 Found 12 hits in this display
Found 12 hits in this display Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Thymidine kinase, cytosolic
(Rattus norvegicus) | BDBM50205415
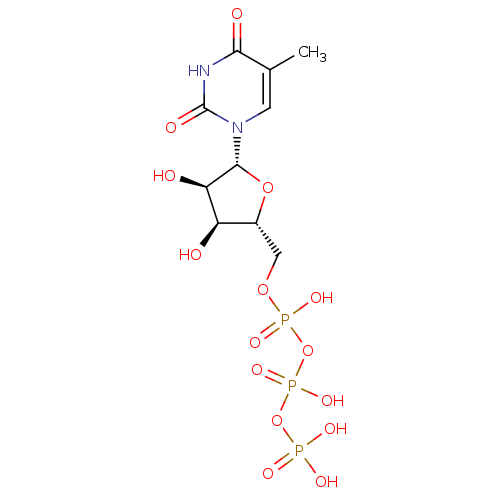
(({[({[(2R,3S,4R,5R)-3,4-dihydroxy-5-(5-methyl-2,4-...)Show SMILES Cc1cn([C@@H]2O[C@H](COP(O)(=O)OP(O)(=O)OP(O)(O)=O)[C@@H](O)[C@H]2O)c(=O)[nH]c1=O Show InChI InChI=1S/C10H17N2O15P3/c1-4-2-12(10(16)11-8(4)15)9-7(14)6(13)5(25-9)3-24-29(20,21)27-30(22,23)26-28(17,18)19/h2,5-7,9,13-14H,3H2,1H3,(H,20,21)(H,22,23)(H,11,15,16)(H2,17,18,19)/t5-,6-,7-,9-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
PDB
UniChem
Similars
| PubMed
| 3.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Evaluated for the mixed objective Non-competitive inhibition constant Ki against TdR varied rat mitochondrial thymidine kinase |
J Med Chem 25: 801-5 (1982)
BindingDB Entry DOI: 10.7270/Q2BZ66M6 |
More data for this
Ligand-Target Pair | |
Thymidine kinase, cytosolic
(Rattus norvegicus) | BDBM50205415

(({[({[(2R,3S,4R,5R)-3,4-dihydroxy-5-(5-methyl-2,4-...)Show SMILES Cc1cn([C@@H]2O[C@H](COP(O)(=O)OP(O)(=O)OP(O)(O)=O)[C@@H](O)[C@H]2O)c(=O)[nH]c1=O Show InChI InChI=1S/C10H17N2O15P3/c1-4-2-12(10(16)11-8(4)15)9-7(14)6(13)5(25-9)3-24-29(20,21)27-30(22,23)26-28(17,18)19/h2,5-7,9,13-14H,3H2,1H3,(H,20,21)(H,22,23)(H,11,15,16)(H2,17,18,19)/t5-,6-,7-,9-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
PDB
UniChem
Similars
| PubMed
| 6.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Evaluated for the Non-competitive inhibition constant Ki against TdR varied rat cytoplasmic soluble thymidine kinase |
J Med Chem 25: 801-5 (1982)
BindingDB Entry DOI: 10.7270/Q2BZ66M6 |
More data for this
Ligand-Target Pair | |
DNA polymerase alpha catalytic subunit
(Homo sapiens (Human)) | BDBM50205415

(({[({[(2R,3S,4R,5R)-3,4-dihydroxy-5-(5-methyl-2,4-...)Show SMILES Cc1cn([C@@H]2O[C@H](COP(O)(=O)OP(O)(=O)OP(O)(O)=O)[C@@H](O)[C@H]2O)c(=O)[nH]c1=O Show InChI InChI=1S/C10H17N2O15P3/c1-4-2-12(10(16)11-8(4)15)9-7(14)6(13)5(25-9)3-24-29(20,21)27-30(22,23)26-28(17,18)19/h2,5-7,9,13-14H,3H2,1H3,(H,20,21)(H,22,23)(H,11,15,16)(H2,17,18,19)/t5-,6-,7-,9-/m1/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Max-Delbr�ck-Centrum f�r Molekulare Medizin
Curated by ChEMBL
| Assay Description
Compound was evaluated for 50% inhibition of DHBV DNA polymerase (duck hepatitis B virus) |
J Med Chem 41: 2040-6 (1998)
Article DOI: 10.1021/jm9704210
BindingDB Entry DOI: 10.7270/Q2FT8MQT |
More data for this
Ligand-Target Pair | |
DNA polymerase alpha catalytic subunit
(Homo sapiens (Human)) | BDBM50205415

