 Found 13 hits in this display
Found 13 hits in this display Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Caspase-1
(Homo sapiens (Human)) | BDBM160794
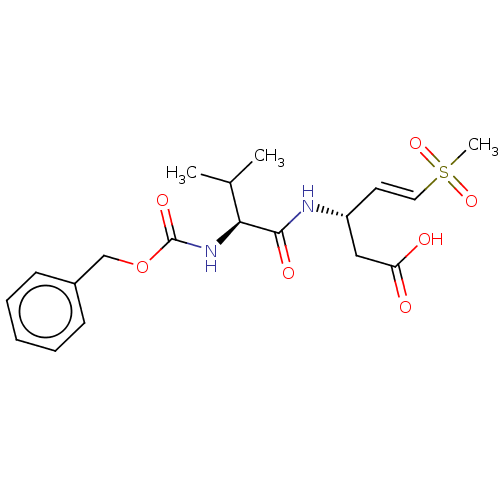
(US10167313, Compound 61 | US9045524, 61)Show SMILES CC(C)[C@H](NC(=O)OCc1ccccc1)C(=O)N[C@@H](CC(O)=O)\C=C\S(C)(=O)=O |r| Show InChI InChI=1S/C19H26N2O7S/c1-13(2)17(21-19(25)28-12-14-7-5-4-6-8-14)18(24)20-15(11-16(22)23)9-10-29(3,26)27/h4-10,13,15,17H,11-12H2,1-3H3,(H,20,24)(H,21,25)(H,22,23)/b10-9+/t15-,17+/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| US Patent
| n/a | n/a | >3.33E+3 | n/a | n/a | n/a | n/a | n/a | 37 |
NOVAGENESIS FOUNDATION
US Patent
| Assay Description
Selectivity of compound 55, compound 63, compound 48, compound 57, compound 88 toward caspase-1 (pro-inflammatory group), caspase-5 (group I), caspas... |
US Patent US9045524 (2015)
BindingDB Entry DOI: 10.7270/Q2GT5KX8 |
More data for this
Ligand-Target Pair | |
Caspase-5
(Homo sapiens (Human)) | BDBM160794

(US10167313, Compound 61 | US9045524, 61)Show SMILES CC(C)[C@H](NC(=O)OCc1ccccc1)C(=O)N[C@@H](CC(O)=O)\C=C\S(C)(=O)=O |r| Show InChI InChI=1S/C19H26N2O7S/c1-13(2)17(21-19(25)28-12-14-7-5-4-6-8-14)18(24)20-15(11-16(22)23)9-10-29(3,26)27/h4-10,13,15,17H,11-12H2,1-3H3,(H,20,24)(H,21,25)(H,22,23)/b10-9+/t15-,17+/m1/s1 | KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| US Patent
| n/a | n/a | >3.33E+3 | n/a | n/a | n/a | n/a | n/a | 37 |
NOVAGENESIS FOUNDATION
US Patent
| Assay Description
Selectivity of compound 55, compound 63, compound 48, compound 57, compound 88 toward caspase-1 (pro-inflammatory group), caspase-5 (group I), caspas... |
US Patent US9045524 (2015)
BindingDB Entry DOI: 10.7270/Q2GT5KX8 |
More data for this
Ligand-Target Pair | |
Caspase-7
(Homo sapiens (Human)) | BDBM160794

(US10167313, Compound 61 | US9045524, 61)Show SMILES CC(C)[C@H](NC(=O)OCc1ccccc1)C(=O)N[C@@H](CC(O)=O)\C=C\S(C)(=O)=O |r| Show InChI InChI=1S/C19H26N2O7S/c1-13(2)17(21-19(25)28-12-14-7-5-4-6-8-14)18(24)20-15(11-16(22)23)9-10-29(3,26)27/h4-10,13,15,17H,11-12H2,1-3H3,(H,20,24)(H,21,25)(H,22,23)/b10-9+/t15-,17+/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| US Patent
| n/a | n/a | >3.33E+3 | n/a | n/a | n/a | n/a | n/a | 37 |
NOVAGENESIS FOUNDATION
US Patent
| Assay Description
Selectivity of compound 55, compound 63, compound 48, compound 57, compound 88 toward caspase-1 (pro-inflammatory group), caspase-5 (group I), caspas... |
US Patent US9045524 (2015)
BindingDB Entry DOI: 10.7270/Q2GT5KX8 |
More data for this
Ligand-Target Pair | |
Caspase-9
(Homo sapiens (Human)) | BDBM160794

(US10167313, Compound 61 | US9045524, 61)Show SMILES CC(C)[C@H](NC(=O)OCc1ccccc1)C(=O)N[C@@H](CC(O)=O)\C=C\S(C)(=O)=O |r| Show InChI InChI=1S/C19H26N2O7S/c1-13(2)17(21-19(25)28-12-14-7-5-4-6-8-14)18(24)20-15(11-16(22)23)9-10-29(3,26)27/h4-10,13,15,17H,11-12H2,1-3H3,(H,20,24)(H,21,25)(H,22,23)/b10-9+/t15-,17+/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| US Patent
| n/a | n/a | >3.33E+3 | n/a | n/a | n/a | n/a | n/a | 37 |
NOVAGENESIS FOUNDATION
US Patent
| Assay Description
Selectivity of compound 55, compound 63, compound 48, compound 57, compound 88 toward caspase-1 (pro-inflammatory group), caspase-5 (group I), caspas... |
US Patent US9045524 (2015)
BindingDB Entry DOI: 10.7270/Q2GT5KX8 |
More data for this
Ligand-Target Pair | |
Caspase-3
(Homo sapiens (Human)) | BDBM160794

