 Found 155 hits with Last Name = 'janovitz' and Initial = 'eb'
Found 155 hits with Last Name = 'janovitz' and Initial = 'eb' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Melanin-concentrating hormone receptor 1
(RAT) | BDBM50194196
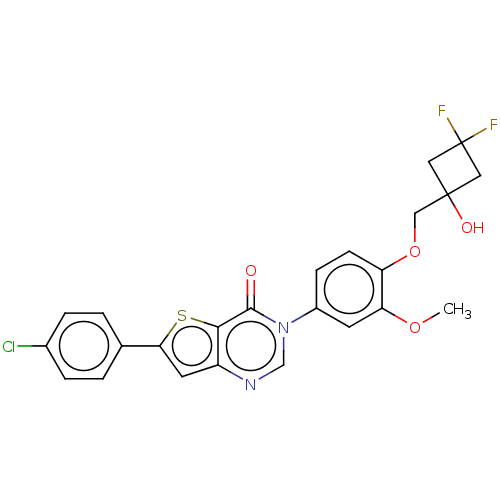
(CHEMBL2147472)Show SMILES COc1cc(ccc1OCC1(O)CC(F)(F)C1)-n1cnc2cc(sc2c1=O)-c1ccc(Cl)cc1 Show InChI InChI=1S/C24H19ClF2N2O4S/c1-32-19-8-16(6-7-18(19)33-12-23(31)10-24(26,27)11-23)29-13-28-17-9-20(34-21(17)22(29)30)14-2-4-15(25)5-3-14/h2-9,13,31H,10-12H2,1H3 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 12 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research& Development
Curated by ChEMBL
| Assay Description
Binding affinity to rat MCHR1 |
J Med Chem 59: 8848-8858 (2016)
Article DOI: 10.1021/acs.jmedchem.6b00676
BindingDB Entry DOI: 10.7270/Q2W95C41 |
More data for this
Ligand-Target Pair | |
Melanin-concentrating hormone receptor 1
(Homo sapiens (Human)) | BDBM50194195

(CHEMBL3945242)Show SMILES COc1cc(ccc1OCC1(CC(F)(F)C1)OP(O)(O)=O)-n1cnc2cc(sc2c1=O)-c1ccc(Cl)cc1 Show InChI InChI=1S/C24H20ClF2N2O7PS/c1-34-19-8-16(6-7-18(19)35-12-23(36-37(31,32)33)10-24(26,27)11-23)29-13-28-17-9-20(38-21(17)22(29)30)14-2-4-15(25)5-3-14/h2-9,13H,10-12H2,1H3,(H2,31,32,33) | Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.92E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research& Development
Curated by ChEMBL
| Assay Description
Binding affinity to MCHR1 (unknown origin) |
J Med Chem 59: 8848-8858 (2016)
Article DOI: 10.1021/acs.jmedchem.6b00676
BindingDB Entry DOI: 10.7270/Q2W95C41 |
More data for this
Ligand-Target Pair | |
Hexokinase-4
(Homo sapiens (Human)) | BDBM50585812
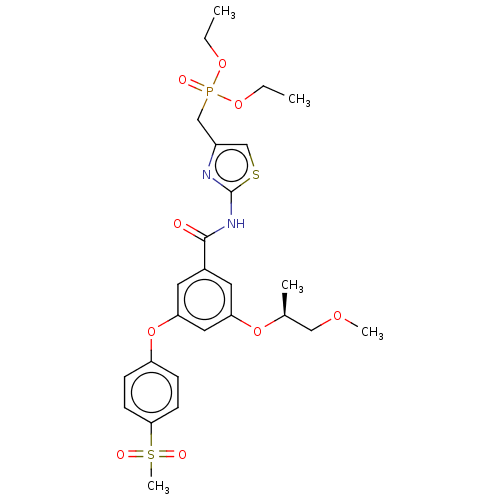
(CHEMBL5091943)Show SMILES CCOP(=O)(Cc1csc(NC(=O)c2cc(O[C@@H](C)COC)cc(Oc3ccc(cc3)S(C)(=O)=O)c2)n1)OCC |r| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| n/a | n/a | 122 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Displacement of fluorescent labeled derivative from recombinant human hepatic glucokinase incubated for 30 mins in presence of 12 mM glucose by fluor... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c02110
BindingDB Entry DOI: 10.7270/Q2057KV7 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Hexokinase-4
(Homo sapiens (Human)) | BDBM50585813
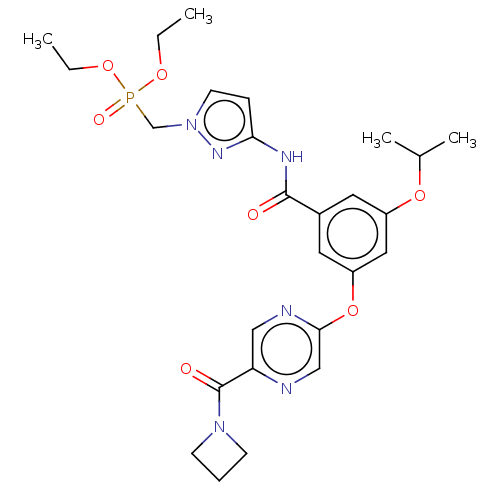
(CHEMBL5072532)Show SMILES CCOP(=O)(Cn1ccc(NC(=O)c2cc(OC(C)C)cc(Oc3cnc(cn3)C(=O)N3CCC3)c2)n1)OCC | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| n/a | n/a | 123 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Displacement of fluorescent labeled derivative from recombinant human hepatic glucokinase incubated for 30 mins in presence of 12 mM glucose by fluor... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c02110
BindingDB Entry DOI: 10.7270/Q2057KV7 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Cytochrome P450 2C8
(Homo sapiens (Human)) | BDBM50243975
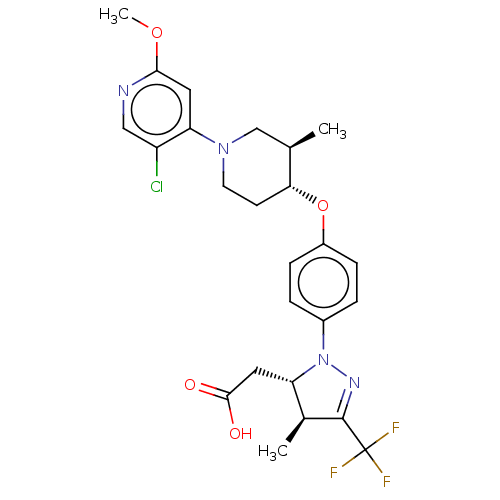
(CHEMBL4080226)Show SMILES COc1cc(N2CC[C@@H](Oc3ccc(cc3)N3N=C([C@@H](C)[C@@H]3CC(O)=O)C(F)(F)F)[C@H](C)C2)c(Cl)cn1 |r,c:18| Show InChI InChI=1S/C25H28ClF3N4O4/c1-14-13-32(20-10-22(36-3)30-12-18(20)26)9-8-21(14)37-17-6-4-16(5-7-17)33-19(11-23(34)35)15(2)24(31-33)25(27,28)29/h4-7,10,12,14-15,19,21H,8-9,11,13H2,1-3H3,(H,34,35)/t14-,15+,19+,21-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| | n/a | n/a | 1.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50243975

