 Found 105 hits with Last Name = 'pavana' and Initial = 'rk'
Found 105 hits with Last Name = 'pavana' and Initial = 'rk' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Epidermal growth factor receptor
(Homo sapiens (Human)) | BDBM50232828
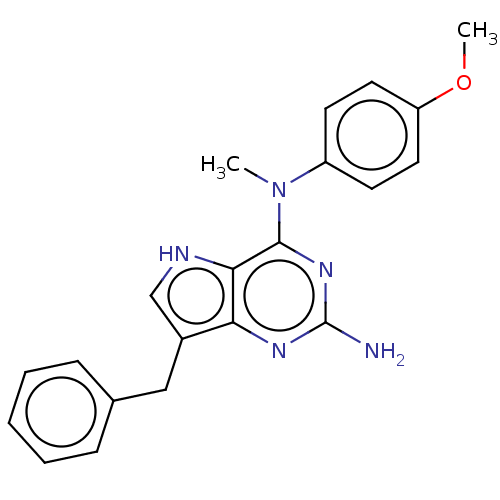
(CHEMBL4099329)Show SMILES COc1ccc(cc1)N(C)c1nc(N)nc2c(Cc3ccccc3)c[nH]c12 Show InChI InChI=1S/C21H21N5O/c1-26(16-8-10-17(27-2)11-9-16)20-19-18(24-21(22)25-20)15(13-23-19)12-14-6-4-3-5-7-14/h3-11,13,23H,12H2,1-2H3,(H2,22,24,25) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.10 | n/a | n/a | n/a | n/a | n/a | n/a |
Duquesne University
Curated by ChEMBL
| Assay Description
Inhibition of EGFR in human A431 cells assessed as reduction in EGF induced phosphorylation preincubated for 60 mins followed by EGF addition measure... |
Bioorg Med Chem 25: 545-556 (2017)
Article DOI: 10.1016/j.bmc.2016.11.026
BindingDB Entry DOI: 10.7270/Q27W6FF7 |
More data for this
Ligand-Target Pair | |
Epidermal growth factor receptor
(Homo sapiens (Human)) | BDBM5446

(CHEMBL553 | ERLOTINIB HYDROCHLORIDE | Erlotinib | ...)Show InChI InChI=1S/C22H23N3O4/c1-4-16-6-5-7-17(12-16)25-22-18-13-20(28-10-8-26-2)21(29-11-9-27-3)14-19(18)23-15-24-22/h1,5-7,12-15H,8-11H2,2-3H3,(H,23,24,25) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
MMDB
PDB
Article
PubMed
| n/a | n/a | 1.20 | n/a | n/a | n/a | n/a | n/a | n/a |
Duquesne University
Curated by ChEMBL
| Assay Description
Inhibition of EGFR in human A431 cells assessed as reduction in EGF induced phosphorylation preincubated for 60 mins followed by EGF addition measure... |
Bioorg Med Chem 25: 545-556 (2017)
Article DOI: 10.1016/j.bmc.2016.11.026
BindingDB Entry DOI: 10.7270/Q27W6FF7 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Epidermal growth factor receptor
(Homo sapiens (Human)) | BDBM50232824
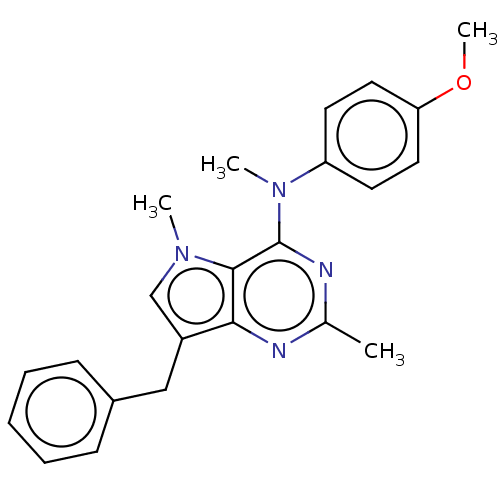
(CHEMBL4060996)Show SMILES Cl.COc1ccc(cc1)N(C)c1nc(C)nc2c(Cc3ccccc3)cn(C)c12 Show InChI InChI=1S/C23H24N4O/c1-16-24-21-18(14-17-8-6-5-7-9-17)15-26(2)22(21)23(25-16)27(3)19-10-12-20(28-4)13-11-19/h5-13,15H,14H2,1-4H3 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.30 | n/a | n/a | n/a | n/a | n/a | n/a |
Duquesne University
Curated by ChEMBL
| Assay Description
Inhibition of EGFR in human A431 cells assessed as reduction in EGF induced phosphorylation preincubated for 60 mins followed by EGF addition measure... |
Bioorg Med Chem 25: 545-556 (2017)
Article DOI: 10.1016/j.bmc.2016.11.026
BindingDB Entry DOI: 10.7270/Q27W6FF7 |
More data for this
Ligand-Target Pair | |
Epidermal growth factor receptor
(Homo sapiens (Human)) | BDBM50232823
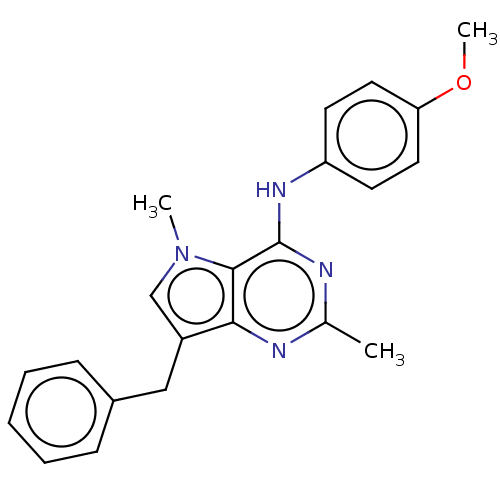
(CHEMBL4099682)Show SMILES Cl.COc1ccc(Nc2nc(C)nc3c(Cc4ccccc4)cn(C)c23)cc1 Show InChI InChI=1S/C22H22N4O/c1-15-23-20-17(13-16-7-5-4-6-8-16)14-26(2)21(20)22(24-15)25-18-9-11-19(27-3)12-10-18/h4-12,14H,13H2,1-3H3,(H,23,24,25) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.60 | n/a | n/a | n/a | n/a | n/a | n/a |
Duquesne University
Curated by ChEMBL
| Assay Description
Inhibition of EGFR in human A431 cells assessed as reduction in EGF induced phosphorylation preincubated for 60 mins followed by EGF addition measure... |
Bioorg Med Chem 25: 545-556 (2017)
Article DOI: 10.1016/j.bmc.2016.11.026
BindingDB Entry DOI: 10.7270/Q27W6FF7 |
More data for this
Ligand-Target Pair | |
Epidermal growth factor receptor
(Homo sapiens (Human)) | BDBM50232827
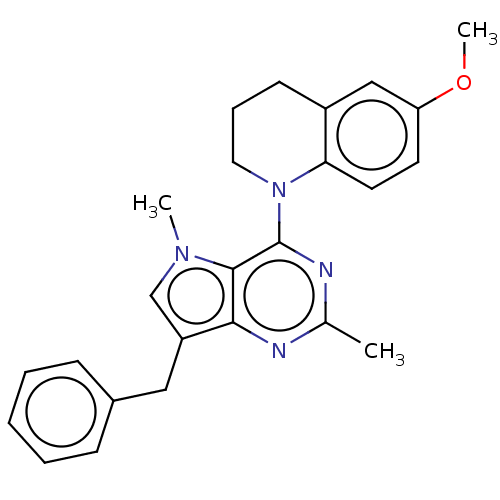
(CHEMBL4104515)Show SMILES Cl.COc1ccc2N(CCCc2c1)c1nc(C)nc2c(Cc3ccccc3)cn(C)c12 Show InChI InChI=1S/C25H26N4O/c1-17-26-23-20(14-18-8-5-4-6-9-18)16-28(2)24(23)25(27-17)29-13-7-10-19-15-21(30-3)11-12-22(19)29/h4-6,8-9,11-12,15-16H,7,10,13-14H2,1-3H3 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4.90 | n/a | n/a | n/a | n/a | n/a | n/a |
Duquesne University
Curated by ChEMBL
| Assay Description
Inhibition of EGFR in human A431 cells assessed as reduction in EGF induced phosphorylation preincubated for 60 mins followed by EGF addition measure... |
Bioorg Med Chem 25: 545-556 (2017)
Article DOI: 10.1016/j.bmc.2016.11.026
BindingDB Entry DOI: 10.7270/Q27W6FF7 |
More data for this
Ligand-Target Pair | |
Platelet-derived growth factor receptor beta
(Homo sapiens (Human)) | BDBM50232823