(({[({[(2R,3S,4R,5R)-3,4-dihydroxy-5-(5-methyl-2,4-...)Show SMILES Cc1cn([C@@H]2O[C@H](COP(O)(=O)OP(O)(=O)OP(O)(O)=O)[C@@H](O)[C@H]2O)c(=O)[nH]c1=O Show InChI InChI=1S/C10H17N2O15P3/c1-4-2-12(10(16)11-8(4)15)9-7(14)6(13)5(25-9)3-24-29(20,21)27-30(22,23)26-28(17,18)19/h2,5-7,9,13-14H,3H2,1H3,(H,20,21)(H,22,23)(H,11,15,16)(H2,17,18,19)/t5-,6-,7-,9-/m1/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Max-Delbr�ck-Centrum f�r Molekulare Medizin
Curated by ChEMBL
| Assay Description
Compound was evaluated for 50% inhibition of DHBV DNA polymerase (duck hepatitis B virus) |
J Med Chem 41: 2040-6 (1998)
Article DOI: 10.1021/jm9704210
BindingDB Entry DOI: 10.7270/Q2FT8MQT |
More data for this
Ligand-Target Pair | |
DNA polymerase subunit gamma-1
(Homo sapiens (Human)) | BDBM50205415

(({[({[(2R,3S,4R,5R)-3,4-dihydroxy-5-(5-methyl-2,4-...)Show SMILES Cc1cn([C@@H]2O[C@H](COP(O)(=O)OP(O)(=O)OP(O)(O)=O)[C@@H](O)[C@H]2O)c(=O)[nH]c1=O Show InChI InChI=1S/C10H17N2O15P3/c1-4-2-12(10(16)11-8(4)15)9-7(14)6(13)5(25-9)3-24-29(20,21)27-30(22,23)26-28(17,18)19/h2,5-7,9,13-14H,3H2,1H3,(H,20,21)(H,22,23)(H,11,15,16)(H2,17,18,19)/t5-,6-,7-,9-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 8.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Max-Delbr�ck-Centrum f�r Molekulare Medizin
Curated by ChEMBL
| Assay Description
Compound was evaluated for the inhibition of cellular DNA polymerase (gamma) |
J Med Chem 41: 2040-6 (1998)
Article DOI: 10.1021/jm9704210
BindingDB Entry DOI: 10.7270/Q2FT8MQT |
More data for this
Ligand-Target Pair | |
Reverse transcriptase/RNaseH
(Human immunodeficiency virus 1) | BDBM50205415

(({[({[(2R,3S,4R,5R)-3,4-dihydroxy-5-(5-methyl-2,4-...)Show SMILES Cc1cn([C@@H]2O[C@H](COP(O)(=O)OP(O)(=O)OP(O)(O)=O)[C@@H](O)[C@H]2O)c(=O)[nH]c1=O Show InChI InChI=1S/C10H17N2O15P3/c1-4-2-12(10(16)11-8(4)15)9-7(14)6(13)5(25-9)3-24-29(20,21)27-30(22,23)26-28(17,18)19/h2,5-7,9,13-14H,3H2,1H3,(H,20,21)(H,22,23)(H,11,15,16)(H2,17,18,19)/t5-,6-,7-,9-/m1/s1 | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | >2.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Max-Delbr�ck-Centrum f�r Molekulare Medizin
Curated by ChEMBL
| Assay Description
Compound was evaluated for 50% inhibition of HIV-RT (HIV reverse transcriptase) |
J Med Chem 41: 2040-6 (1998)
Article DOI: 10.1021/jm9704210
BindingDB Entry DOI: 10.7270/Q2FT8MQT |
More data for this
Ligand-Target Pair | |
DNA polymerase beta
(Homo sapiens (Human)) | BDBM50205415