(CHEMBL4080226)Show SMILES COc1cc(N2CC[C@@H](Oc3ccc(cc3)N3N=C([C@@H](C)[C@@H]3CC(O)=O)C(F)(F)F)[C@H](C)C2)c(Cl)cn1 |r,c:18| Show InChI InChI=1S/C25H28ClF3N4O4/c1-14-13-32(20-10-22(36-3)30-12-18(20)26)9-8-21(14)37-17-6-4-16(5-7-17)33-19(11-23(34)35)15(2)24(31-33)25(27,28)29/h4-7,10,12,14-15,19,21H,8-9,11,13H2,1-3H3,(H,34,35)/t14-,15+,19+,21-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| | n/a | n/a | 3.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C8
(Homo sapiens (Human)) | BDBM50585813

(CHEMBL5072532)Show SMILES CCOP(=O)(Cn1ccc(NC(=O)c2cc(OC(C)C)cc(Oc3cnc(cn3)C(=O)N3CCC3)c2)n1)OCC | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | 7.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of CYP2C8 (unknown origin) |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c02110
BindingDB Entry DOI: 10.7270/Q2057KV7 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2B6
(Homo sapiens (Human)) | BDBM50243975

(CHEMBL4080226)Show SMILES COc1cc(N2CC[C@@H](Oc3ccc(cc3)N3N=C([C@@H](C)[C@@H]3CC(O)=O)C(F)(F)F)[C@H](C)C2)c(Cl)cn1 |r,c:18| Show InChI InChI=1S/C25H28ClF3N4O4/c1-14-13-32(20-10-22(36-3)30-12-18(20)26)9-8-21(14)37-17-6-4-16(5-7-17)33-19(11-23(34)35)15(2)24(31-33)25(27,28)29/h4-7,10,12,14-15,19,21H,8-9,11,13H2,1-3H3,(H,34,35)/t14-,15+,19+,21-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| | n/a | n/a | 8.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C8
(Homo sapiens (Human)) | BDBM50614666
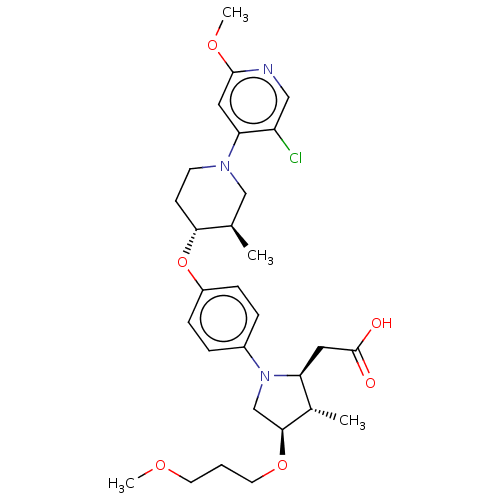
(CHEMBL5285988)Show SMILES COCCCO[C@H]1CN([C@@H](CC(O)=O)[C@@H]1C)c1ccc(O[C@@H]2CCN(C[C@H]2C)c2cc(OC)ncc2Cl)cc1 |r| | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| UniChem
| | n/a | n/a | 8.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C9
(Homo sapiens (Human)) | BDBM50243975