(CHEMBL4099682)Show SMILES Cl.COc1ccc(Nc2nc(C)nc3c(Cc4ccccc4)cn(C)c23)cc1 Show InChI InChI=1S/C22H22N4O/c1-15-23-20-17(13-16-7-5-4-6-8-16)14-26(2)21(20)22(24-15)25-18-9-11-19(27-3)12-10-18/h4-12,14H,13H2,1-3H3,(H,23,24,25) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 7.20 | n/a | n/a | n/a | n/a | n/a | n/a |
Duquesne University
Curated by ChEMBL
| Assay Description
Inhibition of PDGFR-beta in human SF539 cells assessed as reduction in PDGF-BB induced phosphorylation preincubated for 60 mins followed by PDGF-BB a... |
Bioorg Med Chem 25: 545-556 (2017)
Article DOI: 10.1016/j.bmc.2016.11.026
BindingDB Entry DOI: 10.7270/Q27W6FF7 |
More data for this
Ligand-Target Pair | |
Platelet-derived growth factor receptor beta
(Homo sapiens (Human)) | BDBM50232828

(CHEMBL4099329)Show SMILES COc1ccc(cc1)N(C)c1nc(N)nc2c(Cc3ccccc3)c[nH]c12 Show InChI InChI=1S/C21H21N5O/c1-26(16-8-10-17(27-2)11-9-16)20-19-18(24-21(22)25-20)15(13-23-19)12-14-6-4-3-5-7-14/h3-11,13,23H,12H2,1-2H3,(H2,22,24,25) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 8.10 | n/a | n/a | n/a | n/a | n/a | n/a |
Duquesne University
Curated by ChEMBL
| Assay Description
Inhibition of PDGFR-beta in human SF539 cells assessed as reduction in PDGF-BB induced phosphorylation preincubated for 60 mins followed by PDGF-BB a... |
Bioorg Med Chem 25: 545-556 (2017)
Article DOI: 10.1016/j.bmc.2016.11.026
BindingDB Entry DOI: 10.7270/Q27W6FF7 |
More data for this
Ligand-Target Pair | |
Platelet-derived growth factor receptor beta
(Homo sapiens (Human)) | BDBM50232824

(CHEMBL4060996)Show SMILES Cl.COc1ccc(cc1)N(C)c1nc(C)nc2c(Cc3ccccc3)cn(C)c12 Show InChI InChI=1S/C23H24N4O/c1-16-24-21-18(14-17-8-6-5-7-9-17)15-26(2)22(21)23(25-16)27(3)19-10-12-20(28-4)13-11-19/h5-13,15H,14H2,1-4H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
Duquesne University
Curated by ChEMBL
| Assay Description
Inhibition of PDGFR-beta in human SF539 cells assessed as reduction in PDGF-BB induced phosphorylation preincubated for 60 mins followed by PDGF-BB a... |
Bioorg Med Chem 25: 545-556 (2017)
Article DOI: 10.1016/j.bmc.2016.11.026
BindingDB Entry DOI: 10.7270/Q27W6FF7 |
More data for this
Ligand-Target Pair | |
Vascular endothelial growth factor receptor 2
(Homo sapiens (Human)) | BDBM4810
((3Z)-3-[(3,5-dimethyl-1H-pyrrol-2-yl)methylidene]-...)Show InChI InChI=1S/C15H14N2O/c1-9-7-10(2)16-14(9)8-12-11-5-3-4-6-13(11)17-15(12)18/h3-8,16H,1-2H3,(H,17,18)/b12-8- | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 13 | n/a | n/a | n/a | n/a | n/a | n/a |
Duquesne University
Curated by ChEMBL
| Assay Description
Inhibition of VEGFR-2 in human U251 cells assessed as reduction in VEGF induced phosphorylation preincubated for 60 mins followed by VEGF addition me... |
Bioorg Med Chem 25: 545-556 (2017)
Article DOI: 10.1016/j.bmc.2016.11.026
BindingDB Entry DOI: 10.7270/Q27W6FF7 |
More data for this
Ligand-Target Pair | |
Vascular endothelial growth factor receptor 2
(Homo sapiens (Human)) | BDBM4810

((3Z)-3-[(3,5-dimethyl-1H-pyrrol-2-yl)methylidene]-...)Show InChI InChI=1S/C15H14N2O/c1-9-7-10(2)16-14(9)8-12-11-5-3-4-6-13(11)17-15(12)18/h3-8,16H,1-2H3,(H,17,18)/b12-8- | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 13 | n/a | n/a | n/a | n/a | n/a | n/a |
Duquesne University
Curated by ChEMBL
| Assay Description
Inhibition of VEGFR2 in human U251 cells by phosphotyrosine ELISA |
ACS Med Chem Lett 5: 480-4 (2014)
Article DOI: 10.1021/ml4004793
BindingDB Entry DOI: 10.7270/Q2H133KM |
More data for this
Ligand-Target Pair | |
Vascular endothelial growth factor receptor 2
(Homo sapiens (Human)) | BDBM4814
(CHEMBL535 | N-[2-(diethylamino)ethyl]-5-[(Z)-(5-fl...)Show SMILES CCN(CC)CCNC(=O)c1c(C)[nH]c(\C=C2/C(=O)Nc3ccc(F)cc23)c1C Show InChI InChI=1S/C22H27FN4O2/c1-5-27(6-2)10-9-24-22(29)20-13(3)19(25-14(20)4)12-17-16-11-15(23)7-8-18(16)26-21(17)28/h7-8,11-12,25H,5-6,9-10H2,1-4H3,(H,24,29)(H,26,28)/b17-12- | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PDB
Article
PubMed
| n/a | n/a | 19 | n/a | n/a | n/a | n/a | n/a | n/a |
Duquesne University
Curated by ChEMBL
| Assay Description
Inhibition of VEGFR-2 in human U251 cells assessed as reduction in VEGF induced phosphorylation preincubated for 60 mins followed by VEGF addition me... |
Bioorg Med Chem 25: 545-556 (2017)
Article DOI: 10.1016/j.bmc.2016.11.026
BindingDB Entry DOI: 10.7270/Q27W6FF7 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Vascular endothelial growth factor receptor 2
(Homo sapiens (Human)) | BDBM4814

(CHEMBL535 | N-[2-(diethylamino)ethyl]-5-[(Z)-(5-fl...)Show SMILES CCN(CC)CCNC(=O)c1c(C)[nH]c(\C=C2/C(=O)Nc3ccc(F)cc23)c1C Show InChI InChI=1S/C22H27FN4O2/c1-5-27(6-2)10-9-24-22(29)20-13(3)19(25-14(20)4)12-17-16-11-15(23)7-8-18(16)26-21(17)28/h7-8,11-12,25H,5-6,9-10H2,1-4H3,(H,24,29)(H,26,28)/b17-12- | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PDB
Article
PubMed
| n/a | n/a | 19 | n/a | n/a | n/a | n/a | n/a | n/a |
Duquesne University
Curated by ChEMBL
| Assay Description
Inhibition of VEGFR2 in human U251 cells by phosphotyrosine ELISA |
ACS Med Chem Lett 5: 480-4 (2014)
Article DOI: 10.1021/ml4004793
BindingDB Entry DOI: 10.7270/Q2H133KM |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Tryptophan 2,3-dioxygenase
(Homo sapiens (Human)) | BDBM50511732
(CHEMBL4434743) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 20 | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human TDO using L-tryptophan as substrate preincubated for 5 mins followed by substrate addition and measured after 15 mins... |
ACS Med Chem Lett 11: 541-549 (2020)
Article DOI: 10.1021/acsmedchemlett.0c00004
BindingDB Entry DOI: 10.7270/Q2RR22JS |
More data for this
Ligand-Target Pair | |
Tryptophan 2,3-dioxygenase
(Homo sapiens (Human)) | BDBM370419
(2-(5H-imidazo[5,1-a]isoindol-5-yl)ethanol | US1023...)Show InChI InChI=1S/C12H12N2O/c15-6-5-11-9-3-1-2-4-10(9)12-7-13-8-14(11)12/h1-4,7-8,11,15H,5-6H2 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 20 | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human TDO using L-tryptophan as substrate preincubated for 5 mins followed by substrate addition and measured after 15 mins... |
ACS Med Chem Lett 11: 541-549 (2020)
Article DOI: 10.1021/acsmedchemlett.0c00004
BindingDB Entry DOI: 10.7270/Q2RR22JS |
More data for this
Ligand-Target Pair | |
Vascular endothelial growth factor receptor 2
(Homo sapiens (Human)) | BDBM50019928