(US10167313, Compound 61 | US9045524, 61)Show SMILES CC(C)[C@H](NC(=O)OCc1ccccc1)C(=O)N[C@@H](CC(O)=O)\C=C\S(C)(=O)=O |r| Show InChI InChI=1S/C19H26N2O7S/c1-13(2)17(21-19(25)28-12-14-7-5-4-6-8-14)18(24)20-15(11-16(22)23)9-10-29(3,26)27/h4-10,13,15,17H,11-12H2,1-3H3,(H,20,24)(H,21,25)(H,22,23)/b10-9+/t15-,17+/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| US Patent
| n/a | n/a | >3.33E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
NOVAGENESIS FOUNDATION
US Patent
| Assay Description
To test the efficacy of caspase-3 inhibitors at the cellular level, the ability of selected compounds to inhibit the proteolytic cleavage of PARP (po... |
US Patent US9045524 (2015)
BindingDB Entry DOI: 10.7270/Q2GT5KX8 |
More data for this
Ligand-Target Pair | |
Caspase-5
(Homo sapiens (Human)) | BDBM160794

(US10167313, Compound 61 | US9045524, 61)Show SMILES CC(C)[C@H](NC(=O)OCc1ccccc1)C(=O)N[C@@H](CC(O)=O)\C=C\S(C)(=O)=O |r| Show InChI InChI=1S/C19H26N2O7S/c1-13(2)17(21-19(25)28-12-14-7-5-4-6-8-14)18(24)20-15(11-16(22)23)9-10-29(3,26)27/h4-10,13,15,17H,11-12H2,1-3H3,(H,20,24)(H,21,25)(H,22,23)/b10-9+/t15-,17+/m1/s1 | KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| US Patent
| n/a | n/a | >3.33E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Genesis Technologies Limited
US Patent
| Assay Description
Selectivity of compound 55, compound 63, compound 48, compound 57, compound 88 toward caspase-1 (pro-inflammatory group), caspase-5 (group I), caspas... |
US Patent US10167313 (2019)
BindingDB Entry DOI: 10.7270/Q2Q81G59 |
More data for this
Ligand-Target Pair | |
Caspase-9
(Homo sapiens (Human)) | BDBM160794

(US10167313, Compound 61 | US9045524, 61)Show SMILES CC(C)[C@H](NC(=O)OCc1ccccc1)C(=O)N[C@@H](CC(O)=O)\C=C\S(C)(=O)=O |r| Show InChI InChI=1S/C19H26N2O7S/c1-13(2)17(21-19(25)28-12-14-7-5-4-6-8-14)18(24)20-15(11-16(22)23)9-10-29(3,26)27/h4-10,13,15,17H,11-12H2,1-3H3,(H,20,24)(H,21,25)(H,22,23)/b10-9+/t15-,17+/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| US Patent
| n/a | n/a | >3.33E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Genesis Technologies Limited
US Patent
| Assay Description
Selectivity of compound 55, compound 63, compound 48, compound 57, compound 88 toward caspase-1 (pro-inflammatory group), caspase-5 (group I), caspas... |
US Patent US10167313 (2019)
BindingDB Entry DOI: 10.7270/Q2Q81G59 |
More data for this
Ligand-Target Pair | |
Caspase-7
(Homo sapiens (Human)) | BDBM160794

(US10167313, Compound 61 | US9045524, 61)Show SMILES CC(C)[C@H](NC(=O)OCc1ccccc1)C(=O)N[C@@H](CC(O)=O)\C=C\S(C)(=O)=O |r| Show InChI InChI=1S/C19H26N2O7S/c1-13(2)17(21-19(25)28-12-14-7-5-4-6-8-14)18(24)20-15(11-16(22)23)9-10-29(3,26)27/h4-10,13,15,17H,11-12H2,1-3H3,(H,20,24)(H,21,25)(H,22,23)/b10-9+/t15-,17+/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| US Patent
| n/a | n/a | >3.33E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Genesis Technologies Limited
US Patent
| Assay Description
Selectivity of compound 55, compound 63, compound 48, compound 57, compound 88 toward caspase-1 (pro-inflammatory group), caspase-5 (group I), caspas... |
US Patent US10167313 (2019)
BindingDB Entry DOI: 10.7270/Q2Q81G59 |
More data for this
Ligand-Target Pair | |
Caspase-1
(Homo sapiens (Human)) | BDBM160794