(CHEMBL4080226)Show SMILES COc1cc(N2CC[C@@H](Oc3ccc(cc3)N3N=C([C@@H](C)[C@@H]3CC(O)=O)C(F)(F)F)[C@H](C)C2)c(Cl)cn1 |r,c:18| Show InChI InChI=1S/C25H28ClF3N4O4/c1-14-13-32(20-10-22(36-3)30-12-18(20)26)9-8-21(14)37-17-6-4-16(5-7-17)33-19(11-23(34)35)15(2)24(31-33)25(27,28)29/h4-7,10,12,14-15,19,21H,8-9,11,13H2,1-3H3,(H,34,35)/t14-,15+,19+,21-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| | n/a | n/a | 1.40E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Cytochrome P450 2B6
(Homo sapiens (Human)) | BDBM50614666

(CHEMBL5285988)Show SMILES COCCCO[C@H]1CN([C@@H](CC(O)=O)[C@@H]1C)c1ccc(O[C@@H]2CCN(C[C@H]2C)c2cc(OC)ncc2Cl)cc1 |r| | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| UniChem
| | n/a | n/a | 2.90E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C19
(Homo sapiens (Human)) | BDBM50585813

(CHEMBL5072532)Show SMILES CCOP(=O)(Cn1ccc(NC(=O)c2cc(OC(C)C)cc(Oc3cnc(cn3)C(=O)N3CCC3)c2)n1)OCC | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | 3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of CYP2C19 (unknown origin) |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c02110
BindingDB Entry DOI: 10.7270/Q2057KV7 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50585813

(CHEMBL5072532)Show SMILES CCOP(=O)(Cn1ccc(NC(=O)c2cc(OC(C)C)cc(Oc3cnc(cn3)C(=O)N3CCC3)c2)n1)OCC | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | 3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of CYP3A4 (unknown origin) |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c02110
BindingDB Entry DOI: 10.7270/Q2057KV7 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM50585813

(CHEMBL5072532)Show SMILES CCOP(=O)(Cn1ccc(NC(=O)c2cc(OC(C)C)cc(Oc3cnc(cn3)C(=O)N3CCC3)c2)n1)OCC | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | 3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of CYP1A2 (unknown origin) |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c02110
BindingDB Entry DOI: 10.7270/Q2057KV7 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2B6
(Homo sapiens (Human)) | BDBM50585813

(CHEMBL5072532)Show SMILES CCOP(=O)(Cn1ccc(NC(=O)c2cc(OC(C)C)cc(Oc3cnc(cn3)C(=O)N3CCC3)c2)n1)OCC | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | 3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of CYP2B6 (unknown origin) |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c02110
BindingDB Entry DOI: 10.7270/Q2057KV7 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM50585813

(CHEMBL5072532)Show SMILES CCOP(=O)(Cn1ccc(NC(=O)c2cc(OC(C)C)cc(Oc3cnc(cn3)C(=O)N3CCC3)c2)n1)OCC | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | 3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of CYP2D6 (unknown origin) |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c02110
BindingDB Entry DOI: 10.7270/Q2057KV7 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C9
(Homo sapiens (Human)) | BDBM50585813

(CHEMBL5072532)Show SMILES CCOP(=O)(Cn1ccc(NC(=O)c2cc(OC(C)C)cc(Oc3cnc(cn3)C(=O)N3CCC3)c2)n1)OCC | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | 3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of CYP2C9 (unknown origin) |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c02110
BindingDB Entry DOI: 10.7270/Q2057KV7 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50585829
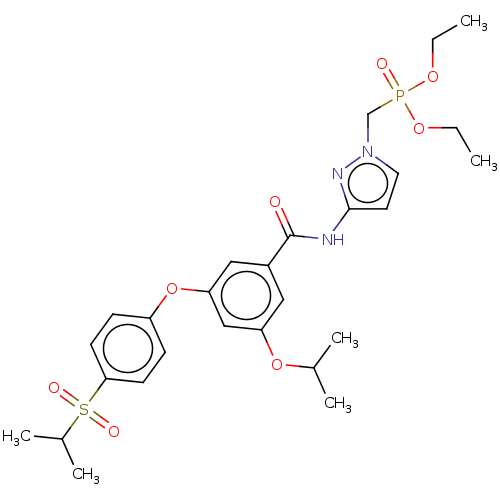
(CHEMBL5092846)Show SMILES CCOP(=O)(Cn1ccc(NC(=O)c2cc(OC(C)C)cc(Oc3ccc(cc3)S(=O)(=O)C(C)C)c2)n1)OCC | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of hERG by patch clamp method |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c02110
BindingDB Entry DOI: 10.7270/Q2057KV7 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50585830
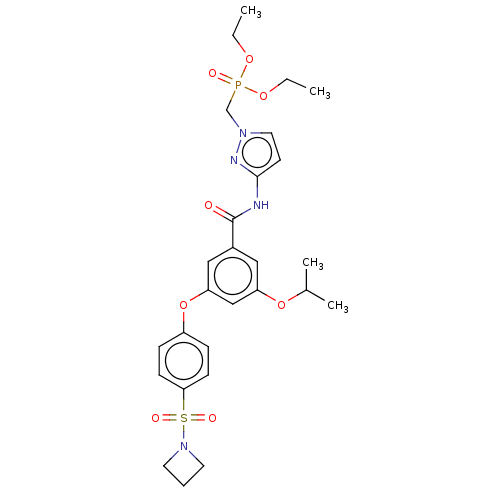
(CHEMBL5072442)Show SMILES CCOP(=O)(Cn1ccc(NC(=O)c2cc(OC(C)C)cc(Oc3ccc(cc3)S(=O)(=O)N3CCC3)c2)n1)OCC | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 3.30E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of hERG by patch clamp method |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c02110
BindingDB Entry DOI: 10.7270/Q2057KV7 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C9
(Homo sapiens (Human)) | BDBM50614666

