

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Inositol 1,4,5-trisphosphate receptor type 1/2/3 (Homo sapiens (Human)) | BDBM50211666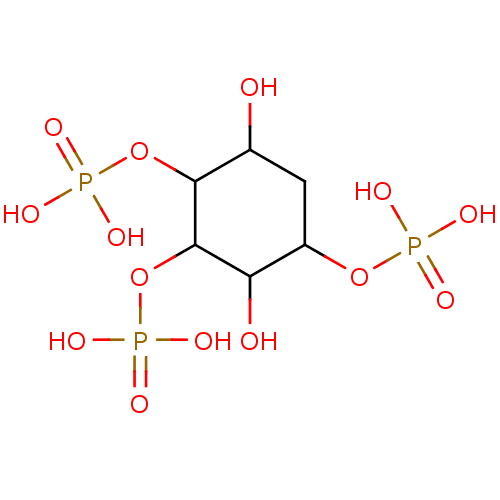 (CHEMBL1161456) | PDB UniProtKB/SwissProt antibodypedia antibodypedia antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid PDB UniChem Similars | Article | 2.92E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Compound was evaluated for its ability to inhibit Inositol phosphorylation | Bioorg Med Chem Lett 1: 705-710 (1991) Article DOI: 10.1016/S0960-894X(01)81052-0 BindingDB Entry DOI: 10.7270/Q2348KV6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Inositol 1,4,5-trisphosphate receptor type 1 (Homo sapiens (Human)) | BDBM50075183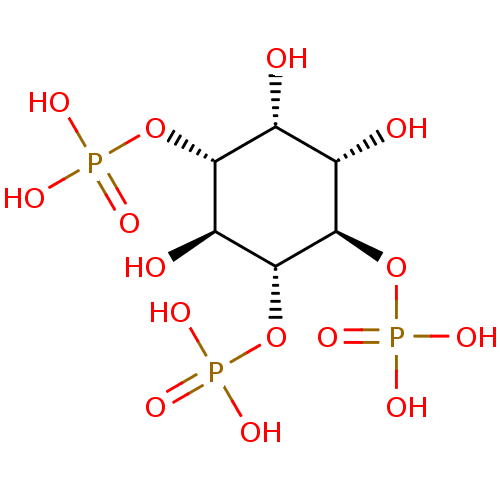 (1,4,5-Insp3 | 1D-myo-inositol 1,4,5-triphosphate |...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | PDB Article | n/a | n/a | 300 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Binding affinity against Ins(1,4,5)P3 receptor in swiss 3T3 cells | Bioorg Med Chem Lett 5: 1225-1230 (1995) Article DOI: 10.1016/0960-894X(95)00200-D BindingDB Entry DOI: 10.7270/Q21836F4 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Inositol 1,4,5-trisphosphate receptor type 1 (Homo sapiens (Human)) | BDBM50111931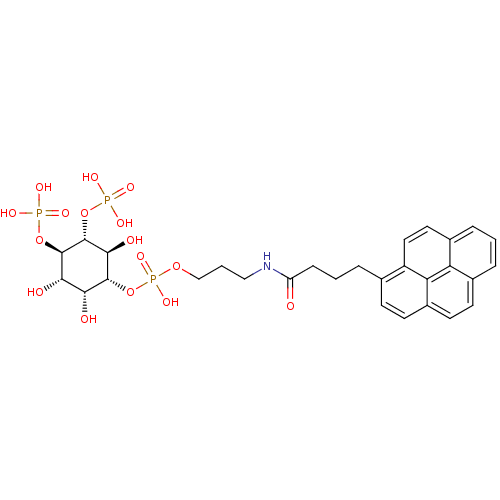 (CHEMBL434103 | Phosphoric acid 3-(4-pyren-1-yl-but...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | PubMed | n/a | n/a | n/a | 140 | n/a | n/a | n/a | n/a | n/a |
The University of Tokyo Curated by ChEMBL | Assay Description Dissociation constant of the compound was determined using IP3-binding domain (IBD) of human Type 1 inositol 1,4,5-trisphosphate receptor | Bioorg Med Chem Lett 12: 911-3 (2002) BindingDB Entry DOI: 10.7270/Q2HX1C0H | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Inositol 1,4,5-trisphosphate receptor type 1 (Homo sapiens (Human)) | BDBM50078573 (CHEMBL297235 | Phosphoric acid 3-amino-propyl este...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | n/a | 1.50E+3 | n/a | n/a | n/a | n/a | n/a |
The University of Tokyo Curated by ChEMBL | Assay Description Dissociation constant of the compound was determined using IP3-binding domain (IBD) of human Type 1 inositol 1,4,5-trisphosphate receptor | Bioorg Med Chem Lett 12: 911-3 (2002) BindingDB Entry DOI: 10.7270/Q2HX1C0H | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Inositol 1,4,5-trisphosphate receptor type 1 (Homo sapiens (Human)) | BDBM50078572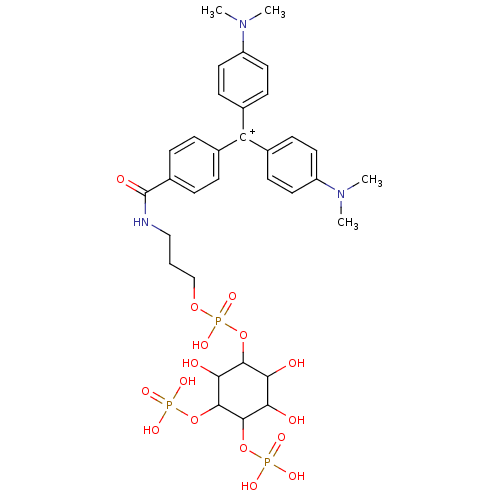 (CHEMBL172534 | CHEMBL297496 | {4-[(4-Dimethylamino...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | n/a | 1.20 | n/a | n/a | n/a | n/a | n/a |
The University of Tokyo Curated by ChEMBL | Assay Description Dissociation constant of the compound was determined using IP3-binding domain (IBD) of human Type 1 inositol 1,4,5-trisphosphate receptor | Bioorg Med Chem Lett 12: 911-3 (2002) BindingDB Entry DOI: 10.7270/Q2HX1C0H | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Inositol 1,4,5-trisphosphate receptor type 1 (Homo sapiens (Human)) | BDBM50111928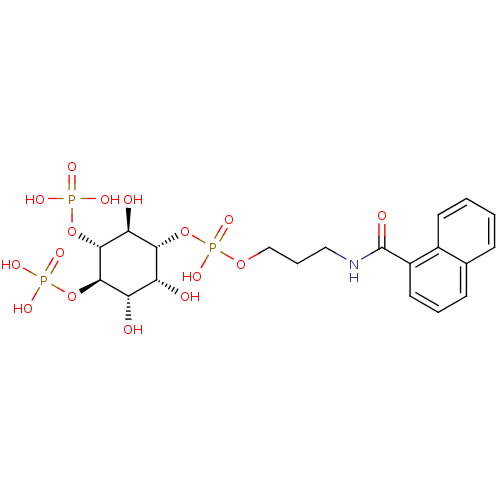 (CHEMBL169923 | Phosphoric acid 3-[(naphthalene-1-c...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | PubMed | n/a | n/a | n/a | 160 | n/a | n/a | n/a | n/a | n/a |