(({[({[(2R,3S,4R,5R)-3,4-dihydroxy-5-(5-methyl-2,4-...)Show SMILES Cc1cn([C@@H]2O[C@H](COP(O)(=O)OP(O)(=O)OP(O)(O)=O)[C@@H](O)[C@H]2O)c(=O)[nH]c1=O Show InChI InChI=1S/C10H17N2O15P3/c1-4-2-12(10(16)11-8(4)15)9-7(14)6(13)5(25-9)3-24-29(20,21)27-30(22,23)26-28(17,18)19/h2,5-7,9,13-14H,3H2,1H3,(H,20,21)(H,22,23)(H,11,15,16)(H2,17,18,19)/t5-,6-,7-,9-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | >2.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Max-Delbr�ck-Centrum f�r Molekulare Medizin
Curated by ChEMBL
| Assay Description
Compound was evaluated for the inhibition of cellular DNA polymerase (beta) |
J Med Chem 41: 2040-6 (1998)
Article DOI: 10.1021/jm9704210
BindingDB Entry DOI: 10.7270/Q2FT8MQT |
More data for this
Ligand-Target Pair | |
P2Y purinoceptor 6
(Homo sapiens (Human)) | BDBM50205415

(({[({[(2R,3S,4R,5R)-3,4-dihydroxy-5-(5-methyl-2,4-...)Show SMILES Cc1cn([C@@H]2O[C@H](COP(O)(=O)OP(O)(=O)OP(O)(O)=O)[C@@H](O)[C@H]2O)c(=O)[nH]c1=O Show InChI InChI=1S/C10H17N2O15P3/c1-4-2-12(10(16)11-8(4)15)9-7(14)6(13)5(25-9)3-24-29(20,21)27-30(22,23)26-28(17,18)19/h2,5-7,9,13-14H,3H2,1H3,(H,20,21)(H,22,23)(H,11,15,16)(H2,17,18,19)/t5-,6-,7-,9-/m1/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 140 | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases
Curated by ChEMBL
| Assay Description
Agonist activity at human recombinant P2Y6 receptor expressed in 1321N1 cells assessed as stimulation of phospholipase C |
J Med Chem 50: 1166-76 (2007)
Article DOI: 10.1021/jm060903o
BindingDB Entry DOI: 10.7270/Q2PK0GZ1 |
More data for this
Ligand-Target Pair | |
P2Y purinoceptor 4
(Homo sapiens (Human)) | BDBM50205415

(({[({[(2R,3S,4R,5R)-3,4-dihydroxy-5-(5-methyl-2,4-...)Show SMILES Cc1cn([C@@H]2O[C@H](COP(O)(=O)OP(O)(=O)OP(O)(O)=O)[C@@H](O)[C@H]2O)c(=O)[nH]c1=O Show InChI InChI=1S/C10H17N2O15P3/c1-4-2-12(10(16)11-8(4)15)9-7(14)6(13)5(25-9)3-24-29(20,21)27-30(22,23)26-28(17,18)19/h2,5-7,9,13-14H,3H2,1H3,(H,20,21)(H,22,23)(H,11,15,16)(H2,17,18,19)/t5-,6-,7-,9-/m1/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 3.90E+3 | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases
Curated by ChEMBL
| Assay Description
Agonist activity at human recombinant P2Y4 receptor expressed in 1321N1 cells assessed as stimulation of phospholipase C |
J Med Chem 50: 1166-76 (2007)
Article DOI: 10.1021/jm060903o
BindingDB Entry DOI: 10.7270/Q2PK0GZ1 |
More data for this
Ligand-Target Pair | |
Reverse transcriptase
(Human immunodeficiency virus 1) | BDBM50205415

