| Show InChI InChI=1S/C28H42O3/c1-5-6-13-23(15-16-24-19-26(29)20-27(30)21(24)2)25-14-10-12-22(18-25)11-8-7-9-17-28(3,4)31/h10,12,14-16,18,26-27,29-31H,2,5-9,11,13,17,19-20H2,1,3-4H3/b23-15+,24-16-/t26-,27+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | n/a | n/a | 9 | n/a | n/a | n/a | n/a |
University of Santiago de Compostela
Curated by ChEMBL
| Assay Description
Agonist activity at VDR in human MCF7 cells assessed as increase in transcription of CYP24A1 gene after 24 hrs by luciferase reporter gene assay |
J Med Chem 61: 4928-4937 (2018)
Article DOI: 10.1021/acs.jmedchem.8b00337
BindingDB Entry DOI: 10.7270/Q22R3V8W |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Homo sapiens (Human)) | BDBM50455852

(CHEMBL4209452)Show SMILES CCCCCCC\C(=C/C=C1/C[C@@H](O)C[C@H](O)C1=C)c1cccc(CCCCCC(C)(C)O)c1 |r| Show InChI InChI=1S/C31H48O3/c1-5-6-7-8-11-16-26(18-19-27-22-29(32)23-30(33)24(27)2)28-17-13-15-25(21-28)14-10-9-12-20-31(3,4)34/h13,15,17-19,21,29-30,32-34H,2,5-12,14,16,20,22-23H2,1,3-4H3/b26-18+,27-19-/t29-,30+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | n/a | n/a | 29 | n/a | n/a | n/a | n/a |
University of Santiago de Compostela
Curated by ChEMBL
| Assay Description
Agonist activity at VDR in human MCF7 cells assessed as increase in transcription of CYP24A1 gene after 24 hrs by luciferase reporter gene assay |
J Med Chem 61: 4928-4937 (2018)
Article DOI: 10.1021/acs.jmedchem.8b00337
BindingDB Entry DOI: 10.7270/Q22R3V8W |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Homo sapiens (Human)) | BDBM50455851

(CHEMBL4203540)Show SMILES CCCCCC\C(=C/C=C1/C[C@@H](O)C[C@H](O)C1=C)c1cccc(CCCCCC(C)(C)O)c1 |r| Show InChI InChI=1S/C30H46O3/c1-5-6-7-10-15-25(17-18-26-21-28(31)22-29(32)23(26)2)27-16-12-14-24(20-27)13-9-8-11-19-30(3,4)33/h12,14,16-18,20,28-29,31-33H,2,5-11,13,15,19,21-22H2,1,3-4H3/b25-17+,26-18-/t28-,29+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | n/a | n/a | 10 | n/a | n/a | n/a | n/a |
University of Santiago de Compostela
Curated by ChEMBL
| Assay Description
Agonist activity at VDR in human MCF7 cells assessed as increase in transcription of CYP24A1 gene after 24 hrs by luciferase reporter gene assay |
J Med Chem 61: 4928-4937 (2018)
Article DOI: 10.1021/acs.jmedchem.8b00337
BindingDB Entry DOI: 10.7270/Q22R3V8W |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Homo sapiens (Human)) | BDBM50397223

(CHEMBL2172539)Show SMILES C[C@H](CO)[C@H]1CC[C@H]2\C(CC[C@@H](CCCCCCC(C)(C)O)[C@]12C)=C\C=C1\C[C@@H](O)C[C@H](O)C1=C |r| Show InChI InChI=1S/C31H52O4/c1-21(20-32)27-15-16-28-23(11-12-24-18-26(33)19-29(34)22(24)2)13-14-25(31(27,28)5)10-8-6-7-9-17-30(3,4)35/h11-12,21,25-29,32-35H,2,6-10,13-20H2,1,3-5H3/b23-11+,24-12-/t21-,25-,26-,27-,28+,29+,31-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 8.19 | n/a | n/a | n/a | n/a |
Universidad de Santiago de Compostela
Curated by ChEMBL
| Assay Description
Agonist activity at VDR in human MCF7 cells assessed as transcription of CYP24A1 gene after 24 hrs by luciferase reporter gene assay |
J Med Chem 55: 8642-56 (2012)
Article DOI: 10.1021/jm3008272
BindingDB Entry DOI: 10.7270/Q24Q7W49 |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Homo sapiens (Human)) | BDBM50397222