The University of Tokyo Curated by ChEMBL | Assay Description Dissociation constant of the compound was determined using IP3-binding domain (IBD) of human Type 1 inositol 1,4,5-trisphosphate receptor | Bioorg Med Chem Lett 12: 911-3 (2002) BindingDB Entry DOI: 10.7270/Q2HX1C0H | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Inositol 1,4,5-trisphosphate receptor type 1 (Homo sapiens (Human)) | BDBM50111932 (CHEMBL369479 | inositol 1,4,5-Trisphosphate analog...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | PubMed | n/a | n/a | n/a | 620 | n/a | n/a | n/a | n/a | n/a |
The University of Tokyo Curated by ChEMBL | Assay Description Dissociation constant of the compound was determined using IP3-binding domain (IBD) of human Type 1 inositol 1,4,5-trisphosphate receptor | Bioorg Med Chem Lett 12: 911-3 (2002) BindingDB Entry DOI: 10.7270/Q2HX1C0H | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Inositol 1,4,5-trisphosphate receptor type 1 (Homo sapiens (Human)) | BDBM50075183 (1,4,5-Insp3 | 1D-myo-inositol 1,4,5-triphosphate |...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | PDB PubMed | n/a | n/a | n/a | 260 | n/a | n/a | n/a | n/a | n/a |
The University of Tokyo Curated by ChEMBL | Assay Description Dissociation constant of the compound was determined using IP3-binding domain (IBD) of human Type 1 inositol 1,4,5-trisphosphate receptor | Bioorg Med Chem Lett 12: 911-3 (2002) BindingDB Entry DOI: 10.7270/Q2HX1C0H | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Inositol 1,4,5-trisphosphate receptor type 1 (Homo sapiens (Human)) | BDBM50111929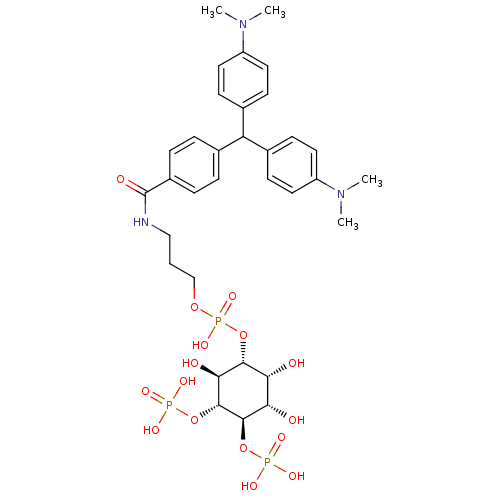 (CHEMBL352326 | Phosphoric acid 3-{4-[bis-(4-dimeth...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | n/a | 3.20E+3 | n/a | n/a | n/a | n/a | n/a |
The University of Tokyo Curated by ChEMBL | Assay Description Dissociation constant of the compound was determined using IP3-binding domain (IBD) of human Type 1 inositol 1,4,5-trisphosphate receptor | Bioorg Med Chem Lett 12: 911-3 (2002) BindingDB Entry DOI: 10.7270/Q2HX1C0H | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Inositol 1,4,5-trisphosphate receptor type 1 (Rattus norvegicus) | BDBM50184323 (CHEMBL379040 | phosphoric acid mono-{(2R,3R,4R,5R,...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 4.60 | n/a | n/a | n/a | n/a |
Hokkaido University Curated by ChEMBL | Assay Description Agonistic potency at rat IP3 type 1 receptor expressed in chicken DT40 cells | J Med Chem 49: 1900-9 (2006) Article DOI: 10.1021/jm051039n BindingDB Entry DOI: 10.7270/Q2MK6CG5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Inositol 1,4,5-trisphosphate receptor type 1 (Rattus norvegicus) | BDBM50184324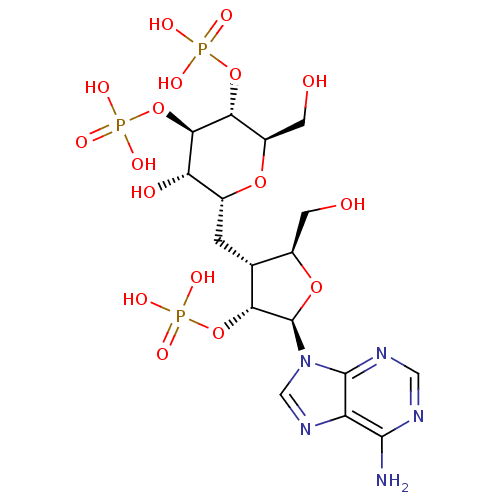 (CHEMBL383049 | phosphoric acid mono-{(2R,3R,4R,5S,...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 4 | n/a | n/a | n/a | n/a |
Hokkaido University Curated by ChEMBL | Assay Description Agonistic potency at rat IP3 type 1 receptor expressed in chicken DT40 cells | J Med Chem 49: 1900-9 (2006) Article DOI: 10.1021/jm051039n BindingDB Entry DOI: 10.7270/Q2MK6CG5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Inositol 1,4,5-trisphosphate receptor type 1 (Rattus norvegicus) | BDBM50184326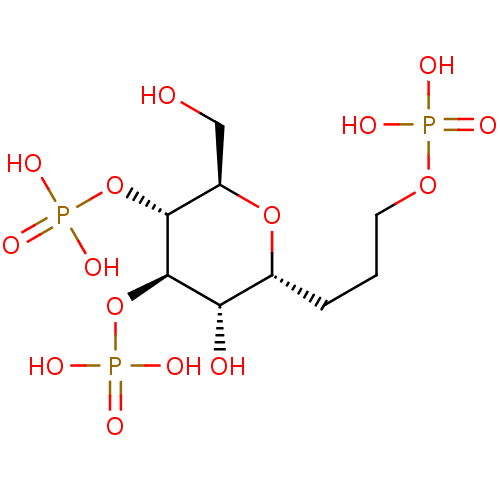 (CHEMBL380941 | phosphoric acid mono-[(2R,3R,4R,5S,...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 213 | n/a | n/a | n/a | n/a |
Hokkaido University Curated by ChEMBL | Assay Description Agonistic potency at rat IP3 type 1 receptor expressed in chicken DT40 cells | J Med Chem 49: 1900-9 (2006) Article DOI: 10.1021/jm051039n BindingDB Entry DOI: 10.7270/Q2MK6CG5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Inositol 1,4,5-trisphosphate receptor type 1 (Rattus norvegicus) | BDBM50184329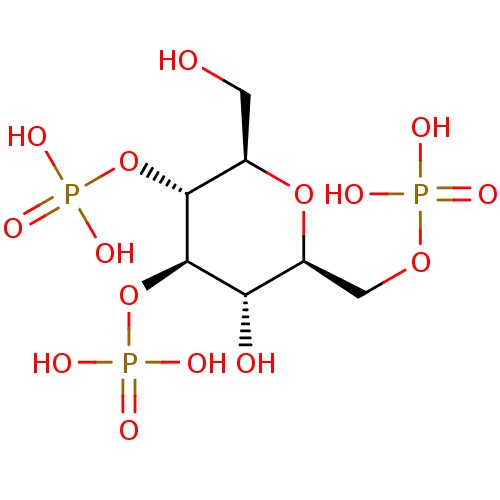 (CHEMBL383659 | phosphoric acid mono-((2R,3R,4R,5S,...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid PDB UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 1.08E+3 | n/a | n/a | n/a | n/a |
