 Found 4 hits in this display
Found 4 hits in this display Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Cysteinyl leukotriene receptor 1/2
(Homo sapiens (Human)) | BDBM50227367
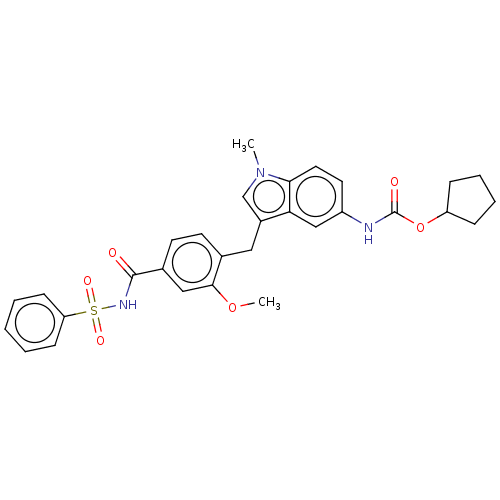
(CHEMBL48435)Show SMILES COc1cc(ccc1Cc1cn(C)c2ccc(NC(=O)OC3CCCC3)cc12)C(=O)NS(=O)(=O)c1ccccc1 Show InChI InChI=1S/C30H31N3O6S/c1-33-19-22(26-18-23(14-15-27(26)33)31-30(35)39-24-8-6-7-9-24)16-20-12-13-21(17-28(20)38-2)29(34)32-40(36,37)25-10-4-3-5-11-25/h3-5,10-15,17-19,24H,6-9,16H2,1-2H3,(H,31,35)(H,32,34) | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
ICI Pharmaceuticals Group
Curated by ChEMBL
| Assay Description
Inhibition constant for displacement of [3H]LTD4 on guinea pig lung parenchymal membranes. |
J Med Chem 33: 1781-90 (1990)
BindingDB Entry DOI: 10.7270/Q2862FDP |
More data for this
Ligand-Target Pair | |
Cysteinyl leukotriene receptor 1
(GUINEA PIG) | BDBM50227367

(CHEMBL48435)Show SMILES COc1cc(ccc1Cc1cn(C)c2ccc(NC(=O)OC3CCCC3)cc12)C(=O)NS(=O)(=O)c1ccccc1 Show InChI InChI=1S/C30H31N3O6S/c1-33-19-22(26-18-23(14-15-27(26)33)31-30(35)39-24-8-6-7-9-24)16-20-12-13-21(17-28(20)38-2)29(34)32-40(36,37)25-10-4-3-5-11-25/h3-5,10-15,17-19,24H,6-9,16H2,1-2H3,(H,31,35)(H,32,34) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Goethe-University Frankfurt
Curated by ChEMBL
| Assay Description
Displacement of [3H]LTD4 from cysteinyl leukotriene receptor 1 in Hartley guinea pig parenchymal membrane after 30 mins by liquid scintillation count... |
J Med Chem 60: 5235-5266 (2017)
Article DOI: 10.1021/acs.jmedchem.6b01287
BindingDB Entry DOI: 10.7270/Q2T43WDQ |
More data for this
Ligand-Target Pair | |
Bifunctional epoxide hydrolase 2
(Homo sapiens (Human)) | BDBM50227367

(CHEMBL48435)Show SMILES COc1cc(ccc1Cc1cn(C)c2ccc(NC(=O)OC3CCCC3)cc12)C(=O)NS(=O)(=O)c1ccccc1 Show InChI InChI=1S/C30H31N3O6S/c1-33-19-22(26-18-23(14-15-27(26)33)31-30(35)39-24-8-6-7-9-24)16-20-12-13-21(17-28(20)38-2)29(34)32-40(36,37)25-10-4-3-5-11-25/h3-5,10-15,17-19,24H,6-9,16H2,1-2H3,(H,31,35)(H,32,34) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Goethe University Frankfurt
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human sEH using PHOME as substrate preincubated for 30 mins followed by substrate addition and measured after 30 mins by sp... |
J Med Chem 61: 5758-5764 (2018)
Article DOI: 10.1021/acs.jmedchem.8b00458
BindingDB Entry DOI: 10.7270/Q2TX3J21 |
More data for this
Ligand-Target Pair | |
Peroxisome proliferator-activated receptor gamma
(Homo sapiens (Human)) | BDBM50227367

(CHEMBL48435)Show SMILES COc1cc(ccc1Cc1cn(C)c2ccc(NC(=O)OC3CCCC3)cc12)C(=O)NS(=O)(=O)c1ccccc1 Show InChI InChI=1S/C30H31N3O6S/c1-33-19-22(26-18-23(14-15-27(26)33)31-30(35)39-24-8-6-7-9-24)16-20-12-13-21(17-28(20)38-2)29(34)32-40(36,37)25-10-4-3-5-11-25/h3-5,10-15,17-19,24H,6-9,16H2,1-2H3,(H,31,35)(H,32,34) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 230 | n/a | n/a | n/a | n/a |
Goethe University Frankfurt
Curated by ChEMBL
| Assay Description
Partial agonist activity at human PPARgamma LBD expressed HEK293T cells after 12 to 14 hrs by dual-glo luciferase reporter gene assay |
J Med Chem 61: 5758-5764 (2018)
Article DOI: 10.1021/acs.jmedchem.8b00458
BindingDB Entry DOI: 10.7270/Q2TX3J21 |
More data for this
Ligand-Target Pair | |