 Found 10 hits in this display
Found 10 hits in this display Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Calcium-dependent protein kinase 1
(Cryptosporidium parvum) | BDBM50148652
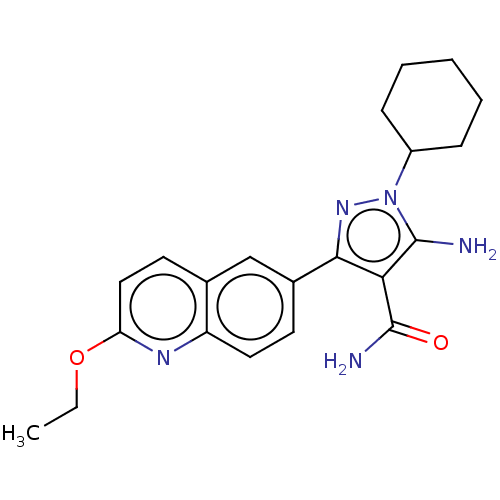
(CHEMBL3770802 | US9518026, Example 57 | US9956214,...)Show SMILES CCOc1ccc2cc(ccc2n1)-c1nn(C2CCCCC2)c(N)c1C(N)=O Show InChI InChI=1S/C21H25N5O2/c1-2-28-17-11-9-13-12-14(8-10-16(13)24-17)19-18(21(23)27)20(22)26(25-19)15-6-4-3-5-7-15/h8-12,15H,2-7,22H2,1H3,(H2,23,27) | UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| US Patent
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Boehringer Ingelheim (Canada) Ltd.
| Assay Description
Inhibition of TgCDPK1 and CpCDPK1 was determined using a luminescent kinase assay which measures ATP depletion in the presence of the Syntide 2 pepti... |
Bioorg Med Chem Lett 19: 1199-205 (2009)
BindingDB Entry DOI: 10.7270/Q23J3G91 |
More data for this
Ligand-Target Pair | |
Uncharacterized protein
(Cryptosporidium parvum) | BDBM50148652

(CHEMBL3770802 | US9518026, Example 57 | US9956214,...)Show SMILES CCOc1ccc2cc(ccc2n1)-c1nn(C2CCCCC2)c(N)c1C(N)=O Show InChI InChI=1S/C21H25N5O2/c1-2-28-17-11-9-13-12-14(8-10-16(13)24-17)19-18(21(23)27)20(22)26(25-19)15-6-4-3-5-7-15/h8-12,15H,2-7,22H2,1H3,(H2,23,27) | PDB
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| US Patent
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Washington Through its Center For Commercialization
US Patent
| Assay Description
Inhibition of TgCDPK1 and CpCDPK1 was determined using a luminescent kinase assay which measures ATP depletion in the presence of the Syntide 2 pepti... |
US Patent US9518026 (2016)
BindingDB Entry DOI: 10.7270/Q2BV7JN4 |
More data for this
Ligand-Target Pair | |
Calmodulin-domain protein kinase 1
(Toxoplasma gondii) | BDBM50148652

(CHEMBL3770802 | US9518026, Example 57 | US9956214,...)Show SMILES CCOc1ccc2cc(ccc2n1)-c1nn(C2CCCCC2)c(N)c1C(N)=O Show InChI InChI=1S/C21H25N5O2/c1-2-28-17-11-9-13-12-14(8-10-16(13)24-17)19-18(21(23)27)20(22)26(25-19)15-6-4-3-5-7-15/h8-12,15H,2-7,22H2,1H3,(H2,23,27) | PDB
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| US Patent
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Boehringer Ingelheim (Canada) Ltd.
| Assay Description
Inhibition of TgCDPK1 and CpCDPK1 was determined using a luminescent kinase assay which measures ATP depletion in the presence of the Syntide 2 pepti... |
Bioorg Med Chem Lett 19: 1199-205 (2009)
BindingDB Entry DOI: 10.7270/Q23J3G91 |
More data for this
Ligand-Target Pair | |
Calmodulin-domain protein kinase 1
(Toxoplasma gondii) | BDBM50148652

(CHEMBL3770802 | US9518026, Example 57 | US9956214,...)Show SMILES CCOc1ccc2cc(ccc2n1)-c1nn(C2CCCCC2)c(N)c1C(N)=O Show InChI InChI=1S/C21H25N5O2/c1-2-28-17-11-9-13-12-14(8-10-16(13)24-17)19-18(21(23)27)20(22)26(25-19)15-6-4-3-5-7-15/h8-12,15H,2-7,22H2,1H3,(H2,23,27) | PDB
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 5.5 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Washington
Curated by ChEMBL
| Assay Description
Inhibition of Toxoplasma gondii CDPK1 assessed as ATP consumption using (Biotin-C6-PLARTLSVAGLPGKK) as substrate after 90 mins by luciferase reporter... |
ACS Med Chem Lett 6: 1184-1189 (2015)
Article DOI: 10.1021/acsmedchemlett.5b00319
BindingDB Entry DOI: 10.7270/Q2NK3GXD |
More data for this
Ligand-Target Pair | |
Calmodulin-domain protein kinase 1
(Toxoplasma gondii) | BDBM50148652