Hokkaido University Curated by ChEMBL | Assay Description Agonistic potency at rat IP3 type 1 receptor expressed in chicken DT40 cells | J Med Chem 49: 1900-9 (2006) Article DOI: 10.1021/jm051039n BindingDB Entry DOI: 10.7270/Q2MK6CG5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Inositol 1,4,5-trisphosphate receptor type 1 (Rattus norvegicus) | BDBM50184325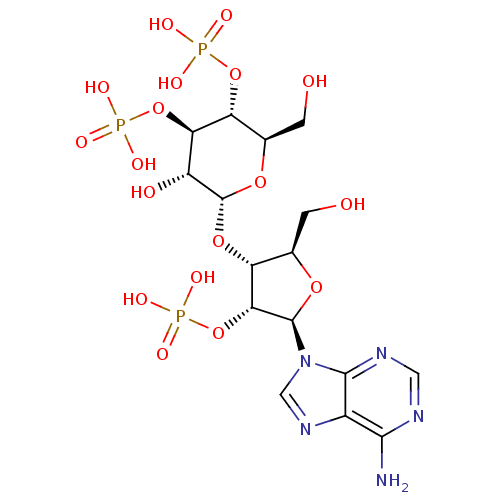 (2-[5-(6-amino-9H-9-purinyl)-2-hydroxymethyl-4-oxyp...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL KEGG PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | 2.10 | n/a | n/a | n/a | n/a |
Hokkaido University Curated by ChEMBL | Assay Description Agonistic potency at rat IP3 type 1 receptor expressed in chicken DT40 cells | J Med Chem 49: 1900-9 (2006) Article DOI: 10.1021/jm051039n BindingDB Entry DOI: 10.7270/Q2MK6CG5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Inositol 1,4,5-trisphosphate receptor type 1 (Rattus norvegicus) | BDBM50184328 (CHEMBL204903 | phosphoric acid mono-[(2R,3R,4R,5S,...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 2.04E+3 | n/a | n/a | n/a | n/a |
Hokkaido University Curated by ChEMBL | Assay Description Agonistic potency at rat IP3 type 1 receptor expressed in chicken DT40 cells | J Med Chem 49: 1900-9 (2006) Article DOI: 10.1021/jm051039n BindingDB Entry DOI: 10.7270/Q2MK6CG5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Inositol 1,4,5-trisphosphate receptor type 1 (Rattus norvegicus) | BDBM50075183 (1,4,5-Insp3 | 1D-myo-inositol 1,4,5-triphosphate |...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | n/a | n/a | n/a | n/a | 24.8 | n/a | n/a | n/a | n/a |
Hokkaido University Curated by ChEMBL | Assay Description Agonistic potency at rat IP3 type 1 receptor expressed in chicken DT40 cells | J Med Chem 49: 1900-9 (2006) Article DOI: 10.1021/jm051039n BindingDB Entry DOI: 10.7270/Q2MK6CG5 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Inositol 1,4,5-trisphosphate receptor type 1 (Rattus norvegicus) | BDBM50184327 (CHEMBL205303 | phosphoric acid mono-[(2R,3R,4R,5S,...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 4.43E+3 | n/a | n/a | n/a | n/a |
Hokkaido University Curated by ChEMBL | Assay Description Agonistic potency at rat IP3 type 1 receptor expressed in chicken DT40 cells | J Med Chem 49: 1900-9 (2006) Article DOI: 10.1021/jm051039n BindingDB Entry DOI: 10.7270/Q2MK6CG5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Inositol 1,4,5-trisphosphate receptor type 1 (Rattus norvegicus) | BDBM50184331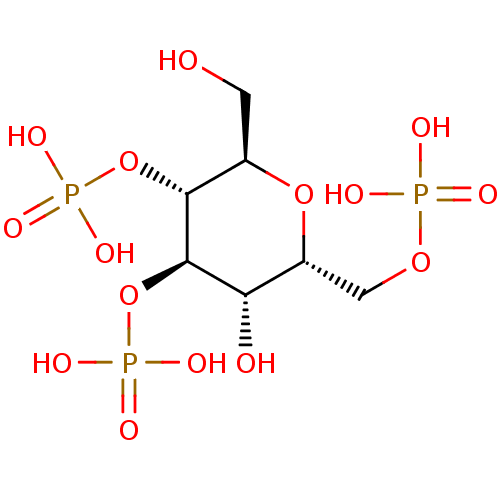 (CHEMBL204253 | phosphoric acid mono-((2R,3R,4R,5S,...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 394 | n/a | n/a | n/a | n/a |
Hokkaido University Curated by ChEMBL | Assay Description Agonistic potency at rat IP3 type 1 receptor expressed in chicken DT40 cells | J Med Chem 49: 1900-9 (2006) Article DOI: 10.1021/jm051039n BindingDB Entry DOI: 10.7270/Q2MK6CG5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Inositol 1,4,5-trisphosphate receptor type 1 (Rattus norvegicus) | BDBM50184332 (CHEMBL382185 | phosphoric acid mono-{(2R,3R,4R,5S,...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 11.3 | n/a | n/a | n/a | n/a |