(CHEMBL3287222)Show SMILES Cl.COc1ccc(cc1)N(C)c1nc(C)nc2c(Cc3ccccc3)c[nH]c12 Show InChI InChI=1S/C22H22N4O.ClH/c1-15-24-20-17(13-16-7-5-4-6-8-16)14-23-21(20)22(25-15)26(2)18-9-11-19(27-3)12-10-18;/h4-12,14,23H,13H2,1-3H3;1H | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 21 | n/a | n/a | n/a | n/a | n/a | n/a |
Duquesne University
Curated by ChEMBL
| Assay Description
Inhibition of VEGFR2 in human U251 cells by phosphotyrosine ELISA |
ACS Med Chem Lett 5: 480-4 (2014)
Article DOI: 10.1021/ml4004793
BindingDB Entry DOI: 10.7270/Q2H133KM |
More data for this
Ligand-Target Pair | |
Vascular endothelial growth factor receptor 2
(Homo sapiens (Human)) | BDBM50232823

(CHEMBL4099682)Show SMILES Cl.COc1ccc(Nc2nc(C)nc3c(Cc4ccccc4)cn(C)c23)cc1 Show InChI InChI=1S/C22H22N4O/c1-15-23-20-17(13-16-7-5-4-6-8-16)14-26(2)21(20)22(24-15)25-18-9-11-19(27-3)12-10-18/h4-12,14H,13H2,1-3H3,(H,23,24,25) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 27 | n/a | n/a | n/a | n/a | n/a | n/a |
Duquesne University
Curated by ChEMBL
| Assay Description
Inhibition of VEGFR-2 in human U251 cells assessed as reduction in VEGF induced phosphorylation preincubated for 60 mins followed by VEGF addition me... |
Bioorg Med Chem 25: 545-556 (2017)
Article DOI: 10.1016/j.bmc.2016.11.026
BindingDB Entry DOI: 10.7270/Q27W6FF7 |
More data for this
Ligand-Target Pair | |
Vascular endothelial growth factor receptor 2
(Homo sapiens (Human)) | BDBM50232828

(CHEMBL4099329)Show SMILES COc1ccc(cc1)N(C)c1nc(N)nc2c(Cc3ccccc3)c[nH]c12 Show InChI InChI=1S/C21H21N5O/c1-26(16-8-10-17(27-2)11-9-16)20-19-18(24-21(22)25-20)15(13-23-19)12-14-6-4-3-5-7-14/h3-11,13,23H,12H2,1-2H3,(H2,22,24,25) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 27 | n/a | n/a | n/a | n/a | n/a | n/a |
Duquesne University
Curated by ChEMBL
| Assay Description
Inhibition of VEGFR-2 in human U251 cells assessed as reduction in VEGF induced phosphorylation preincubated for 60 mins followed by VEGF addition me... |
Bioorg Med Chem 25: 545-556 (2017)
Article DOI: 10.1016/j.bmc.2016.11.026
BindingDB Entry DOI: 10.7270/Q27W6FF7 |
More data for this
Ligand-Target Pair | |
Vascular endothelial growth factor receptor 2
(Homo sapiens (Human)) | BDBM50019931

(CHEMBL3287219)Show SMILES Cl.COc1ccc(Nc2nc(C)nc3c(Cc4ccccc4)c[nH]c23)c(OC)c1 Show InChI InChI=1S/C22H22N4O2.ClH/c1-14-24-20-16(11-15-7-5-4-6-8-15)13-23-21(20)22(25-14)26-18-10-9-17(27-2)12-19(18)28-3;/h4-10,12-13,23H,11H2,1-3H3,(H,24,25,26);1H | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 30 | n/a | n/a | n/a | n/a | n/a | n/a |
Duquesne University
Curated by ChEMBL
| Assay Description
Inhibition of VEGFR2 in human U251 cells by phosphotyrosine ELISA |
ACS Med Chem Lett 5: 480-4 (2014)
Article DOI: 10.1021/ml4004793
BindingDB Entry DOI: 10.7270/Q2H133KM |
More data for this
Ligand-Target Pair | |
Platelet-derived growth factor receptor beta
(Homo sapiens (Human)) | BDBM50232827

(CHEMBL4104515)Show SMILES Cl.COc1ccc2N(CCCc2c1)c1nc(C)nc2c(Cc3ccccc3)cn(C)c12 Show InChI InChI=1S/C25H26N4O/c1-17-26-23-20(14-18-8-5-4-6-9-18)16-28(2)24(23)25(27-17)29-13-7-10-19-15-21(30-3)11-12-22(19)29/h4-6,8-9,11-12,15-16H,7,10,13-14H2,1-3H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 30 | n/a | n/a | n/a | n/a | n/a | n/a |
Duquesne University
Curated by ChEMBL
| Assay Description
Inhibition of PDGFR-beta in human SF539 cells assessed as reduction in PDGF-BB induced phosphorylation preincubated for 60 mins followed by PDGF-BB a... |
Bioorg Med Chem 25: 545-556 (2017)
Article DOI: 10.1016/j.bmc.2016.11.026
BindingDB Entry DOI: 10.7270/Q27W6FF7 |
More data for this
Ligand-Target Pair | |
Vascular endothelial growth factor receptor 2
(Homo sapiens (Human)) | BDBM50232824

(CHEMBL4060996)Show SMILES Cl.COc1ccc(cc1)N(C)c1nc(C)nc2c(Cc3ccccc3)cn(C)c12 Show InChI InChI=1S/C23H24N4O/c1-16-24-21-18(14-17-8-6-5-7-9-17)15-26(2)22(21)23(25-16)27(3)19-10-12-20(28-4)13-11-19/h5-13,15H,14H2,1-4H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 33 | n/a | n/a | n/a | n/a | n/a | n/a |
Duquesne University
Curated by ChEMBL
| Assay Description
Inhibition of VEGFR-2 in human U251 cells assessed as reduction in VEGF induced phosphorylation preincubated for 60 mins followed by VEGF addition me... |
Bioorg Med Chem 25: 545-556 (2017)
Article DOI: 10.1016/j.bmc.2016.11.026
BindingDB Entry DOI: 10.7270/Q27W6FF7 |
More data for this
Ligand-Target Pair | |
Vascular endothelial growth factor receptor 2
(Homo sapiens (Human)) | BDBM50232827

(CHEMBL4104515)Show SMILES Cl.COc1ccc2N(CCCc2c1)c1nc(C)nc2c(Cc3ccccc3)cn(C)c12 Show InChI InChI=1S/C25H26N4O/c1-17-26-23-20(14-18-8-5-4-6-9-18)16-28(2)24(23)25(27-17)29-13-7-10-19-15-21(30-3)11-12-22(19)29/h4-6,8-9,11-12,15-16H,7,10,13-14H2,1-3H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 33 | n/a | n/a | n/a | n/a | n/a | n/a |
Duquesne University
Curated by ChEMBL
| Assay Description
Inhibition of VEGFR-2 in human U251 cells assessed as reduction in VEGF induced phosphorylation preincubated for 60 mins followed by VEGF addition me... |
Bioorg Med Chem 25: 545-556 (2017)
Article DOI: 10.1016/j.bmc.2016.11.026
BindingDB Entry DOI: 10.7270/Q27W6FF7 |
More data for this
Ligand-Target Pair | |
Epidermal growth factor receptor
(Homo sapiens (Human)) | BDBM50232826
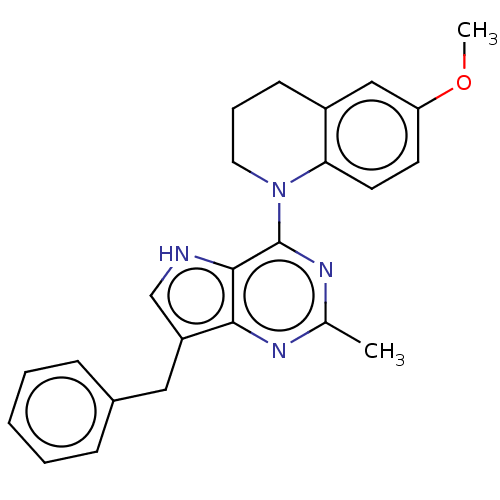
(CHEMBL4069672)Show SMILES Cl.COc1ccc2N(CCCc2c1)c1nc(C)nc2c(Cc3ccccc3)c[nH]c12 Show InChI InChI=1S/C24H24N4O/c1-16-26-22-19(13-17-7-4-3-5-8-17)15-25-23(22)24(27-16)28-12-6-9-18-14-20(29-2)10-11-21(18)28/h3-5,7-8,10-11,14-15,25H,6,9,12-13H2,1-2H3 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 34 | n/a | n/a | n/a | n/a | n/a | n/a |
Duquesne University
Curated by ChEMBL
| Assay Description
Inhibition of EGFR in human A431 cells assessed as reduction in EGF induced phosphorylation preincubated for 60 mins followed by EGF addition measure... |
Bioorg Med Chem 25: 545-556 (2017)
Article DOI: 10.1016/j.bmc.2016.11.026
BindingDB Entry DOI: 10.7270/Q27W6FF7 |
More data for this
Ligand-Target Pair | |
Vascular endothelial growth factor receptor 2
(Homo sapiens (Human)) | BDBM50232826