(CHEMBL2172538)Show SMILES C[C@H](CCO)[C@H]1CC[C@H]2\C(CC[C@@H](CCCCCCC(C)(C)O)[C@]12C)=C\C=C1\C[C@@H](O)C[C@H](O)C1=C |r| Show InChI InChI=1S/C32H54O4/c1-22(17-19-33)28-15-16-29-24(11-12-25-20-27(34)21-30(35)23(25)2)13-14-26(32(28,29)5)10-8-6-7-9-18-31(3,4)36/h11-12,22,26-30,33-36H,2,6-10,13-21H2,1,3-5H3/b24-11+,25-12-/t22-,26-,27-,28-,29+,30+,32-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 9.04 | n/a | n/a | n/a | n/a |
Universidad de Santiago de Compostela
Curated by ChEMBL
| Assay Description
Agonist activity at VDR in human MCF7 cells assessed as transcription of CYP24A1 gene after 24 hrs by luciferase reporter gene assay |
J Med Chem 55: 8642-56 (2012)
Article DOI: 10.1021/jm3008272
BindingDB Entry DOI: 10.7270/Q24Q7W49 |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Homo sapiens (Human)) | BDBM50200182

((1S,3R,5Z,7E)-9,10-secocholesta-5,7,10(19)-triene-...)Show SMILES C[C@H](CCCC(C)(C)O)[C@H]1CC[C@H]2\C(CCC[C@]12C)=C\C=C1\C[C@@H](O)C[C@H](O)C1=C |r| Show InChI InChI=1S/C27H44O3/c1-18(8-6-14-26(3,4)30)23-12-13-24-20(9-7-15-27(23,24)5)10-11-21-16-22(28)17-25(29)19(21)2/h10-11,18,22-25,28-30H,2,6-9,12-17H2,1,3-5H3/b20-10+,21-11-/t18-,22-,23-,24+,25+,27-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
Article
PubMed
| n/a | n/a | n/a | n/a | 3.28 | n/a | n/a | n/a | n/a |
Universidad de Santiago de Compostela
Curated by ChEMBL
| Assay Description
Agonist activity at VDR in human MCF7 cells assessed as transcription of CYP24A1 gene after 24 hrs by luciferase reporter gene assay |
J Med Chem 55: 8642-56 (2012)
Article DOI: 10.1021/jm3008272
BindingDB Entry DOI: 10.7270/Q24Q7W49 |
More data for this
Ligand-Target Pair | |
1,25-dihydroxyvitamin D(3) 24-hydroxylase, mitochondrial
(Homo sapiens (Human)) | BDBM50200182

((1S,3R,5Z,7E)-9,10-secocholesta-5,7,10(19)-triene-...)Show SMILES C[C@H](CCCC(C)(C)O)[C@H]1CC[C@H]2\C(CCC[C@]12C)=C\C=C1\C[C@@H](O)C[C@H](O)C1=C |r| Show InChI InChI=1S/C27H44O3/c1-18(8-6-14-26(3,4)30)23-12-13-24-20(9-7-15-27(23,24)5)10-11-21-16-22(28)17-25(29)19(21)2/h10-11,18,22-25,28-30H,2,6-9,12-17H2,1,3-5H3/b20-10+,21-11-/t18-,22-,23-,24+,25+,27-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
Article
PubMed
| n/a | n/a | n/a | n/a | 3 | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00900
BindingDB Entry DOI: 10.7270/Q27W6H5G |
More data for this
Ligand-Target Pair | |
1,25-dihydroxyvitamin D(3) 24-hydroxylase, mitochondrial
(Homo sapiens (Human)) | BDBM50589995

(CHEMBL5175578)Show SMILES CC(C)(O)CCCCC\C(=C/C=C1/C[C@@H](O)C[C@H](O)C1=C)c1cccc(CCCCCC(C)(C)O)c1 |r| | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | n/a | n/a | 5.60 | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00900
BindingDB Entry DOI: 10.7270/Q27W6H5G |
More data for this
Ligand-Target Pair | |
1,25-dihydroxyvitamin D(3) 24-hydroxylase, mitochondrial
(Homo sapiens (Human)) | BDBM50589996