(CHEMBL5285988)Show SMILES COCCCO[C@H]1CN([C@@H](CC(O)=O)[C@@H]1C)c1ccc(O[C@@H]2CCN(C[C@H]2C)c2cc(OC)ncc2Cl)cc1 |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| UniChem
| | n/a | n/a | 3.40E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C19
(Homo sapiens (Human)) | BDBM50243975

(CHEMBL4080226)Show SMILES COc1cc(N2CC[C@@H](Oc3ccc(cc3)N3N=C([C@@H](C)[C@@H]3CC(O)=O)C(F)(F)F)[C@H](C)C2)c(Cl)cn1 |r,c:18| Show InChI InChI=1S/C25H28ClF3N4O4/c1-14-13-32(20-10-22(36-3)30-12-18(20)26)9-8-21(14)37-17-6-4-16(5-7-17)33-19(11-23(34)35)15(2)24(31-33)25(27,28)29/h4-7,10,12,14-15,19,21H,8-9,11,13H2,1-3H3,(H,34,35)/t14-,15+,19+,21-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| | n/a | n/a | 3.80E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM50614666

(CHEMBL5285988)Show SMILES COCCCO[C@H]1CN([C@@H](CC(O)=O)[C@@H]1C)c1ccc(O[C@@H]2CCN(C[C@H]2C)c2cc(OC)ncc2Cl)cc1 |r| | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| UniChem
| | n/a | n/a | 4.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C19
(Homo sapiens (Human)) | BDBM50614666

(CHEMBL5285988)Show SMILES COCCCO[C@H]1CN([C@@H](CC(O)=O)[C@@H]1C)c1ccc(O[C@@H]2CCN(C[C@H]2C)c2cc(OC)ncc2Cl)cc1 |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| UniChem
| | n/a | n/a | 4.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50614666

(CHEMBL5285988)Show SMILES COCCCO[C@H]1CN([C@@H](CC(O)=O)[C@@H]1C)c1ccc(O[C@@H]2CCN(C[C@H]2C)c2cc(OC)ncc2Cl)cc1 |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| UniChem
| | n/a | n/a | 4.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM50243975

(CHEMBL4080226)Show SMILES COc1cc(N2CC[C@@H](Oc3ccc(cc3)N3N=C([C@@H](C)[C@@H]3CC(O)=O)C(F)(F)F)[C@H](C)C2)c(Cl)cn1 |r,c:18| Show InChI InChI=1S/C25H28ClF3N4O4/c1-14-13-32(20-10-22(36-3)30-12-18(20)26)9-8-21(14)37-17-6-4-16(5-7-17)33-19(11-23(34)35)15(2)24(31-33)25(27,28)29/h4-7,10,12,14-15,19,21H,8-9,11,13H2,1-3H3,(H,34,35)/t14-,15+,19+,21-/m1/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| | n/a | n/a | 4.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50585823
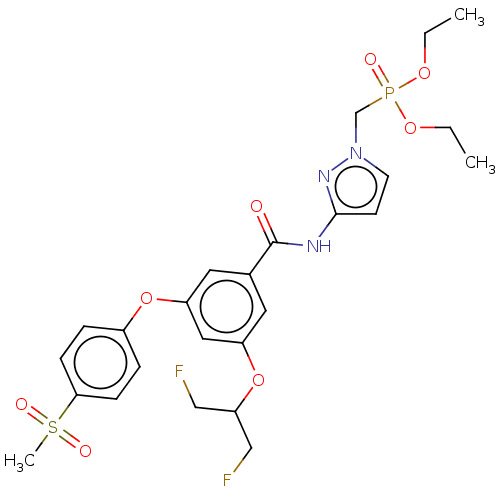
(CHEMBL5081517)Show SMILES CCOP(=O)(Cn1ccc(NC(=O)c2cc(OC(CF)CF)cc(Oc3ccc(cc3)S(C)(=O)=O)c2)n1)OCC | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 4.40E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of hERG by patch clamp method |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c02110
BindingDB Entry DOI: 10.7270/Q2057KV7 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50585812