(CHEMBL4069672)Show SMILES Cl.COc1ccc2N(CCCc2c1)c1nc(C)nc2c(Cc3ccccc3)c[nH]c12 Show InChI InChI=1S/C24H24N4O/c1-16-26-22-19(13-17-7-4-3-5-8-17)15-25-23(22)24(27-16)28-12-6-9-18-14-20(29-2)10-11-21(18)28/h3-5,7-8,10-11,14-15,25H,6,9,12-13H2,1-2H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 39 | n/a | n/a | n/a | n/a | n/a | n/a |
Duquesne University
Curated by ChEMBL
| Assay Description
Inhibition of VEGFR-2 in human U251 cells assessed as reduction in VEGF induced phosphorylation preincubated for 60 mins followed by VEGF addition me... |
Bioorg Med Chem 25: 545-556 (2017)
Article DOI: 10.1016/j.bmc.2016.11.026
BindingDB Entry DOI: 10.7270/Q27W6FF7 |
More data for this
Ligand-Target Pair | |
Vascular endothelial growth factor receptor 2
(Homo sapiens (Human)) | BDBM50019930
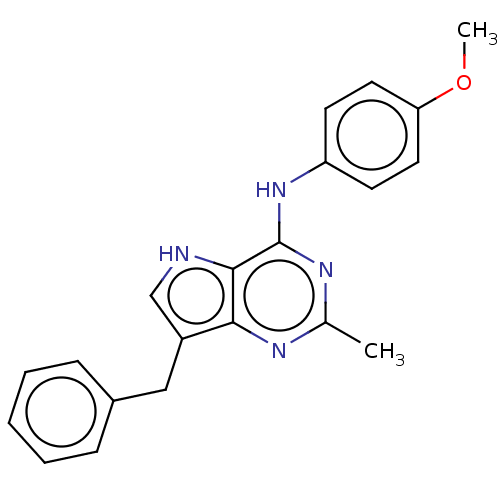
(CHEMBL3287220)Show SMILES Cl.COc1ccc(Nc2nc(C)nc3c(Cc4ccccc4)c[nH]c23)cc1 Show InChI InChI=1S/C21H20N4O.ClH/c1-14-23-19-16(12-15-6-4-3-5-7-15)13-22-20(19)21(24-14)25-17-8-10-18(26-2)11-9-17;/h3-11,13,22H,12H2,1-2H3,(H,23,24,25);1H | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 41 | n/a | n/a | n/a | n/a | n/a | n/a |
Duquesne University
Curated by ChEMBL
| Assay Description
Inhibition of VEGFR2 in human U251 cells by phosphotyrosine ELISA |
ACS Med Chem Lett 5: 480-4 (2014)
Article DOI: 10.1021/ml4004793
BindingDB Entry DOI: 10.7270/Q2H133KM |
More data for this
Ligand-Target Pair | |
Epidermal growth factor receptor
(Homo sapiens (Human)) | BDBM50232825
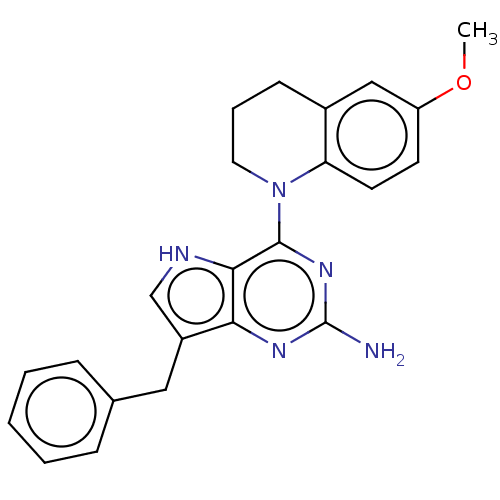
(CHEMBL4072383)Show SMILES COc1ccc2N(CCCc2c1)c1nc(N)nc2c(Cc3ccccc3)c[nH]c12 Show InChI InChI=1S/C23H23N5O/c1-29-18-9-10-19-16(13-18)8-5-11-28(19)22-21-20(26-23(24)27-22)17(14-25-21)12-15-6-3-2-4-7-15/h2-4,6-7,9-10,13-14,25H,5,8,11-12H2,1H3,(H2,24,26,27) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 47 | n/a | n/a | n/a | n/a | n/a | n/a |
Duquesne University
Curated by ChEMBL
| Assay Description
Inhibition of EGFR in human A431 cells assessed as reduction in EGF induced phosphorylation preincubated for 60 mins followed by EGF addition measure... |
Bioorg Med Chem 25: 545-556 (2017)
Article DOI: 10.1016/j.bmc.2016.11.026
BindingDB Entry DOI: 10.7270/Q27W6FF7 |
More data for this
Ligand-Target Pair | |
Vascular endothelial growth factor receptor 2
(Homo sapiens (Human)) | BDBM50019929

(CHEMBL3287221)Show SMILES Cl.CN(c1ccccc1)c1nc(C)nc2c(Cc3ccccc3)c[nH]c12 Show InChI InChI=1S/C21H20N4.ClH/c1-15-23-19-17(13-16-9-5-3-6-10-16)14-22-20(19)21(24-15)25(2)18-11-7-4-8-12-18;/h3-12,14,22H,13H2,1-2H3;1H | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 50 | n/a | n/a | n/a | n/a | n/a | n/a |
Duquesne University
Curated by ChEMBL
| Assay Description
Inhibition of VEGFR2 in human U251 cells by phosphotyrosine ELISA |
ACS Med Chem Lett 5: 480-4 (2014)
Article DOI: 10.1021/ml4004793
BindingDB Entry DOI: 10.7270/Q2H133KM |
More data for this
Ligand-Target Pair | |
Tryptophan 2,3-dioxygenase
(Homo sapiens (Human)) | BDBM50511733
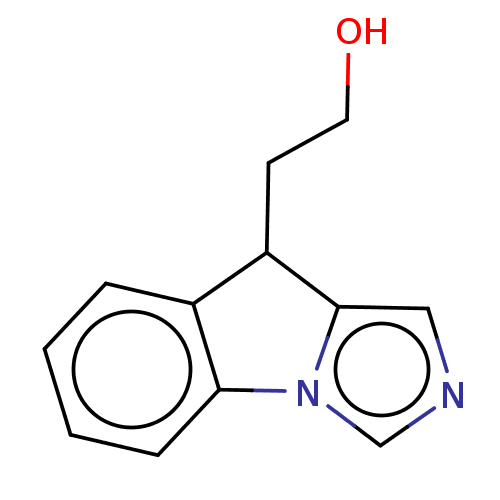
(CHEMBL4443553)Show InChI InChI=1S/C12H12N2O/c15-6-5-10-9-3-1-2-4-11(9)14-8-13-7-12(10)14/h1-4,7-8,10,15H,5-6H2 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 60 | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human TDO using L-tryptophan as substrate preincubated for 5 mins followed by substrate addition and measured after 15 mins... |
ACS Med Chem Lett 11: 541-549 (2020)
Article DOI: 10.1021/acsmedchemlett.0c00004
BindingDB Entry DOI: 10.7270/Q2RR22JS |
More data for this
Ligand-Target Pair | |
Platelet-derived growth factor receptor beta
(Homo sapiens (Human)) | BDBM4814