Hokkaido University Curated by ChEMBL | Assay Description Agonistic potency at rat IP3 type 1 receptor expressed in chicken DT40 cells | J Med Chem 49: 1900-9 (2006) Article DOI: 10.1021/jm051039n BindingDB Entry DOI: 10.7270/Q2MK6CG5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Inositol 1,4,5-trisphosphate receptor type 1 (Rattus norvegicus) | BDBM50184330 (CHEMBL205008 | phosphoric acid mono-[(2R,3R,4R,5S,...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 49.2 | n/a | n/a | n/a | n/a |
Hokkaido University Curated by ChEMBL | Assay Description Agonistic potency at rat IP3 type 1 receptor expressed in chicken DT40 cells | J Med Chem 49: 1900-9 (2006) Article DOI: 10.1021/jm051039n BindingDB Entry DOI: 10.7270/Q2MK6CG5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Inositol 1,4,5-trisphosphate receptor type 1 (Rattus norvegicus) | BDBM50075183 (1,4,5-Insp3 | 1D-myo-inositol 1,4,5-triphosphate |...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | n/a | n/a | n/a | n/a | 20 | n/a | n/a | n/a | n/a |
University of Cambridge Curated by ChEMBL | Assay Description Agonist activity at rat recombinant IP3R1 expressed in chicken DT40 cells assessed as calcium release from intracellular stores | Nat Chem Biol 5: 631-9 (2009) Article DOI: 10.1038/nchembio.195 BindingDB Entry DOI: 10.7270/Q2C53M27 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Inositol 1,4,5-trisphosphate receptor type 1 (Rattus norvegicus) | BDBM50184325 (2-[5-(6-amino-9H-9-purinyl)-2-hydroxymethyl-4-oxyp...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL KEGG PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | 1.5 | n/a | n/a | n/a | n/a |
University of Cambridge Curated by ChEMBL | Assay Description Agonist activity at rat recombinant IP3R1 expressed in chicken DT40 cells assessed as calcium release from intracellular stores | Nat Chem Biol 5: 631-9 (2009) Article DOI: 10.1038/nchembio.195 BindingDB Entry DOI: 10.7270/Q2C53M27 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Inositol 1,4,5-trisphosphate receptor type 1 (Rattus norvegicus) | BDBM50323704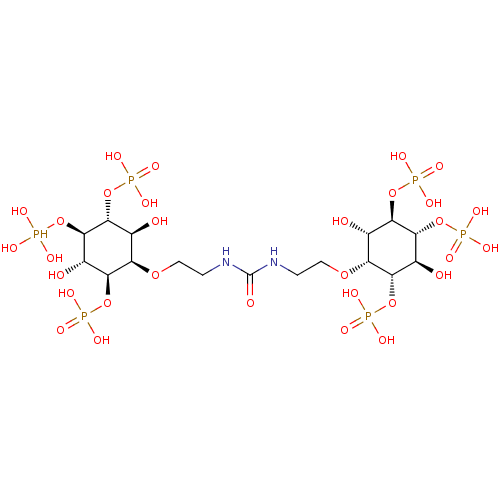 (CHEMBL1213158) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 2.70 | n/a | n/a | n/a | n/a |
University of Cambridge Curated by ChEMBL | Assay Description Agonist activity at rat recombinant IP3R1 expressed in chicken DT40 cells assessed as calcium release from intracellular stores | Nat Chem Biol 5: 631-9 (2009) Article DOI: 10.1038/nchembio.195 BindingDB Entry DOI: 10.7270/Q2C53M27 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Inositol 1,4,5-trisphosphate receptor type 1 (Rattus norvegicus) | BDBM50323705 (CHEMBL1213159) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | n/a | n/a | n/a | n/a | 4.90 | n/a | n/a | n/a | n/a |
University of Cambridge Curated by ChEMBL | Assay Description Agonist activity at rat recombinant IP3R1 expressed in chicken DT40 cells assessed as calcium release from intracellular stores | Nat Chem Biol 5: 631-9 (2009) Article DOI: 10.1038/nchembio.195 BindingDB Entry DOI: 10.7270/Q2C53M27 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Inositol 1,4,5-trisphosphate receptor type 1 (Rattus norvegicus) | BDBM50323706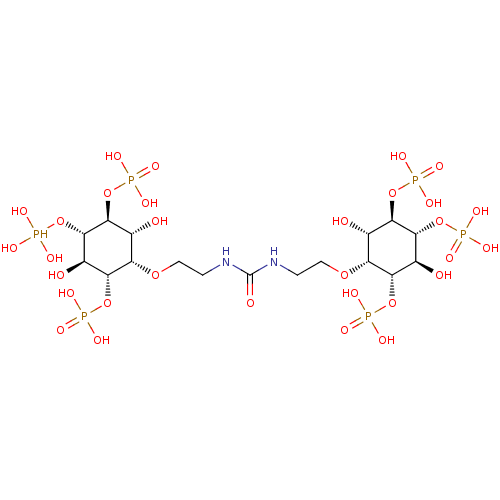 (CHEMBL1212943) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 5 | n/a | n/a | n/a | n/a |
University of Cambridge Curated by ChEMBL | Assay Description Agonist activity at rat recombinant IP3R1 expressed in chicken DT40 cells assessed as calcium release from intracellular stores | Nat Chem Biol 5: 631-9 (2009) Article DOI: 10.1038/nchembio.195 BindingDB Entry DOI: 10.7270/Q2C53M27 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Inositol 1,4,5-trisphosphate receptor type 1 (Rattus norvegicus) | BDBM50323707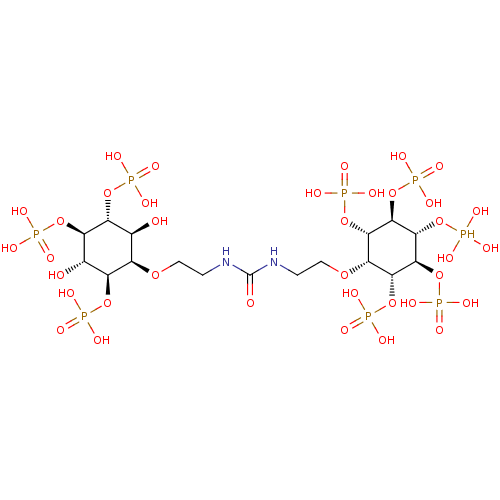 (CHEMBL1213160) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 5 | n/a | n/a | n/a | n/a |