(US10167313, Compound 61 | US9045524, 61)Show SMILES CC(C)[C@H](NC(=O)OCc1ccccc1)C(=O)N[C@@H](CC(O)=O)\C=C\S(C)(=O)=O |r| Show InChI InChI=1S/C19H26N2O7S/c1-13(2)17(21-19(25)28-12-14-7-5-4-6-8-14)18(24)20-15(11-16(22)23)9-10-29(3,26)27/h4-10,13,15,17H,11-12H2,1-3H3,(H,20,24)(H,21,25)(H,22,23)/b10-9+/t15-,17+/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| US Patent
| n/a | n/a | >3.33E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Genesis Technologies Limited
US Patent
| Assay Description
Selectivity of compound 55, compound 63, compound 48, compound 57, compound 88 toward caspase-1 (pro-inflammatory group), caspase-5 (group I), caspas... |
US Patent US10167313 (2019)
BindingDB Entry DOI: 10.7270/Q2Q81G59 |
More data for this
Ligand-Target Pair | |
Caspase-3
(Homo sapiens (Human)) | BDBM160794

(US10167313, Compound 61 | US9045524, 61)Show SMILES CC(C)[C@H](NC(=O)OCc1ccccc1)C(=O)N[C@@H](CC(O)=O)\C=C\S(C)(=O)=O |r| Show InChI InChI=1S/C19H26N2O7S/c1-13(2)17(21-19(25)28-12-14-7-5-4-6-8-14)18(24)20-15(11-16(22)23)9-10-29(3,26)27/h4-10,13,15,17H,11-12H2,1-3H3,(H,20,24)(H,21,25)(H,22,23)/b10-9+/t15-,17+/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| US Patent
| n/a | n/a | >3.33E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Genesis Technologies Limited
US Patent
| Assay Description
Selectivity of compound 55, compound 63, compound 48, compound 57, compound 88 toward caspase-1 (pro-inflammatory group), caspase-5 (group I), caspas... |
US Patent US10167313 (2019)
BindingDB Entry DOI: 10.7270/Q2Q81G59 |
More data for this
Ligand-Target Pair | |
Caspase-3
(Homo sapiens (Human)) | BDBM50326094

((S)-3-((S)-2-(benzyloxycarbonylamino)-3-methylbuta...)Show SMILES CC(C)[C@H](NC(=O)OCc1ccccc1)C(=O)N[C@@H](CC(O)=O)C=CS(C)(=O)=O |r,w:24.25| Show InChI InChI=1S/C19H26N2O7S/c1-13(2)17(21-19(25)28-12-14-7-5-4-6-8-14)18(24)20-15(11-16(22)23)9-10-29(3,26)27/h4-10,13,15,17H,11-12H2,1-3H3,(H,20,24)(H,21,25)(H,22,23)/t15-,17+/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3.53E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Lisbon
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant caspase-3 expressed in Saccharomyces cerevisiae CG379 assessed as growth stimulation after 2 days |
Eur J Med Chem 46: 2141-6 (2011)
Article DOI: 10.1016/j.ejmech.2011.02.067
BindingDB Entry DOI: 10.7270/Q2QV3MW0 |
More data for this
Ligand-Target Pair | |
Caspase-3
(Homo sapiens (Human)) | BDBM50326094

((S)-3-((S)-2-(benzyloxycarbonylamino)-3-methylbuta...)Show SMILES CC(C)[C@H](NC(=O)OCc1ccccc1)C(=O)N[C@@H](CC(O)=O)C=CS(C)(=O)=O |r,w:24.25| Show InChI InChI=1S/C19H26N2O7S/c1-13(2)17(21-19(25)28-12-14-7-5-4-6-8-14)18(24)20-15(11-16(22)23)9-10-29(3,26)27/h4-10,13,15,17H,11-12H2,1-3H3,(H,20,24)(H,21,25)(H,22,23)/t15-,17+/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.33E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Lisbon
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant caspase 3 assessed as fluorescent 7-amido-4-methylcoumarin release |
Eur J Med Chem 45: 3858-63 (2010)
Article DOI: 10.1016/j.ejmech.2010.05.039
BindingDB Entry DOI: 10.7270/Q2X63N59 |
More data for this
Ligand-Target Pair | |
Caspase-7
(Homo sapiens (Human)) | BDBM50326094

((S)-3-((S)-2-(benzyloxycarbonylamino)-3-methylbuta...)Show SMILES CC(C)[C@H](NC(=O)OCc1ccccc1)C(=O)N[C@@H](CC(O)=O)C=CS(C)(=O)=O |r,w:24.25| Show InChI InChI=1S/C19H26N2O7S/c1-13(2)17(21-19(25)28-12-14-7-5-4-6-8-14)18(24)20-15(11-16(22)23)9-10-29(3,26)27/h4-10,13,15,17H,11-12H2,1-3H3,(H,20,24)(H,21,25)(H,22,23)/t15-,17+/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >2.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Lisbon
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant caspase 7 assessed as fluorescent 7-amido-4-methylcoumarin release |
Eur J Med Chem 45: 3858-63 (2010)
Article DOI: 10.1016/j.ejmech.2010.05.039
BindingDB Entry DOI: 10.7270/Q2X63N59 |
More data for this
Ligand-Target Pair | |