(CHEMBL5091943)Show SMILES CCOP(=O)(Cc1csc(NC(=O)c2cc(O[C@@H](C)COC)cc(Oc3ccc(cc3)S(C)(=O)=O)c2)n1)OCC |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | 5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of hERG by patch clamp method |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c02110
BindingDB Entry DOI: 10.7270/Q2057KV7 |
More data for this
Ligand-Target Pair | |
ATP-dependent translocase ABCB1
(Homo sapiens (Human)) | BDBM50585813

(CHEMBL5072532)Show SMILES CCOP(=O)(Cn1ccc(NC(=O)c2cc(OC(C)C)cc(Oc3cnc(cn3)C(=O)N3CCC3)c2)n1)OCC | PDB
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | 5.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of P-gp transporter (unknown origin) |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c02110
BindingDB Entry DOI: 10.7270/Q2057KV7 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50585827
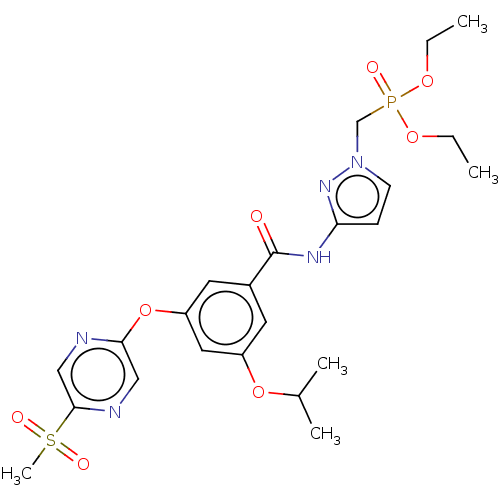
(CHEMBL5088522)Show SMILES CCOP(=O)(Cn1ccc(NC(=O)c2cc(OC(C)C)cc(Oc3cnc(cn3)S(C)(=O)=O)c2)n1)OCC | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >8.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of hERG by patch clamp method |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c02110
BindingDB Entry DOI: 10.7270/Q2057KV7 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50585826