University of Cambridge Curated by ChEMBL | Assay Description Agonist activity at rat recombinant IP3R1 expressed in chicken DT40 cells assessed as calcium release from intracellular stores | Nat Chem Biol 5: 631-9 (2009) Article DOI: 10.1038/nchembio.195 BindingDB Entry DOI: 10.7270/Q2C53M27 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Inositol 1,4,5-trisphosphate receptor type 1 (Rattus norvegicus) | BDBM50323708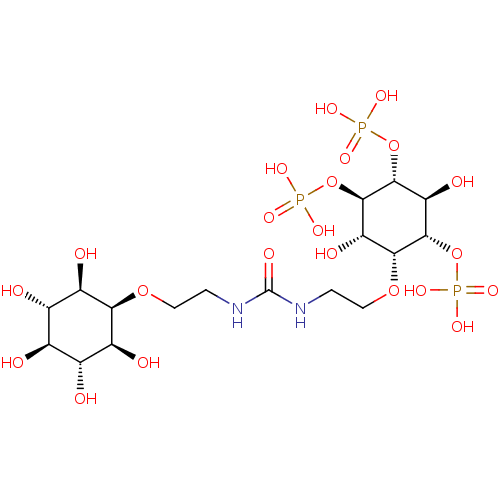 (CHEMBL1213161) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 21 | n/a | n/a | n/a | n/a |
University of Cambridge Curated by ChEMBL | Assay Description Agonist activity at rat recombinant IP3R1 expressed in chicken DT40 cells assessed as calcium release from intracellular stores | Nat Chem Biol 5: 631-9 (2009) Article DOI: 10.1038/nchembio.195 BindingDB Entry DOI: 10.7270/Q2C53M27 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Inositol 1,4,5-trisphosphate receptor type 1 (Rattus norvegicus) | BDBM50323709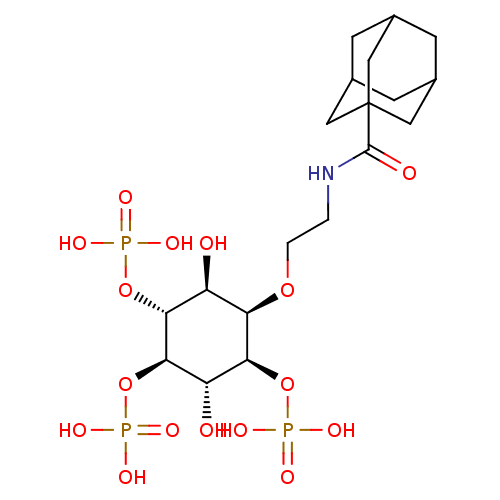 (CHEMBL1213163) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | n/a | n/a | n/a | n/a | 30 | n/a | n/a | n/a | n/a |
University of Cambridge Curated by ChEMBL | Assay Description Agonist activity at rat recombinant IP3R1 expressed in chicken DT40 cells assessed as calcium release from intracellular stores | Nat Chem Biol 5: 631-9 (2009) Article DOI: 10.1038/nchembio.195 BindingDB Entry DOI: 10.7270/Q2C53M27 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Inositol 1,4,5-trisphosphate receptor type 1 (Rattus norvegicus) | BDBM50323710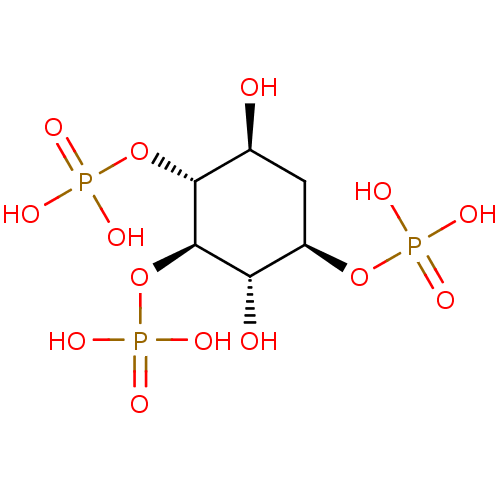 ((1R,2R,3S,4R,6S)-3,6-dihydroxycyclohexane-1,2,4-tr...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 32 | n/a | n/a | n/a | n/a |
University of Cambridge Curated by ChEMBL | Assay Description Agonist activity at rat recombinant IP3R1 expressed in chicken DT40 cells assessed as calcium release from intracellular stores | Nat Chem Biol 5: 631-9 (2009) Article DOI: 10.1038/nchembio.195 BindingDB Entry DOI: 10.7270/Q2C53M27 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Inositol 1,4,5-trisphosphate receptor type 1 (Rattus norvegicus) | BDBM50323711 ((1R,2R,3S,4S,5R,6S)-5-(2-aminoethoxy)-3,6-dihydrox...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 42 | n/a | n/a | n/a | n/a |
University of Cambridge Curated by ChEMBL | Assay Description Agonist activity at rat recombinant IP3R1 expressed in chicken DT40 cells assessed as calcium release from intracellular stores | Nat Chem Biol 5: 631-9 (2009) Article DOI: 10.1038/nchembio.195 BindingDB Entry DOI: 10.7270/Q2C53M27 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Inositol 1,4,5-trisphosphate receptor type 1 (Rattus norvegicus) | BDBM50075183 (1,4,5-Insp3 | 1D-myo-inositol 1,4,5-triphosphate |...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | n/a | n/a | n/a | 12.5 | n/a | n/a | n/a | n/a | n/a |
University of Cambridge Curated by ChEMBL | Assay Description Displacement of [3H]IP3 from full-length rat recombinant IP3R1 expressed in chicken DT40 cells by equilibrium competitive binding assay | Nat Chem Biol 5: 631-9 (2009) Article DOI: 10.1038/nchembio.195 BindingDB Entry DOI: 10.7270/Q2C53M27 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Inositol 1,4,5-trisphosphate receptor type 1 (Rattus norvegicus) | BDBM50184325 (2-[5-(6-amino-9H-9-purinyl)-2-hydroxymethyl-4-oxyp...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL KEGG PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | 1.26 | n/a | n/a | n/a | n/a | n/a |
