 Found 5 hits in this display
Found 5 hits in this display Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Cytosol aminopeptidase
(Homo sapiens (Human)) | BDBM50046325
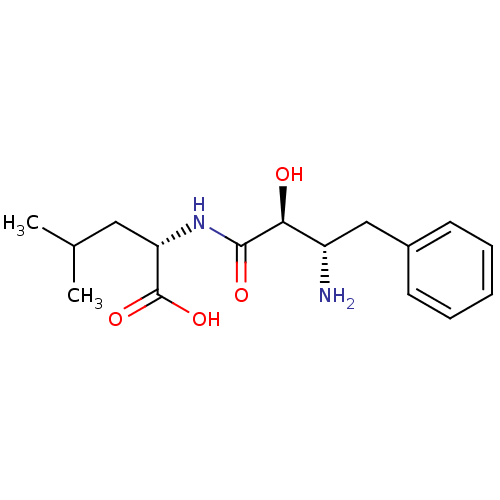
((S)-2-((2S,3S)-3-Amino-2-hydroxy-4-phenyl-butyryla...)Show SMILES CC(C)C[C@H](NC(=O)[C@@H](O)[C@@H](N)Cc1ccccc1)C(O)=O Show InChI InChI=1S/C16H24N2O4/c1-10(2)8-13(16(21)22)18-15(20)14(19)12(17)9-11-6-4-3-5-7-11/h3-7,10,12-14,19H,8-9,17H2,1-2H3,(H,18,20)(H,21,22)/t12-,13-,14-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| 1.70E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Compound was evaluated for the inhibition of Leucine aminopeptidase and the inhibition constant was determined after preincubating the enzyme and inh... |
J Med Chem 27: 417-22 (1984)
BindingDB Entry DOI: 10.7270/Q280535G |
More data for this
Ligand-Target Pair | |
Aminopeptidase N
(Sus scrofa (Pig)) | BDBM50046325

((S)-2-((2S,3S)-3-Amino-2-hydroxy-4-phenyl-butyryla...)Show SMILES CC(C)C[C@H](NC(=O)[C@@H](O)[C@@H](N)Cc1ccccc1)C(O)=O Show InChI InChI=1S/C16H24N2O4/c1-10(2)8-13(16(21)22)18-15(20)14(19)12(17)9-11-6-4-3-5-7-11/h3-7,10,12-14,19H,8-9,17H2,1-2H3,(H,18,20)(H,21,22)/t12-,13-,14-/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
PubMed
| 2.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Effect of inhibitor structure on the slow binding inhibition of aminopeptidase M was determined and Ki* was reported which is obtained by the equatio... |
J Med Chem 27: 417-22 (1984)
BindingDB Entry DOI: 10.7270/Q280535G |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Aminopeptidase N
(Sus scrofa (Pig)) | BDBM50046325

((S)-2-((2S,3S)-3-Amino-2-hydroxy-4-phenyl-butyryla...)Show SMILES CC(C)C[C@H](NC(=O)[C@@H](O)[C@@H](N)Cc1ccccc1)C(O)=O Show InChI InChI=1S/C16H24N2O4/c1-10(2)8-13(16(21)22)18-15(20)14(19)12(17)9-11-6-4-3-5-7-11/h3-7,10,12-14,19H,8-9,17H2,1-2H3,(H,18,20)(H,21,22)/t12-,13-,14-/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
PubMed
| 5.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Effect of inhibitor structure on the slow binding inhibition of aminopeptidase M was determined and the Ki was reported which is = k2/k1 |
J Med Chem 27: 417-22 (1984)
BindingDB Entry DOI: 10.7270/Q280535G |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Aminopeptidase B
(Rattus norvegicus) | BDBM50046325

((S)-2-((2S,3S)-3-Amino-2-hydroxy-4-phenyl-butyryla...)Show SMILES CC(C)C[C@H](NC(=O)[C@@H](O)[C@@H](N)Cc1ccccc1)C(O)=O Show InChI InChI=1S/C16H24N2O4/c1-10(2)8-13(16(21)22)18-15(20)14(19)12(17)9-11-6-4-3-5-7-11/h3-7,10,12-14,19H,8-9,17H2,1-2H3,(H,18,20)(H,21,22)/t12-,13-,14-/m0/s1 | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 4.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of rat liver aminopeptidase B using 0.2 mM L-leucine-beta-naphthylamide by colorimetry |
J Med Chem 20: 510-5 (1977)
BindingDB Entry DOI: 10.7270/Q29W0H11 |
More data for this
Ligand-Target Pair | |
Leukotriene A-4 hydrolase
(Homo sapiens (Human)) | BDBM50046325

((S)-2-((2S,3S)-3-Amino-2-hydroxy-4-phenyl-butyryla...)Show SMILES CC(C)C[C@H](NC(=O)[C@@H](O)[C@@H](N)Cc1ccccc1)C(O)=O Show InChI InChI=1S/C16H24N2O4/c1-10(2)8-13(16(21)22)18-15(20)14(19)12(17)9-11-6-4-3-5-7-11/h3-7,10,12-14,19H,8-9,17H2,1-2H3,(H,18,20)(H,21,22)/t12-,13-,14-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
PubMed
| n/a | n/a | 2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Scripps Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of amidase activity of LTA4 hydrolase purified from human leukocytes |
J Med Chem 36: 211-20 (1993)
BindingDB Entry DOI: 10.7270/Q2T152P3 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |