 Found 10 hits with Last Name = 'reyes-moreno' and Initial = 'c'
Found 10 hits with Last Name = 'reyes-moreno' and Initial = 'c' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50576969
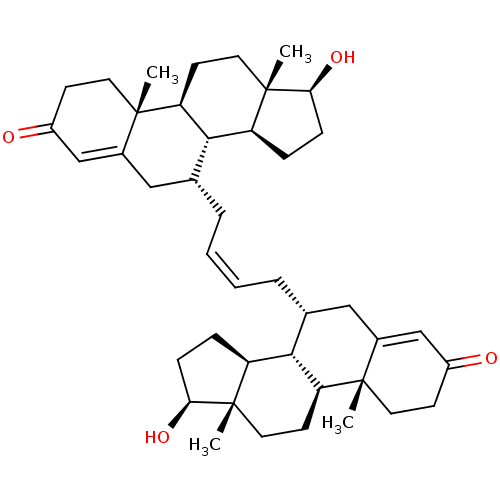
(CHEMBL4863362)Show SMILES [H][C@@]12CC[C@H](O)[C@@]1(C)CC[C@@]1([H])[C@@]2([H])[C@H](C\C=C/C[C@@H]2CC3=CC(=O)CC[C@]3(C)[C@@]3([H])CC[C@]4(C)[C@@H](O)CC[C@@]4([H])[C@]23[H])CC2=CC(=O)CC[C@]12C |r,t:23,50| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| n/a | n/a | 800 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human CYP3A4 using BFC as substrate |
Citation and Details
Article DOI: 10.1016/j.ejmech.2021.113496
BindingDB Entry DOI: 10.7270/Q2S1869N |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50576967

(CHEMBL4862783)Show SMILES [H][C@@]12CC[C@H](O)[C@@]1(C)CC[C@@]1([H])[C@@]2([H])[C@@H](CC2=CC(=O)CC[C@]12C)\C=C\C=C\C=C\[C@@H]1CC2=CC(=O)CC[C@]2(C)[C@@]2([H])CC[C@]3(C)[C@@H](O)CC[C@@]3([H])[C@]12[H] |r,t:18,36| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human CYP3A4 using BFC as substrate |
Citation and Details
Article DOI: 10.1016/j.ejmech.2021.113496
BindingDB Entry DOI: 10.7270/Q2S1869N |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50576968
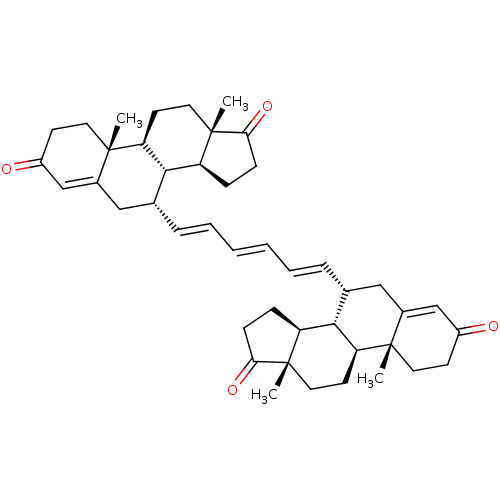
(CHEMBL4871524)Show SMILES [H][C@@]12CCC(=O)[C@@]1(C)CC[C@@]1([H])[C@@]2([H])[C@@H](CC2=CC(=O)CC[C@]12C)\C=C\C=C\C=C\[C@@H]1CC2=CC(=O)CC[C@]2(C)[C@@]2([H])CC[C@]3(C)C(=O)CC[C@@]3([H])[C@]12[H] |r,t:18,36| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 3.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human CYP3A4 using BFC as substrate |
Citation and Details
Article DOI: 10.1016/j.ejmech.2021.113496
BindingDB Entry DOI: 10.7270/Q2S1869N |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50576970

(CHEMBL4863066)Show SMILES [H][C@@]12CC[C@H](O)[C@@]1(C)CC[C@@]1([H])[C@@]2([H])[C@H](C\C=C\C[C@@H]2CC3=CC(=O)CC[C@]3(C)[C@@]3([H])CC[C@]4(C)[C@@H](O)CC[C@@]4([H])[C@]23[H])CC2=CC(=O)CC[C@]12C |r,t:23,50| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 6.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human CYP3A4 using BFC as substrate |
Citation and Details
Article DOI: 10.1016/j.ejmech.2021.113496
BindingDB Entry DOI: 10.7270/Q2S1869N |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50576966