(CHEMBL535 | N-[2-(diethylamino)ethyl]-5-[(Z)-(5-fl...)Show SMILES CCN(CC)CCNC(=O)c1c(C)[nH]c(\C=C2/C(=O)Nc3ccc(F)cc23)c1C Show InChI InChI=1S/C22H27FN4O2/c1-5-27(6-2)10-9-24-22(29)20-13(3)19(25-14(20)4)12-17-16-11-15(23)7-8-18(16)26-21(17)28/h7-8,11-12,25H,5-6,9-10H2,1-4H3,(H,24,29)(H,26,28)/b17-12- | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
Article
PubMed
| n/a | n/a | 83 | n/a | n/a | n/a | n/a | n/a | n/a |
Duquesne University
Curated by ChEMBL
| Assay Description
Inhibition of PDGFR-beta in human SF539 cells assessed as reduction in PDGF-BB induced phosphorylation preincubated for 60 mins followed by PDGF-BB a... |
Bioorg Med Chem 25: 545-556 (2017)
Article DOI: 10.1016/j.bmc.2016.11.026
BindingDB Entry DOI: 10.7270/Q27W6FF7 |
More data for this
Ligand-Target Pair | |
Vascular endothelial growth factor receptor 2
(Homo sapiens (Human)) | BDBM50232825

(CHEMBL4072383)Show SMILES COc1ccc2N(CCCc2c1)c1nc(N)nc2c(Cc3ccccc3)c[nH]c12 Show InChI InChI=1S/C23H23N5O/c1-29-18-9-10-19-16(13-18)8-5-11-28(19)22-21-20(26-23(24)27-22)17(14-25-21)12-15-6-3-2-4-7-15/h2-4,6-7,9-10,13-14,25H,5,8,11-12H2,1H3,(H2,24,26,27) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 84 | n/a | n/a | n/a | n/a | n/a | n/a |
Duquesne University
Curated by ChEMBL
| Assay Description
Inhibition of VEGFR-2 in human U251 cells assessed as reduction in VEGF induced phosphorylation preincubated for 60 mins followed by VEGF addition me... |
Bioorg Med Chem 25: 545-556 (2017)
Article DOI: 10.1016/j.bmc.2016.11.026
BindingDB Entry DOI: 10.7270/Q27W6FF7 |
More data for this
Ligand-Target Pair | |
Platelet-derived growth factor receptor beta
(Homo sapiens (Human)) | BDBM50232826

(CHEMBL4069672)Show SMILES Cl.COc1ccc2N(CCCc2c1)c1nc(C)nc2c(Cc3ccccc3)c[nH]c12 Show InChI InChI=1S/C24H24N4O/c1-16-26-22-19(13-17-7-4-3-5-8-17)15-25-23(22)24(27-16)28-12-6-9-18-14-20(29-2)10-11-21(18)28/h3-5,7-8,10-11,14-15,25H,6,9,12-13H2,1-2H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 90 | n/a | n/a | n/a | n/a | n/a | n/a |
Duquesne University
Curated by ChEMBL
| Assay Description
Inhibition of PDGFR-beta in human SF539 cells assessed as reduction in PDGF-BB induced phosphorylation preincubated for 60 mins followed by PDGF-BB a... |
Bioorg Med Chem 25: 545-556 (2017)
Article DOI: 10.1016/j.bmc.2016.11.026
BindingDB Entry DOI: 10.7270/Q27W6FF7 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50511717
(CHEMBL4437257)Show SMILES [H][C@@]1(c2ccccc2-c2cncn12)[C@]1([H])CCc2cnccc2[C@H]1O |r| Show InChI InChI=1S/C19H17N3O/c23-19-13-7-8-20-9-12(13)5-6-16(19)18-15-4-2-1-3-14(15)17-10-21-11-22(17)18/h1-4,7-11,16,18-19,23H,5-6H2/t16-,18+,19+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | <100 | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 (unknown origin) |
ACS Med Chem Lett 11: 541-549 (2020)
Article DOI: 10.1021/acsmedchemlett.0c00004
BindingDB Entry DOI: 10.7270/Q2RR22JS |
More data for this
Ligand-Target Pair | |
Epidermal growth factor receptor
(Homo sapiens (Human)) | BDBM50232822
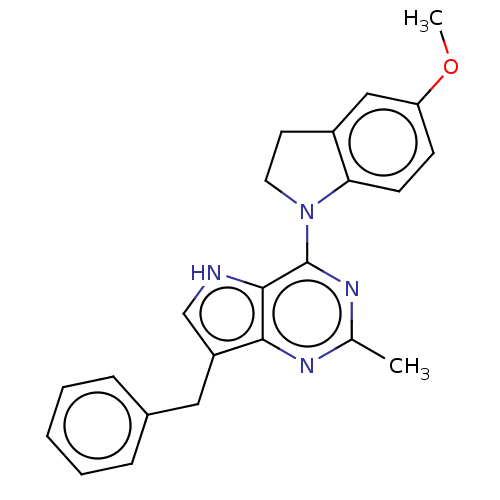
(CHEMBL4098409)Show SMILES Cl.COc1ccc2N(CCc2c1)c1nc(C)nc2c(Cc3ccccc3)c[nH]c12 Show InChI InChI=1S/C23H22N4O/c1-15-25-21-18(12-16-6-4-3-5-7-16)14-24-22(21)23(26-15)27-11-10-17-13-19(28-2)8-9-20(17)27/h3-9,13-14,24H,10-12H2,1-2H3 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 167 | n/a | n/a | n/a | n/a | n/a | n/a |
Duquesne University
Curated by ChEMBL
| Assay Description
Inhibition of EGFR in human A431 cells assessed as reduction in EGF induced phosphorylation preincubated for 60 mins followed by EGF addition measure... |
Bioorg Med Chem 25: 545-556 (2017)
Article DOI: 10.1016/j.bmc.2016.11.026
BindingDB Entry DOI: 10.7270/Q27W6FF7 |
More data for this
Ligand-Target Pair | |
Vascular endothelial growth factor receptor 2
(Homo sapiens (Human)) | BDBM50019927
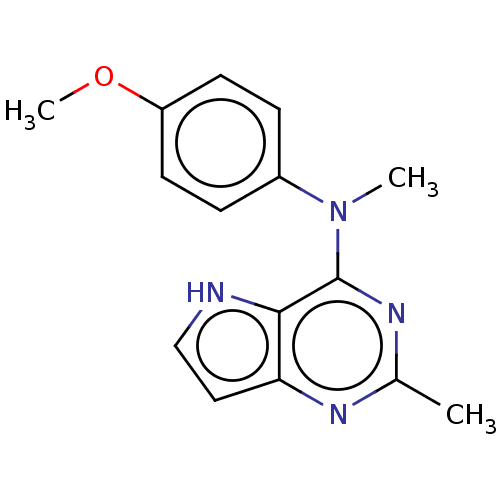
(CHEMBL3287223)Show InChI InChI=1S/C15H16N4O.ClH/c1-10-17-13-8-9-16-14(13)15(18-10)19(2)11-4-6-12(20-3)7-5-11;/h4-9,16H,1-3H3;1H | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 182 | n/a | n/a | n/a | n/a | n/a | n/a |
Duquesne University
Curated by ChEMBL
| Assay Description
Inhibition of VEGFR2 in human U251 cells by phosphotyrosine ELISA |
ACS Med Chem Lett 5: 480-4 (2014)
Article DOI: 10.1021/ml4004793
BindingDB Entry DOI: 10.7270/Q2H133KM |
More data for this
Ligand-Target Pair | |
Platelet-derived growth factor receptor beta
(Homo sapiens (Human)) | BDBM50232822

(CHEMBL4098409)Show SMILES Cl.COc1ccc2N(CCc2c1)c1nc(C)nc2c(Cc3ccccc3)c[nH]c12 Show InChI InChI=1S/C23H22N4O/c1-15-25-21-18(12-16-6-4-3-5-7-16)14-24-22(21)23(26-15)27-11-10-17-13-19(28-2)8-9-20(17)27/h3-9,13-14,24H,10-12H2,1-2H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >200 | n/a | n/a | n/a | n/a | n/a | n/a |
Duquesne University
Curated by ChEMBL
| Assay Description
Inhibition of PDGFR-beta in human SF539 cells assessed as reduction in PDGF-BB induced phosphorylation preincubated for 60 mins followed by PDGF-BB a... |
Bioorg Med Chem 25: 545-556 (2017)
Article DOI: 10.1016/j.bmc.2016.11.026
BindingDB Entry DOI: 10.7270/Q27W6FF7 |
More data for this
Ligand-Target Pair | |
Vascular endothelial growth factor receptor 2
(Homo sapiens (Human)) | BDBM50232822