(CHEMBL5085491)Show SMILES CCOP(=O)(Cn1ccc(NC(=O)c2cc(OC(C)C)cc(Oc3cnc(nc3)S(C)(=O)=O)c2)n1)OCC | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >8.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of hERG by patch clamp method |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c02110
BindingDB Entry DOI: 10.7270/Q2057KV7 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50585825
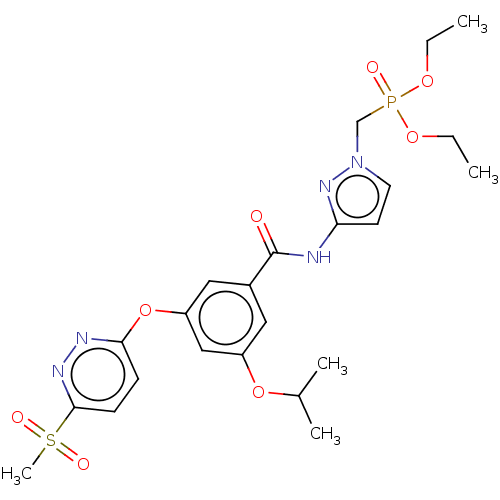
(CHEMBL5086686)Show SMILES CCOP(=O)(Cn1ccc(NC(=O)c2cc(OC(C)C)cc(Oc3ccc(nn3)S(C)(=O)=O)c2)n1)OCC | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >8.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of hERG by patch clamp method |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c02110
BindingDB Entry DOI: 10.7270/Q2057KV7 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50585824
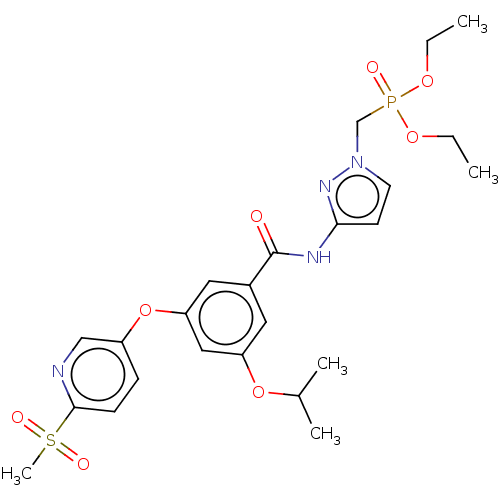
(CHEMBL5084870)Show SMILES CCOP(=O)(Cn1ccc(NC(=O)c2cc(OC(C)C)cc(Oc3ccc(nc3)S(C)(=O)=O)c2)n1)OCC | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >8.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of hERG by patch clamp method |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c02110
BindingDB Entry DOI: 10.7270/Q2057KV7 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50585822
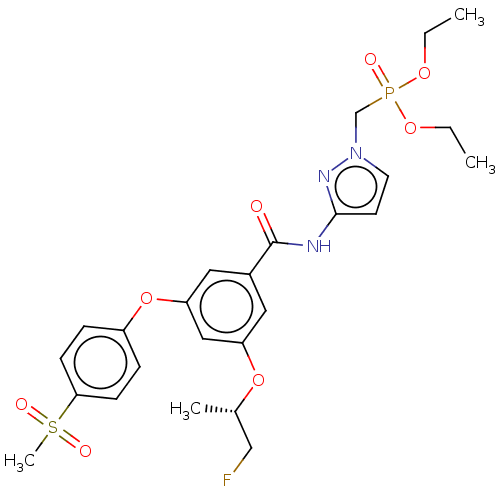
(CHEMBL5083369)Show SMILES CCOP(=O)(Cn1ccc(NC(=O)c2cc(O[C@@H](C)CF)cc(Oc3ccc(cc3)S(C)(=O)=O)c2)n1)OCC |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >8.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of hERG by patch clamp method |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c02110
BindingDB Entry DOI: 10.7270/Q2057KV7 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50585821
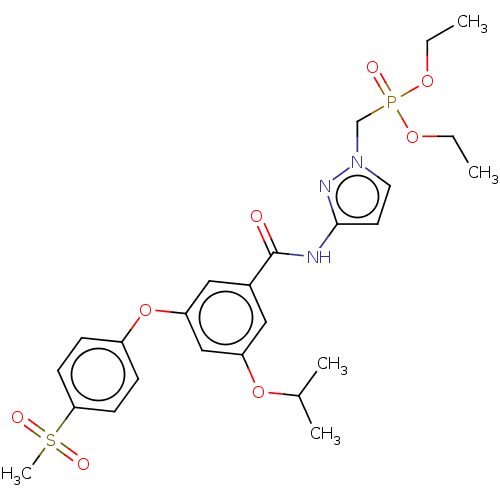
(CHEMBL5093978)Show SMILES CCOP(=O)(Cn1ccc(NC(=O)c2cc(OC(C)C)cc(Oc3ccc(cc3)S(C)(=O)=O)c2)n1)OCC | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >8.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of hERG by patch clamp method |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c02110
BindingDB Entry DOI: 10.7270/Q2057KV7 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50585820
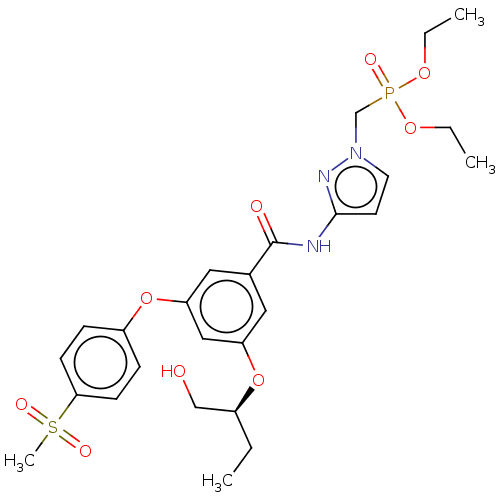
(CHEMBL5093208)Show SMILES CCOP(=O)(Cn1ccc(NC(=O)c2cc(O[C@@H](CC)CO)cc(Oc3ccc(cc3)S(C)(=O)=O)c2)n1)OCC |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >8.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of hERG by patch clamp method |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c02110
BindingDB Entry DOI: 10.7270/Q2057KV7 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50585819
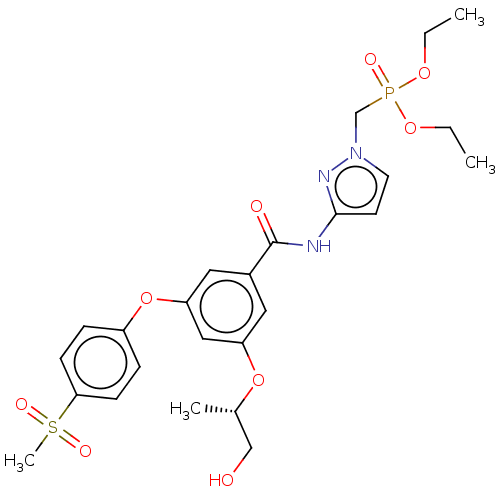
(CHEMBL5088988)Show SMILES CCOP(=O)(Cn1ccc(NC(=O)c2cc(O[C@@H](C)CO)cc(Oc3ccc(cc3)S(C)(=O)=O)c2)n1)OCC |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >8.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of hERG by patch clamp method |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c02110
BindingDB Entry DOI: 10.7270/Q2057KV7 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50585818
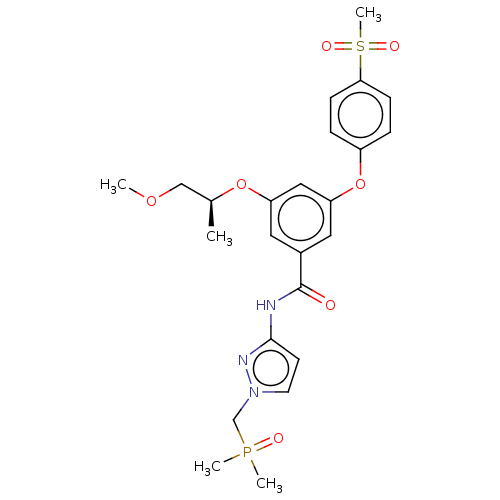
(CHEMBL5080889)Show SMILES COC[C@H](C)Oc1cc(Oc2ccc(cc2)S(C)(=O)=O)cc(c1)C(=O)Nc1ccn(CP(C)(C)=O)n1 |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >8.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of hERG by patch clamp method |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c02110
BindingDB Entry DOI: 10.7270/Q2057KV7 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50585817