(({[({[(2R,3S,4R,5R)-3,4-dihydroxy-5-(5-methyl-2,4-...)Show SMILES Cc1cn([C@@H]2O[C@H](COP(O)(=O)OP(O)(=O)OP(O)(O)=O)[C@@H](O)[C@H]2O)c(=O)[nH]c1=O Show InChI InChI=1S/C10H17N2O15P3/c1-4-2-12(10(16)11-8(4)15)9-7(14)6(13)5(25-9)3-24-29(20,21)27-30(22,23)26-28(17,18)19/h2,5-7,9,13-14H,3H2,1H3,(H,20,21)(H,22,23)(H,11,15,16)(H2,17,18,19)/t5-,6-,7-,9-/m1/s1 | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | 2.10E+3 | n/a | n/a | n/a | n/a | n/a |
Veterans Affairs Medical Center
Curated by ChEMBL
| Assay Description
Binding affinity to HIV1 reverse transcriptase assessed as L-3'-azido-NTP incorporation in nascent DNA |
Eur J Med Chem 46: 3832-44 (2011)
Article DOI: 10.1016/j.ejmech.2011.05.051
BindingDB Entry DOI: 10.7270/Q2RN3BQP |
More data for this
Ligand-Target Pair | |
Reverse transcriptase protein
(Human immunodeficiency virus 1) | BDBM50205415

(({[({[(2R,3S,4R,5R)-3,4-dihydroxy-5-(5-methyl-2,4-...)Show SMILES Cc1cn([C@@H]2O[C@H](COP(O)(=O)OP(O)(=O)OP(O)(O)=O)[C@@H](O)[C@H]2O)c(=O)[nH]c1=O Show InChI InChI=1S/C10H17N2O15P3/c1-4-2-12(10(16)11-8(4)15)9-7(14)6(13)5(25-9)3-24-29(20,21)27-30(22,23)26-28(17,18)19/h2,5-7,9,13-14H,3H2,1H3,(H,20,21)(H,22,23)(H,11,15,16)(H2,17,18,19)/t5-,6-,7-,9-/m1/s1 | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | 2.90E+3 | n/a | n/a | n/a | n/a | n/a |
Veterans Affairs Medical Center
Curated by ChEMBL
| Assay Description
Binding affinity to HIV1 reverse transcriptase M184V mutant assessed as L-3'-azido-NTP incorporation in nascent DNA |
Eur J Med Chem 46: 3832-44 (2011)
Article DOI: 10.1016/j.ejmech.2011.05.051
BindingDB Entry DOI: 10.7270/Q2RN3BQP |
More data for this
Ligand-Target Pair | |
P2Y purinoceptor 2
(Homo sapiens (Human)) | BDBM50205415

(({[({[(2R,3S,4R,5R)-3,4-dihydroxy-5-(5-methyl-2,4-...)Show SMILES Cc1cn([C@@H]2O[C@H](COP(O)(=O)OP(O)(=O)OP(O)(O)=O)[C@@H](O)[C@H]2O)c(=O)[nH]c1=O Show InChI InChI=1S/C10H17N2O15P3/c1-4-2-12(10(16)11-8(4)15)9-7(14)6(13)5(25-9)3-24-29(20,21)27-30(22,23)26-28(17,18)19/h2,5-7,9,13-14H,3H2,1H3,(H,20,21)(H,22,23)(H,11,15,16)(H2,17,18,19)/t5-,6-,7-,9-/m1/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 480 | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases
Curated by ChEMBL
| Assay Description
Agonist activity at human recombinant P2Y2 receptor expressed in 1321N1 cells assessed as stimulation of phospholipase C |
J Med Chem 50: 1166-76 (2007)
Article DOI: 10.1021/jm060903o
BindingDB Entry DOI: 10.7270/Q2PK0GZ1 |
More data for this
Ligand-Target Pair | |