(CHEMBL4864434)Show SMILES [H][C@@]12CC[C@@H](O)[C@@]1(C)CC[C@@]1([H])[C@@]2([H])[C@@H](CC2=CC(=O)CC[C@]12C)\C=C\C=C\C=C\[C@@H]1CC2=CC(=O)CC[C@]2(C)[C@@]2([H])CC[C@]3(C)[C@H](O)CC[C@@]3([H])[C@]12[H] |r,t:18,36| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 2.20E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human CYP3A4 using BFC as substrate |
Citation and Details
Article DOI: 10.1016/j.ejmech.2021.113496
BindingDB Entry DOI: 10.7270/Q2S1869N |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50576969

(CHEMBL4863362)Show SMILES [H][C@@]12CC[C@H](O)[C@@]1(C)CC[C@@]1([H])[C@@]2([H])[C@H](C\C=C/C[C@@H]2CC3=CC(=O)CC[C@]3(C)[C@@]3([H])CC[C@]4(C)[C@@H](O)CC[C@@]4([H])[C@]23[H])CC2=CC(=O)CC[C@]12C |r,t:23,50| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| n/a | n/a | n/a | 370 | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human CYP3A4 assessed as dissociation constant |
Citation and Details
Article DOI: 10.1016/j.ejmech.2021.113496
BindingDB Entry DOI: 10.7270/Q2S1869N |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50576968

(CHEMBL4871524)Show SMILES [H][C@@]12CCC(=O)[C@@]1(C)CC[C@@]1([H])[C@@]2([H])[C@@H](CC2=CC(=O)CC[C@]12C)\C=C\C=C\C=C\[C@@H]1CC2=CC(=O)CC[C@]2(C)[C@@]2([H])CC[C@]3(C)C(=O)CC[C@@]3([H])[C@]12[H] |r,t:18,36| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | n/a | 1.40E+3 | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human CYP3A4 assessed as dissociation constant |
Citation and Details
Article DOI: 10.1016/j.ejmech.2021.113496
BindingDB Entry DOI: 10.7270/Q2S1869N |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50576967

(CHEMBL4862783)Show SMILES [H][C@@]12CC[C@H](O)[C@@]1(C)CC[C@@]1([H])[C@@]2([H])[C@@H](CC2=CC(=O)CC[C@]12C)\C=C\C=C\C=C\[C@@H]1CC2=CC(=O)CC[C@]2(C)[C@@]2([H])CC[C@]3(C)[C@@H](O)CC[C@@]3([H])[C@]12[H] |r,t:18,36| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | n/a | 5.50E+3 | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human CYP3A4 assessed as dissociation constant |
Citation and Details
Article DOI: 10.1016/j.ejmech.2021.113496
BindingDB Entry DOI: 10.7270/Q2S1869N |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50576966

(CHEMBL4864434)Show SMILES [H][C@@]12CC[C@@H](O)[C@@]1(C)CC[C@@]1([H])[C@@]2([H])[C@@H](CC2=CC(=O)CC[C@]12C)\C=C\C=C\C=C\[C@@H]1CC2=CC(=O)CC[C@]2(C)[C@@]2([H])CC[C@]3(C)[C@H](O)CC[C@@]3([H])[C@]12[H] |r,t:18,36| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | n/a | 1.60E+3 | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human CYP3A4 assessed as dissociation constant |
Citation and Details
Article DOI: 10.1016/j.ejmech.2021.113496
BindingDB Entry DOI: 10.7270/Q2S1869N |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50576970

(CHEMBL4863066)Show SMILES [H][C@@]12CC[C@H](O)[C@@]1(C)CC[C@@]1([H])[C@@]2([H])[C@H](C\C=C\C[C@@H]2CC3=CC(=O)CC[C@]3(C)[C@@]3([H])CC[C@]4(C)[C@@H](O)CC[C@@]4([H])[C@]23[H])CC2=CC(=O)CC[C@]12C |r,t:23,50| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | n/a | 7.10E+3 | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human CYP3A4 assessed as dissociation constant |
Citation and Details
Article DOI: 10.1016/j.ejmech.2021.113496
BindingDB Entry DOI: 10.7270/Q2S1869N |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data