(CHEMBL5075882)Show SMILES CCO[P@](C)(=O)Cn1ccc(NC(=O)c2cc(O[C@@H](C)COC)cc(Oc3ccc(cc3)S(C)(=O)=O)c2)n1 |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >8.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of hERG by patch clamp method |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c02110
BindingDB Entry DOI: 10.7270/Q2057KV7 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50585816

(CHEMBL5086985)Show SMILES CCOP(=O)(Cc1cnc(NC(=O)c2cc(O[C@@H](C)COC)cc(Oc3ccc(cc3)S(C)(=O)=O)c2)cn1)OCC |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >8.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of hERG by patch clamp method |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c02110
BindingDB Entry DOI: 10.7270/Q2057KV7 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50585814
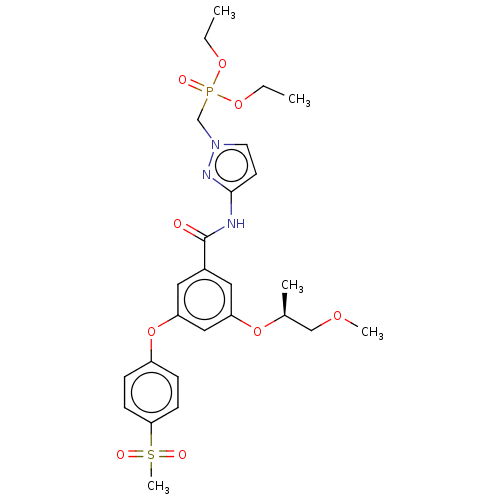
(CHEMBL5071861)Show SMILES CCOP(=O)(Cn1ccc(NC(=O)c2cc(O[C@@H](C)COC)cc(Oc3ccc(cc3)S(C)(=O)=O)c2)n1)OCC |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >8.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of hERG by patch clamp method |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c02110
BindingDB Entry DOI: 10.7270/Q2057KV7 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50585831
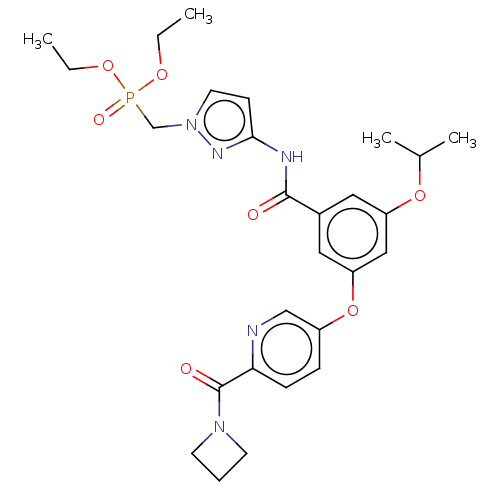
(CHEMBL5076377)Show SMILES CCOP(=O)(Cn1ccc(NC(=O)c2cc(OC(C)C)cc(Oc3ccc(nc3)C(=O)N3CCC3)c2)n1)OCC | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >8.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of hERG by patch clamp method |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c02110
BindingDB Entry DOI: 10.7270/Q2057KV7 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50585832
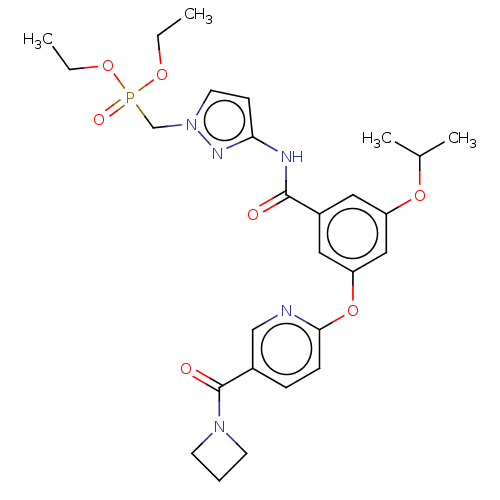
(CHEMBL5087489)Show SMILES CCOP(=O)(Cn1ccc(NC(=O)c2cc(OC(C)C)cc(Oc3ccc(cn3)C(=O)N3CCC3)c2)n1)OCC | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >8.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of hERG by patch clamp method |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c02110
BindingDB Entry DOI: 10.7270/Q2057KV7 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50585837