(CHEMBL3770802 | US9518026, Example 57 | US9956214,...)Show SMILES CCOc1ccc2cc(ccc2n1)-c1nn(C2CCCCC2)c(N)c1C(N)=O Show InChI InChI=1S/C21H25N5O2/c1-2-28-17-11-9-13-12-14(8-10-16(13)24-17)19-18(21(23)27)20(22)26(25-19)15-6-4-3-5-7-15/h8-12,15H,2-7,22H2,1H3,(H2,23,27) | PDB
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Washington
Curated by ChEMBL
| Assay Description
Inhibition of recombinant Toxoplasma gondii CDPK1 using Syntide 2 as substrate assessed as decrease in depletion of ATP after 90 mins by Kinase glo l... |
Bioorg Med Chem Lett 26: 5487-5491 (2016)
Article DOI: 10.1016/j.bmcl.2016.10.014
BindingDB Entry DOI: 10.7270/Q2J968BR |
More data for this
Ligand-Target Pair | |
Calmodulin-domain protein kinase 1
(Toxoplasma gondii) | BDBM50148652

(CHEMBL3770802 | US9518026, Example 57 | US9956214,...)Show SMILES CCOc1ccc2cc(ccc2n1)-c1nn(C2CCCCC2)c(N)c1C(N)=O Show InChI InChI=1S/C21H25N5O2/c1-2-28-17-11-9-13-12-14(8-10-16(13)24-17)19-18(21(23)27)20(22)26(25-19)15-6-4-3-5-7-15/h8-12,15H,2-7,22H2,1H3,(H2,23,27) | PDB
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| US Patent
| n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Washington Through its Center For Commercialization
US Patent
| Assay Description
Inhibition of TgCDPK1 and CpCDPK1 was determined using a luminescent kinase assay which measures ATP depletion in the presence of the Syntide 2 pepti... |
US Patent US9518026 (2016)
BindingDB Entry DOI: 10.7270/Q2BV7JN4 |
More data for this
Ligand-Target Pair | |
cGMP-dependent protein kinase
(Plasmodium falciparum) | BDBM50148652

(CHEMBL3770802 | US9518026, Example 57 | US9956214,...)Show SMILES CCOc1ccc2cc(ccc2n1)-c1nn(C2CCCCC2)c(N)c1C(N)=O Show InChI InChI=1S/C21H25N5O2/c1-2-28-17-11-9-13-12-14(8-10-16(13)24-17)19-18(21(23)27)20(22)26(25-19)15-6-4-3-5-7-15/h8-12,15H,2-7,22H2,1H3,(H2,23,27) | PDB
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2.36E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Washington
Curated by ChEMBL
| Assay Description
Inhibition of recombinant Plasmodium falciparum PKG assessed as decrease in depletion of ATP by Kinase glo luciferase assay |
Bioorg Med Chem Lett 26: 5487-5491 (2016)
Article DOI: 10.1016/j.bmcl.2016.10.014
BindingDB Entry DOI: 10.7270/Q2J968BR |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase Src
(Homo sapiens (Human)) | BDBM50148652

(CHEMBL3770802 | US9518026, Example 57 | US9956214,...)Show SMILES CCOc1ccc2cc(ccc2n1)-c1nn(C2CCCCC2)c(N)c1C(N)=O Show InChI InChI=1S/C21H25N5O2/c1-2-28-17-11-9-13-12-14(8-10-16(13)24-17)19-18(21(23)27)20(22)26(25-19)15-6-4-3-5-7-15/h8-12,15H,2-7,22H2,1H3,(H2,23,27) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Washington
Curated by ChEMBL
| Assay Description
Inhibition of human SRC using Ac-EIYGEFKKK as substrate after 90 mins by luciferase reporter assay |
ACS Med Chem Lett 6: 1184-1189 (2015)
Article DOI: 10.1021/acsmedchemlett.5b00319
BindingDB Entry DOI: 10.7270/Q2NK3GXD |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase Src
(Homo sapiens (Human)) | BDBM50148652

(CHEMBL3770802 | US9518026, Example 57 | US9956214,...)Show SMILES CCOc1ccc2cc(ccc2n1)-c1nn(C2CCCCC2)c(N)c1C(N)=O Show InChI InChI=1S/C21H25N5O2/c1-2-28-17-11-9-13-12-14(8-10-16(13)24-17)19-18(21(23)27)20(22)26(25-19)15-6-4-3-5-7-15/h8-12,15H,2-7,22H2,1H3,(H2,23,27) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Washington
Curated by ChEMBL
| Assay Description
Inhibition of human SRC using Ac-EIYGEFKKK as substrate after 90 mins by Kinase glo luciferase assay |
Bioorg Med Chem Lett 26: 5487-5491 (2016)
Article DOI: 10.1016/j.bmcl.2016.10.014
BindingDB Entry DOI: 10.7270/Q2J968BR |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50148652

(CHEMBL3770802 | US9518026, Example 57 | US9956214,...)Show SMILES CCOc1ccc2cc(ccc2n1)-c1nn(C2CCCCC2)c(N)c1C(N)=O Show InChI InChI=1S/C21H25N5O2/c1-2-28-17-11-9-13-12-14(8-10-16(13)24-17)19-18(21(23)27)20(22)26(25-19)15-6-4-3-5-7-15/h8-12,15H,2-7,22H2,1H3,(H2,23,27) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 1.41E+4 | n/a | n/a | n/a | n/a |
University of Washington
Curated by ChEMBL
| Assay Description
Inhibition of human ERG |
Bioorg Med Chem Lett 26: 5487-5491 (2016)
Article DOI: 10.1016/j.bmcl.2016.10.014
BindingDB Entry DOI: 10.7270/Q2J968BR |
More data for this
Ligand-Target Pair | |