(CHEMBL4098409)Show SMILES Cl.COc1ccc2N(CCc2c1)c1nc(C)nc2c(Cc3ccccc3)c[nH]c12 Show InChI InChI=1S/C23H22N4O/c1-15-25-21-18(12-16-6-4-3-5-7-16)14-24-22(21)23(26-15)27-11-10-17-13-19(28-2)8-9-20(17)27/h3-9,13-14,24H,10-12H2,1-2H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >200 | n/a | n/a | n/a | n/a | n/a | n/a |
Duquesne University
Curated by ChEMBL
| Assay Description
Inhibition of VEGFR-2 in human U251 cells assessed as reduction in VEGF induced phosphorylation preincubated for 60 mins followed by VEGF addition me... |
Bioorg Med Chem 25: 545-556 (2017)
Article DOI: 10.1016/j.bmc.2016.11.026
BindingDB Entry DOI: 10.7270/Q27W6FF7 |
More data for this
Ligand-Target Pair | |
Platelet-derived growth factor receptor beta
(Homo sapiens (Human)) | BDBM50232825

(CHEMBL4072383)Show SMILES COc1ccc2N(CCCc2c1)c1nc(N)nc2c(Cc3ccccc3)c[nH]c12 Show InChI InChI=1S/C23H23N5O/c1-29-18-9-10-19-16(13-18)8-5-11-28(19)22-21-20(26-23(24)27-22)17(14-25-21)12-15-6-3-2-4-7-15/h2-4,6-7,9-10,13-14,25H,5,8,11-12H2,1H3,(H2,24,26,27) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >200 | n/a | n/a | n/a | n/a | n/a | n/a |
Duquesne University
Curated by ChEMBL
| Assay Description
Inhibition of PDGFR-beta in human SF539 cells assessed as reduction in PDGF-BB induced phosphorylation preincubated for 60 mins followed by PDGF-BB a... |
Bioorg Med Chem 25: 545-556 (2017)
Article DOI: 10.1016/j.bmc.2016.11.026
BindingDB Entry DOI: 10.7270/Q27W6FF7 |
More data for this
Ligand-Target Pair | |
Indoleamine 2,3-dioxygenase 1
(Homo sapiens (Human)) | BDBM370555
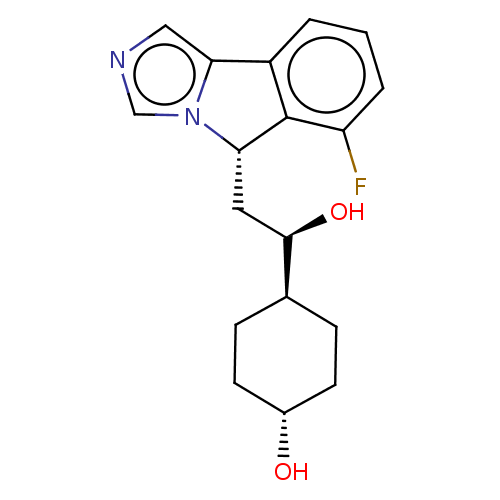
((1R,4r)-4-((R)-2-((S)-6-fluoro-5H-imidazo[5,1- a]i...)Show SMILES O[C@H](C[C@H]1c2c(cccc2F)-c2cncn12)[C@H]1CC[C@H](O)CC1 |r,wU:19.22,wD:3.2,1.0,16.19,(-.99,-3.03,;-.22,-1.7,;-.99,-.37,;-2.53,-.37,;-3.37,.92,;-4.85,.53,;-5.94,1.61,;-5.54,3.1,;-4.06,3.5,;-2.97,2.41,;-1.48,2.81,;-4.93,-1.01,;-5.9,-2.21,;-5.06,-3.5,;-3.58,-3.1,;-3.5,-1.56,;1.32,-1.7,;2.09,-3.03,;3.63,-3.03,;4.4,-1.7,;5.94,-1.7,;3.63,-.37,;2.09,-.37,)| Show InChI InChI=1S/C18H21FN2O2/c19-14-3-1-2-13-16-9-20-10-21(16)15(18(13)14)8-17(23)11-4-6-12(22)7-5-11/h1-3,9-12,15,17,22-23H,4-8H2/t11-,12-,15-,17+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| n/a | n/a | 450 | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of IDO1 (unknown origin) |
ACS Med Chem Lett 11: 541-549 (2020)
Article DOI: 10.1021/acsmedchemlett.0c00004
BindingDB Entry DOI: 10.7270/Q2RR22JS |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
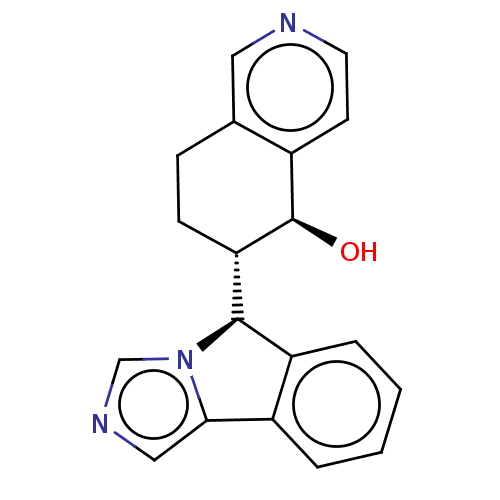
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50511719
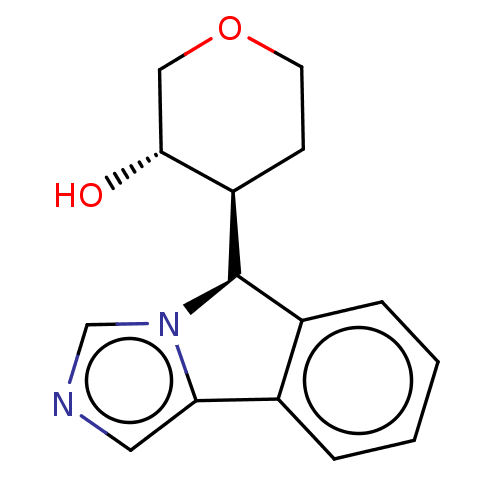
(CHEMBL4548068)Show SMILES [H][C@@]1(c2ccccc2-c2cncn12)[C@]1([H])CCOC[C@H]1O |r| Show InChI InChI=1S/C15H16N2O2/c18-14-8-19-6-5-12(14)15-11-4-2-1-3-10(11)13-7-16-9-17(13)15/h1-4,7,9,12,14-15,18H,5-6,8H2/t12-,14-,15-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | 600 | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 (unknown origin) |
ACS Med Chem Lett 11: 541-549 (2020)
Article DOI: 10.1021/acsmedchemlett.0c00004
BindingDB Entry DOI: 10.7270/Q2RR22JS |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50511715
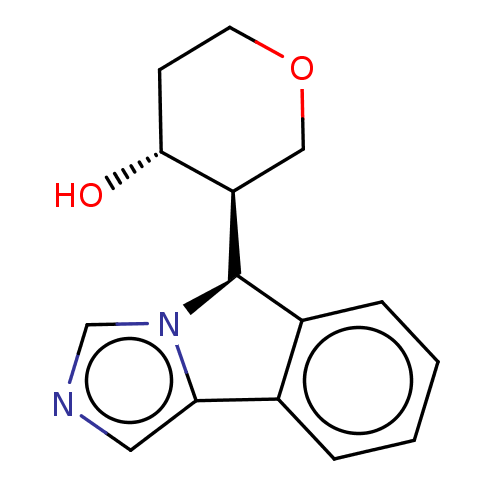
(CHEMBL4540245)Show SMILES [H][C@@]1(c2ccccc2-c2cncn12)[C@]1([H])COCC[C@H]1O |r| Show InChI InChI=1S/C15H16N2O2/c18-14-5-6-19-8-12(14)15-11-4-2-1-3-10(11)13-7-16-9-17(13)15/h1-4,7,9,12,14-15,18H,5-6,8H2/t12-,14-,15-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 650 | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 (unknown origin) |
ACS Med Chem Lett 11: 541-549 (2020)