University of Cambridge Curated by ChEMBL | Assay Description Displacement of [3H]IP3 from full-length rat recombinant IP3R1 expressed in chicken DT40 cells by equilibrium competitive binding assay | Nat Chem Biol 5: 631-9 (2009) Article DOI: 10.1038/nchembio.195 BindingDB Entry DOI: 10.7270/Q2C53M27 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Inositol 1,4,5-trisphosphate receptor type 1 (Rattus norvegicus) | BDBM50323704 (CHEMBL1213158) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | 0.450 | n/a | n/a | n/a | n/a | n/a |
University of Cambridge Curated by ChEMBL | Assay Description Displacement of [3H]IP3 from full-length rat recombinant IP3R1 expressed in chicken DT40 cells by equilibrium competitive binding assay | Nat Chem Biol 5: 631-9 (2009) Article DOI: 10.1038/nchembio.195 BindingDB Entry DOI: 10.7270/Q2C53M27 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Inositol 1,4,5-trisphosphate receptor type 1 (Rattus norvegicus) | BDBM50323705 (CHEMBL1213159) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | n/a | n/a | n/a | 0.860 | n/a | n/a | n/a | n/a | n/a |
University of Cambridge Curated by ChEMBL | Assay Description Displacement of [3H]IP3 from full-length rat recombinant IP3R1 expressed in chicken DT40 cells by equilibrium competitive binding assay | Nat Chem Biol 5: 631-9 (2009) Article DOI: 10.1038/nchembio.195 BindingDB Entry DOI: 10.7270/Q2C53M27 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Inositol 1,4,5-trisphosphate receptor type 1 (Rattus norvegicus) | BDBM50323706 (CHEMBL1212943) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | 0.900 | n/a | n/a | n/a | n/a | n/a |
University of Cambridge Curated by ChEMBL | Assay Description Displacement of [3H]IP3 from full-length rat recombinant IP3R1 expressed in chicken DT40 cells by equilibrium competitive binding assay | Nat Chem Biol 5: 631-9 (2009) Article DOI: 10.1038/nchembio.195 BindingDB Entry DOI: 10.7270/Q2C53M27 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Inositol 1,4,5-trisphosphate receptor type 1 (Rattus norvegicus) | BDBM50323707 (CHEMBL1213160) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | 0.890 | n/a | n/a | n/a | n/a | n/a |
University of Cambridge Curated by ChEMBL | Assay Description Displacement of [3H]IP3 from full-length rat recombinant IP3R1 expressed in chicken DT40 cells by equilibrium competitive binding assay | Nat Chem Biol 5: 631-9 (2009) Article DOI: 10.1038/nchembio.195 BindingDB Entry DOI: 10.7270/Q2C53M27 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Inositol 1,4,5-trisphosphate receptor type 1 (Rattus norvegicus) | BDBM50323708 (CHEMBL1213161) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | 7.82 | n/a | n/a | n/a | n/a | n/a |
University of Cambridge Curated by ChEMBL | Assay Description Displacement of [3H]IP3 from full-length rat recombinant IP3R1 expressed in chicken DT40 cells by equilibrium competitive binding assay | Nat Chem Biol 5: 631-9 (2009) Article DOI: 10.1038/nchembio.195 BindingDB Entry DOI: 10.7270/Q2C53M27 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Inositol 1,4,5-trisphosphate receptor type 1 (Rattus norvegicus) | BDBM50323709 (CHEMBL1213163) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | n/a | n/a | n/a | 7.62 | n/a | n/a | n/a | n/a | n/a |