(CHEMBL5182683)Show SMILES CC(C)(O)CCCCCc1cc(CCCCCC(C)(C)O)cc(\C=C\C=C2\C[C@@H](O)C[C@H](O)C2=C)c1 |r| | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | n/a | n/a | 3 | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00900
BindingDB Entry DOI: 10.7270/Q27W6H5G |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Homo sapiens (Human)) | BDBM50397221

(CHEMBL2172537)Show SMILES C[C@H](CCCO)[C@H]1CC[C@H]2\C(CC[C@@H](CCCCCCC(C)(C)O)[C@]12C)=C\C=C1\C[C@@H](O)C[C@H](O)C1=C |r| Show InChI InChI=1S/C33H56O4/c1-23(11-10-20-34)29-17-18-30-25(13-14-26-21-28(35)22-31(36)24(26)2)15-16-27(33(29,30)5)12-8-6-7-9-19-32(3,4)37/h13-14,23,27-31,34-37H,2,6-12,15-22H2,1,3-5H3/b25-13+,26-14-/t23-,27-,28-,29-,30+,31+,33-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 5.74 | n/a | n/a | n/a | n/a |
Universidad de Santiago de Compostela
Curated by ChEMBL
| Assay Description
Agonist activity at VDR in human MCF7 cells assessed as transcription of CYP24A1 gene after 24 hrs by luciferase reporter gene assay |
J Med Chem 55: 8642-56 (2012)
Article DOI: 10.1021/jm3008272
BindingDB Entry DOI: 10.7270/Q24Q7W49 |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Homo sapiens (Human)) | BDBM50200182

((1S,3R,5Z,7E)-9,10-secocholesta-5,7,10(19)-triene-...)Show SMILES C[C@H](CCCC(C)(C)O)[C@H]1CC[C@H]2\C(CCC[C@]12C)=C\C=C1\C[C@@H](O)C[C@H](O)C1=C |r| Show InChI InChI=1S/C27H44O3/c1-18(8-6-14-26(3,4)30)23-12-13-24-20(9-7-15-27(23,24)5)10-11-21-16-22(28)17-25(29)19(21)2/h10-11,18,22-25,28-30H,2,6-9,12-17H2,1,3-5H3/b20-10+,21-11-/t18-,22-,23-,24+,25+,27-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
Article
PubMed
| n/a | n/a | n/a | n/a | 1.60 | n/a | n/a | n/a | n/a |
University of Santiago de Compostela
Curated by ChEMBL
| Assay Description
Agonist activity at VDR in human MCF7 cells assessed as increase in transcription of CYP24A1 gene after 24 hrs by luciferase reporter gene assay |
J Med Chem 61: 4928-4937 (2018)
Article DOI: 10.1021/acs.jmedchem.8b00337
BindingDB Entry DOI: 10.7270/Q22R3V8W |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Homo sapiens (Human)) | BDBM50455855

(CHEMBL4202601)Show SMILES CCC\C(=C/C=C1/C[C@@H](O)C[C@H](O)C1=C)c1cccc(CCCCCC(C)(C)O)c1 |r| Show InChI InChI=1S/C27H40O3/c1-5-10-22(14-15-23-18-25(28)19-26(29)20(23)2)24-13-9-12-21(17-24)11-7-6-8-16-27(3,4)30/h9,12-15,17,25-26,28-30H,2,5-8,10-11,16,18-19H2,1,3-4H3/b22-14+,23-15-/t25-,26+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | n/a | n/a | 8.60 | n/a | n/a | n/a | n/a |
University of Santiago de Compostela
Curated by ChEMBL
| Assay Description
Agonist activity at VDR in human MCF7 cells assessed as increase in transcription of CYP24A1 gene after 24 hrs by luciferase reporter gene assay |
J Med Chem 61: 4928-4937 (2018)
Article DOI: 10.1021/acs.jmedchem.8b00337
BindingDB Entry DOI: 10.7270/Q22R3V8W |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data