Article DOI: 10.1021/acsmedchemlett.0c00004
BindingDB Entry DOI: 10.7270/Q2RR22JS |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50511728
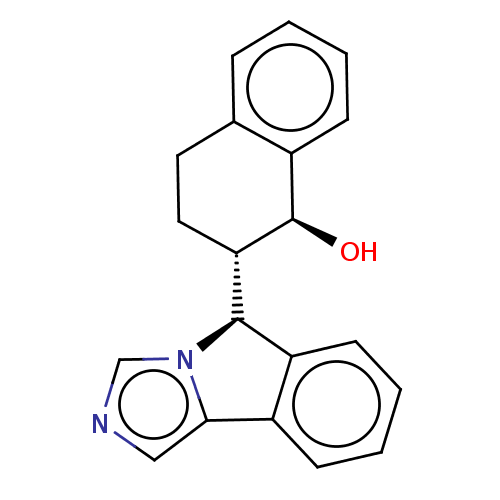
(CHEMBL4577396)Show SMILES [H][C@@]1(c2ccccc2-c2cncn12)[C@]1([H])CCc2ccccc2[C@H]1O |r| Show InChI InChI=1S/C20H18N2O/c23-20-14-6-2-1-5-13(14)9-10-17(20)19-16-8-4-3-7-15(16)18-11-21-12-22(18)19/h1-8,11-12,17,19-20,23H,9-10H2/t17-,19+,20+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 840 | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 (unknown origin) |
ACS Med Chem Lett 11: 541-549 (2020)
Article DOI: 10.1021/acsmedchemlett.0c00004
BindingDB Entry DOI: 10.7270/Q2RR22JS |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50511729
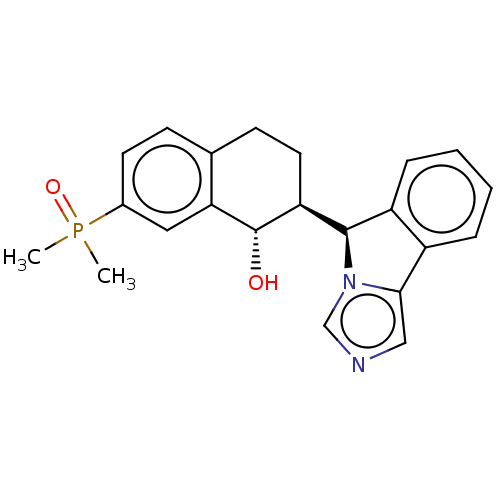
(CHEMBL4589215)Show SMILES [H][C@@]1(c2ccccc2-c2cncn12)[C@]1([H])CCc2ccc(cc2[C@H]1O)P(C)(C)=O |r| Show InChI InChI=1S/C22H23N2O2P/c1-27(2,26)15-9-7-14-8-10-18(22(25)19(14)11-15)21-17-6-4-3-5-16(17)20-12-23-13-24(20)21/h3-7,9,11-13,18,21-22,25H,8,10H2,1-2H3/t18-,21+,22-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 (unknown origin) |
ACS Med Chem Lett 11: 541-549 (2020)
Article DOI: 10.1021/acsmedchemlett.0c00004
BindingDB Entry DOI: 10.7270/Q2RR22JS |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50511727
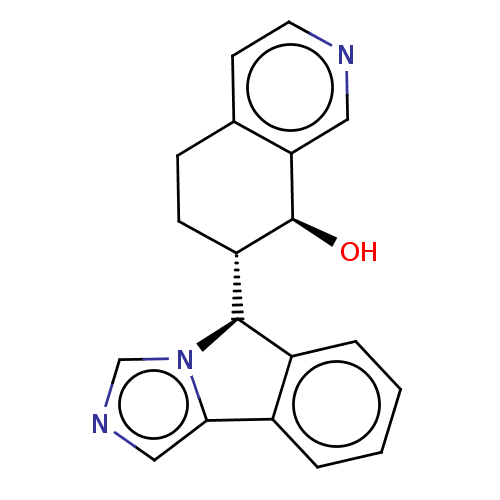
(CHEMBL4447185)Show SMILES [H][C@@]1(c2ccccc2-c2cncn12)[C@]1([H])CCc2ccncc2[C@H]1O |r| Show InChI InChI=1S/C19H17N3O/c23-19-15(6-5-12-7-8-20-9-16(12)19)18-14-4-2-1-3-13(14)17-10-21-11-22(17)18/h1-4,7-11,15,18-19,23H,5-6H2/t15-,18+,19-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 2.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 (unknown origin) |
ACS Med Chem Lett 11: 541-549 (2020)
Article DOI: 10.1021/acsmedchemlett.0c00004
BindingDB Entry DOI: 10.7270/Q2RR22JS |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
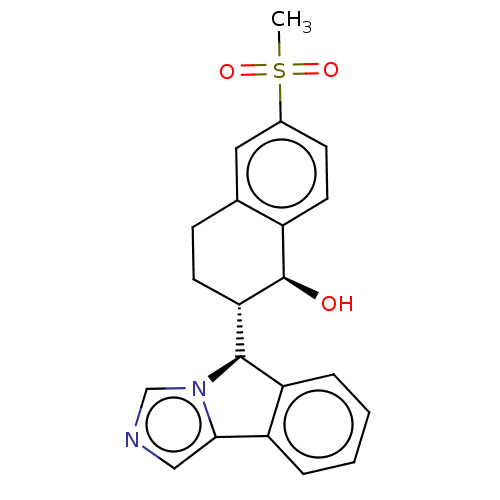
(Homo sapiens (Human)) | BDBM50511716
(CHEMBL4539108)Show SMILES [H][C@@]1(c2ccccc2-c2cncn12)[C@]1([H])CCc2cc(ccc2[C@H]1O)S(C)(=O)=O |r| Show InChI InChI=1S/C21H20N2O3S/c1-27(25,26)14-7-9-15-13(10-14)6-8-18(21(15)24)20-17-5-3-2-4-16(17)19-11-22-12-23(19)20/h2-5,7,9-12,18,20-21,24H,6,8H2,1H3/t18-,20+,21+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 2.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 (unknown origin) |
ACS Med Chem Lett 11: 541-549 (2020)
Article DOI: 10.1021/acsmedchemlett.0c00004
BindingDB Entry DOI: 10.7270/Q2RR22JS |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50511710
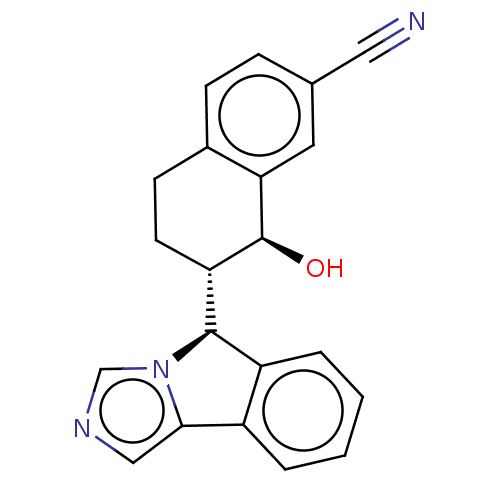
(CHEMBL4541824)Show SMILES [H][C@@]1(c2ccccc2-c2cncn12)[C@]1([H])CCc2ccc(cc2[C@H]1O)C#N |r| Show InChI InChI=1S/C21H17N3O/c22-10-13-5-6-14-7-8-17(21(25)18(14)9-13)20-16-4-2-1-3-15(16)19-11-23-12-24(19)20/h1-6,9,11-12,17,20-21,25H,7-8H2/t17-,20+,21-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 3.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 (unknown origin) |
ACS Med Chem Lett 11: 541-549 (2020)
Article DOI: 10.1021/acsmedchemlett.0c00004
BindingDB Entry DOI: 10.7270/Q2RR22JS |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50511726
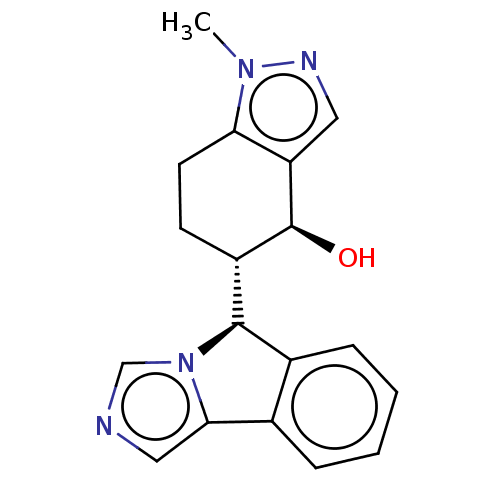
(CHEMBL4440730)Show SMILES [H][C@@]1(c2ccccc2-c2cncn12)[C@]1([H])CCc2c(cnn2C)[C@H]1O |r| Show InChI InChI=1S/C18H18N4O/c1-21-15-7-6-13(18(23)14(15)8-20-21)17-12-5-3-2-4-11(12)16-9-19-10-22(16)17/h2-5,8-10,13,17-18,23H,6-7H2,1H3/t13-,17+,18-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 3.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 (unknown origin) |
ACS Med Chem Lett 11: 541-549 (2020)
Article DOI: 10.1021/acsmedchemlett.0c00004