University of Cambridge Curated by ChEMBL | Assay Description Displacement of [3H]IP3 from full-length rat recombinant IP3R1 expressed in chicken DT40 cells by equilibrium competitive binding assay | Nat Chem Biol 5: 631-9 (2009) Article DOI: 10.1038/nchembio.195 BindingDB Entry DOI: 10.7270/Q2C53M27 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Inositol 1,4,5-trisphosphate receptor type 1 (Rattus norvegicus) | BDBM50323710 ((1R,2R,3S,4R,6S)-3,6-dihydroxycyclohexane-1,2,4-tr...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | 17.1 | n/a | n/a | n/a | n/a | n/a |
University of Cambridge Curated by ChEMBL | Assay Description Displacement of [3H]IP3 from full-length rat recombinant IP3R1 expressed in chicken DT40 cells by equilibrium competitive binding assay | Nat Chem Biol 5: 631-9 (2009) Article DOI: 10.1038/nchembio.195 BindingDB Entry DOI: 10.7270/Q2C53M27 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Inositol 1,4,5-trisphosphate receptor type 1 (Rattus norvegicus) | BDBM50323711 ((1R,2R,3S,4S,5R,6S)-5-(2-aminoethoxy)-3,6-dihydrox...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | 43.6 | n/a | n/a | n/a | n/a | n/a |
University of Cambridge Curated by ChEMBL | Assay Description Displacement of [3H]IP3 from full-length rat recombinant IP3R1 expressed in chicken DT40 cells by equilibrium competitive binding assay | Nat Chem Biol 5: 631-9 (2009) Article DOI: 10.1038/nchembio.195 BindingDB Entry DOI: 10.7270/Q2C53M27 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Inositol 1,4,5-trisphosphate receptor type 1 (Rattus norvegicus) | BDBM50075183 (1,4,5-Insp3 | 1D-myo-inositol 1,4,5-triphosphate |...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | n/a | n/a | n/a | 2.32 | n/a | n/a | n/a | n/a | n/a |
University of Cambridge Curated by ChEMBL | Assay Description Displacement of [3H]IP3 from rat recombinant N-terminal IP3R1 (1-604) expressed in chicken DT40 cells by equilibrium competitive binding assay | Nat Chem Biol 5: 631-9 (2009) Article DOI: 10.1038/nchembio.195 BindingDB Entry DOI: 10.7270/Q2C53M27 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Inositol 1,4,5-trisphosphate receptor type 1 (Rattus norvegicus) | BDBM50323704 (CHEMBL1213158) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | 0.410 | n/a | n/a | n/a | n/a | n/a |
University of Cambridge Curated by ChEMBL | Assay Description Displacement of [3H]IP3 from rat recombinant N-terminal IP3R1 (1-604) expressed in chicken DT40 cells by equilibrium competitive binding assay | Nat Chem Biol 5: 631-9 (2009) Article DOI: 10.1038/nchembio.195 BindingDB Entry DOI: 10.7270/Q2C53M27 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Inositol 1,4,5-trisphosphate receptor type 1 (Rattus norvegicus) | BDBM50323705 (CHEMBL1213159) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | n/a | n/a | n/a | 0.470 | n/a | n/a | n/a | n/a | n/a |
University of Cambridge Curated by ChEMBL | Assay Description Displacement of [3H]IP3 from rat recombinant N-terminal IP3R1 (1-604) expressed in chicken DT40 cells by equilibrium competitive binding assay | Nat Chem Biol 5: 631-9 (2009) Article DOI: 10.1038/nchembio.195 BindingDB Entry DOI: 10.7270/Q2C53M27 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Inositol 1,4,5-trisphosphate receptor type 1 (Rattus norvegicus) | BDBM50323706 (CHEMBL1212943) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | 0.420 | n/a | n/a | n/a | n/a | n/a |
University of Cambridge Curated by ChEMBL | Assay Description Displacement of [3H]IP3 from rat recombinant N-terminal IP3R1 (1-604) expressed in chicken DT40 cells by equilibrium competitive binding assay | Nat Chem Biol 5: 631-9 (2009) Article DOI: 10.1038/nchembio.195 BindingDB Entry DOI: 10.7270/Q2C53M27 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Inositol 1,4,5-trisphosphate receptor type 1 (Rattus norvegicus) | BDBM50323707 (CHEMBL1213160) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | 0.360 | n/a | n/a | n/a | n/a | n/a |