(CHEMBL5080166)Show SMILES CCOP(=O)(Cn1ccc(NC(=O)c2cc(OC(C)C)cc(Oc3cnc(cn3)C(=O)N(C)C)c2)n1)OCC | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >8.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of hERG by patch clamp method |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c02110
BindingDB Entry DOI: 10.7270/Q2057KV7 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50585836
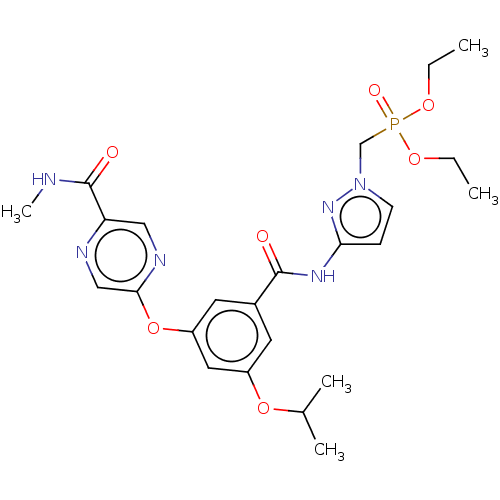
(CHEMBL5082691)Show SMILES CCOP(=O)(Cn1ccc(NC(=O)c2cc(OC(C)C)cc(Oc3cnc(cn3)C(=O)NC)c2)n1)OCC | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >8.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of hERG by patch clamp method |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c02110
BindingDB Entry DOI: 10.7270/Q2057KV7 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50585835
(CHEMBL5079866)Show SMILES CCOP(=O)(Cn1ccc(NC(=O)c2cc(OC(C)C)cc(Oc3cnc(cn3)C(N)=O)c2)n1)OCC | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >8.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of hERG by patch clamp method |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c02110
BindingDB Entry DOI: 10.7270/Q2057KV7 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50585813

(CHEMBL5072532)Show SMILES CCOP(=O)(Cn1ccc(NC(=O)c2cc(OC(C)C)cc(Oc3cnc(cn3)C(=O)N3CCC3)c2)n1)OCC | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | >8.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of hERG by patch clamp method |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c02110
BindingDB Entry DOI: 10.7270/Q2057KV7 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50585833
(CHEMBL5094606)Show SMILES CCOP(=O)(Cn1ccc(NC(=O)c2cc(OC(C)C)cc(Oc3cnc(nc3)C(=O)N3CCC3)c2)n1)OCC | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >8.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of hERG by patch clamp method |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c02110
BindingDB Entry DOI: 10.7270/Q2057KV7 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50585834

(CHEMBL5077438)Show SMILES CCOP(=O)(Cn1ccc(NC(=O)c2cc(OC(C)C)cc(Oc3ccc(nn3)C(=O)N3CCC3)c2)n1)OCC | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >8.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of hERG by patch clamp method |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c02110
BindingDB Entry DOI: 10.7270/Q2057KV7 |
More data for this
Ligand-Target Pair | |
Sodium/glucose cotransporter 2
(Homo sapiens (Human)) | BDBM20875
(1-(2,4-dihydroxy-6-{[(2S,3R,4S,5S,6R)-3,4,5-trihyd...)Show SMILES OC[C@H]1O[C@@H](Oc2cc(O)cc(O)c2C(=O)CCc2ccc(O)cc2)[C@H](O)[C@@H](O)[C@@H]1O Show InChI InChI=1S/C21H24O10/c22-9-16-18(27)19(28)20(29)21(31-16)30-15-8-12(24)7-14(26)17(15)13(25)6-3-10-1-4-11(23)5-2-10/h1-2,4-5,7-8,16,18-24,26-29H,3,6,9H2/t16-,18-,19+,20-,21-/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | n/a | n/a | 35.6 | n/a | n/a | 7.2 | 22 |
Bristol-Myers Squibb Company
| Assay Description
Inhibitors were assayed for the ability to inhibit [14C]AMG uptake in a protein-free buffer over a 2 h incubation period. The response curve was fitt... |
J Med Chem 51: 1145-9 (2008)
Article DOI: 10.1021/jm701272q
BindingDB Entry DOI: 10.7270/Q2PN93X4 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Sodium/glucose cotransporter 2
(Homo sapiens (Human)) | BDBM20877
(3-(1-benzofuran-5-yl)-1-(2-hydroxy-4-methyl-6-{[(2...)Show SMILES Cc1cc(O)c(C(=O)CCc2ccc3occc3c2)c(O[C@@H]2O[C@H](CO)[C@@H](O)[C@H](O)[C@H]2O)c1 Show InChI InChI=1S/C24H26O9/c1-12-8-16(27)20(15(26)4-2-13-3-5-17-14(10-13)6-7-31-17)18(9-12)32-24-23(30)22(29)21(28)19(11-25)33-24/h3,5-10,19,21-25,27-30H,2,4,11H2,1H3/t19-,21-,22+,23-,24-/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 6.60 | n/a | n/a | 7.2 | 22 |
Bristol-Myers Squibb Company
| Assay Description
Inhibitors were assayed for the ability to inhibit [14C]AMG uptake in a protein-free buffer over a 2 h incubation period. The response curve was fitt... |
J Med Chem 51: 1145-9 (2008)
Article DOI: 10.1021/jm701272q
BindingDB Entry DOI: 10.7270/Q2PN93X4 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data