BindingDB Entry DOI: 10.7270/Q2RR22JS |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
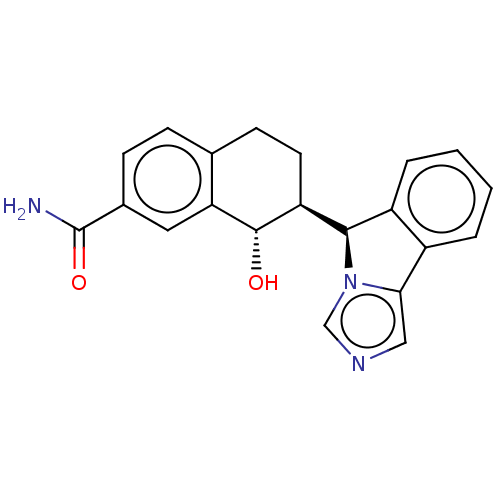
(Homo sapiens (Human)) | BDBM50511731
(CHEMBL4544695)Show SMILES [H][C@@]1(c2ccccc2-c2cncn12)[C@]1([H])CCc2ccc(cc2[C@H]1O)C(N)=O |r| Show InChI InChI=1S/C21H19N3O2/c22-21(26)13-6-5-12-7-8-16(20(25)17(12)9-13)19-15-4-2-1-3-14(15)18-10-23-11-24(18)19/h1-6,9-11,16,19-20,25H,7-8H2,(H2,22,26)/t16-,19+,20-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 3.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 (unknown origin) |
ACS Med Chem Lett 11: 541-549 (2020)
Article DOI: 10.1021/acsmedchemlett.0c00004
BindingDB Entry DOI: 10.7270/Q2RR22JS |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
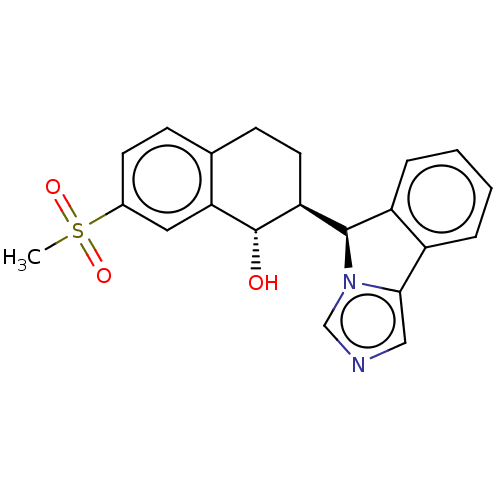
(Homo sapiens (Human)) | BDBM50511714
(CHEMBL4520696)Show SMILES [H][C@@]1(c2ccccc2-c2cncn12)[C@]1([H])CCc2ccc(cc2[C@H]1O)S(C)(=O)=O |r| Show InChI InChI=1S/C21H20N2O3S/c1-27(25,26)14-8-6-13-7-9-17(21(24)18(13)10-14)20-16-5-3-2-4-15(16)19-11-22-12-23(19)20/h2-6,8,10-12,17,20-21,24H,7,9H2,1H3/t17-,20+,21-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 6.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 (unknown origin) |
ACS Med Chem Lett 11: 541-549 (2020)
Article DOI: 10.1021/acsmedchemlett.0c00004
BindingDB Entry DOI: 10.7270/Q2RR22JS |
More data for this
Ligand-Target Pair | |
Tryptophan 2,3-dioxygenase
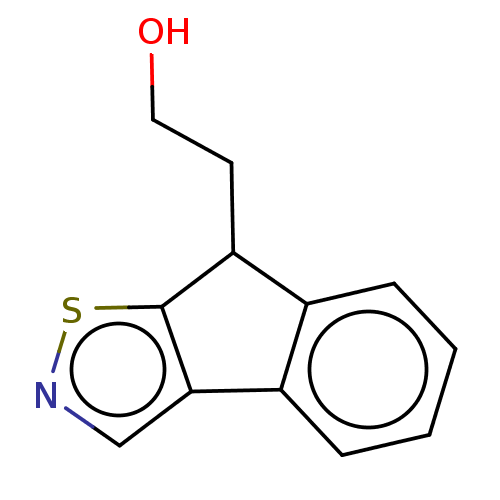
(Homo sapiens (Human)) | BDBM50511736
(CHEMBL4572482)Show InChI InChI=1S/C12H11NOS/c14-6-5-10-8-3-1-2-4-9(8)11-7-13-15-12(10)11/h1-4,7,10,14H,5-6H2 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 7.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human TDO using L-tryptophan as substrate preincubated for 5 mins followed by substrate addition and measured after 15 mins... |
ACS Med Chem Lett 11: 541-549 (2020)
Article DOI: 10.1021/acsmedchemlett.0c00004
BindingDB Entry DOI: 10.7270/Q2RR22JS |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
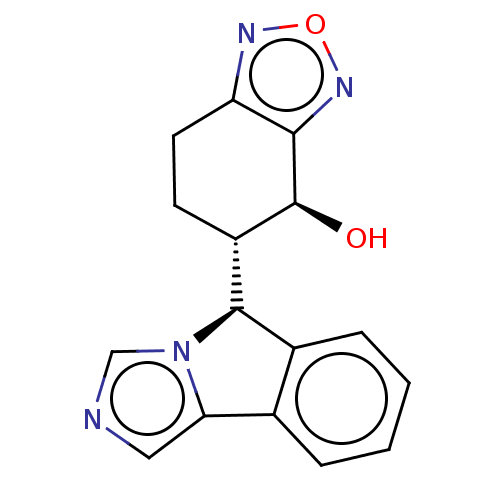
(Homo sapiens (Human)) | BDBM50511730
(CHEMBL4590789)Show SMILES [H][C@@]1(c2ccccc2-c2cncn12)[C@]1([H])CCc2nonc2[C@H]1O |r| Show InChI InChI=1S/C16H14N4O2/c21-16-11(5-6-12-14(16)19-22-18-12)15-10-4-2-1-3-9(10)13-7-17-8-20(13)15/h1-4,7-8,11,15-16,21H,5-6H2/t11-,15+,16-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 8.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 (unknown origin) |
ACS Med Chem Lett 11: 541-549 (2020)
Article DOI: 10.1021/acsmedchemlett.0c00004
BindingDB Entry DOI: 10.7270/Q2RR22JS |
More data for this
Ligand-Target Pair | |
Tryptophan 2,3-dioxygenase
(Homo sapiens (Human)) | BDBM50285416
(CHEMBL4161733)Show SMILES [H][C@]1(CC[C@@H](CC1)c1ccnc2ccc(F)cc12)[C@@H](C)C(=O)Nc1ccc(Cl)cc1 |r,wU:18.21,1.0,wD:4.7,(66.16,-9.22,;66.17,-7.68,;66.16,-6.15,;67.5,-5.38,;68.84,-6.15,;68.83,-7.69,;67.5,-8.46,;70.18,-5.39,;71.5,-6.16,;72.84,-5.4,;72.84,-3.85,;71.5,-3.08,;71.5,-1.55,;70.17,-.79,;68.84,-1.56,;67.5,-.8,;68.85,-3.09,;70.18,-3.85,;64.83,-8.46,;64.84,-10,;63.5,-7.69,;63.49,-6.14,;62.16,-8.46,;60.83,-7.69,;59.49,-8.47,;58.15,-7.69,;58.16,-6.14,;56.82,-5.38,;59.49,-5.38,;60.83,-6.14,)| Show InChI InChI=1S/C24H24ClFN2O/c1-15(24(29)28-20-9-6-18(25)7-10-20)16-2-4-17(5-3-16)21-12-13-27-23-11-8-19(26)14-22(21)23/h6-17H,2-5H2,1H3,(H,28,29)/t15-,16-,17+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of TDO (unknown origin) |
ACS Med Chem Lett 11: 541-549 (2020)
Article DOI: 10.1021/acsmedchemlett.0c00004
BindingDB Entry DOI: 10.7270/Q2RR22JS |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data