University of Cambridge Curated by ChEMBL | Assay Description Displacement of [3H]IP3 from rat recombinant N-terminal IP3R1 (1-604) expressed in chicken DT40 cells by equilibrium competitive binding assay | Nat Chem Biol 5: 631-9 (2009) Article DOI: 10.1038/nchembio.195 BindingDB Entry DOI: 10.7270/Q2C53M27 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Inositol 1,4,5-trisphosphate receptor type 1 (Rattus norvegicus) | BDBM50323708 (CHEMBL1213161) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | 2.81 | n/a | n/a | n/a | n/a | n/a |
University of Cambridge Curated by ChEMBL | Assay Description Displacement of [3H]IP3 from rat recombinant N-terminal IP3R1 (1-604) expressed in chicken DT40 cells by equilibrium competitive binding assay | Nat Chem Biol 5: 631-9 (2009) Article DOI: 10.1038/nchembio.195 BindingDB Entry DOI: 10.7270/Q2C53M27 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Inositol 1,4,5-trisphosphate receptor type 1 (Rattus norvegicus) | BDBM50323709 (CHEMBL1213163) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | n/a | n/a | n/a | 1.63 | n/a | n/a | n/a | n/a | n/a |
University of Cambridge Curated by ChEMBL | Assay Description Displacement of [3H]IP3 from rat recombinant N-terminal IP3R1 (1-604) expressed in chicken DT40 cells by equilibrium competitive binding assay | Nat Chem Biol 5: 631-9 (2009) Article DOI: 10.1038/nchembio.195 BindingDB Entry DOI: 10.7270/Q2C53M27 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Inositol 1,4,5-trisphosphate receptor type 1 (Rattus norvegicus) | BDBM50323710 ((1R,2R,3S,4R,6S)-3,6-dihydroxycyclohexane-1,2,4-tr...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | 4.01 | n/a | n/a | n/a | n/a | n/a |
University of Cambridge Curated by ChEMBL | Assay Description Displacement of [3H]IP3 from rat recombinant N-terminal IP3R1 (1-604) expressed in chicken DT40 cells by equilibrium competitive binding assay | Nat Chem Biol 5: 631-9 (2009) Article DOI: 10.1038/nchembio.195 BindingDB Entry DOI: 10.7270/Q2C53M27 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Inositol 1,4,5-trisphosphate receptor type 1 (Rattus norvegicus) | BDBM50323711 ((1R,2R,3S,4S,5R,6S)-5-(2-aminoethoxy)-3,6-dihydrox...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | 13.5 | n/a | n/a | n/a | n/a | n/a |
University of Cambridge Curated by ChEMBL | Assay Description Displacement of [3H]IP3 from rat recombinant N-terminal IP3R1 (1-604) expressed in chicken DT40 cells by equilibrium competitive binding assay | Nat Chem Biol 5: 631-9 (2009) Article DOI: 10.1038/nchembio.195 BindingDB Entry DOI: 10.7270/Q2C53M27 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Inositol 1,4,5-trisphosphate receptor type 1 (Rattus norvegicus) | BDBM50075183 (1,4,5-Insp3 | 1D-myo-inositol 1,4,5-triphosphate |...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | n/a | n/a | n/a | 0.210 | n/a | n/a | n/a | n/a | n/a |
University of Cambridge Curated by ChEMBL | Assay Description Displacement of [3H]IP3 from rat recombinant IP3R1 binding core residue (224-604) expressed in chicken DT40 cells by equilibrium competitive binding ... | Nat Chem Biol 5: 631-9 (2009) Article DOI: 10.1038/nchembio.195 BindingDB Entry DOI: 10.7270/Q2C53M27 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Displayed 1 to 50 (of 122 total ) | Next | Last >> |