 Found 37 hits Enz. Inhib. hit(s) with all data for entry = 50010563
Found 37 hits Enz. Inhib. hit(s) with all data for entry = 50010563 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Beta-secretase 2
(Homo sapiens (Human)) | BDBM50540172
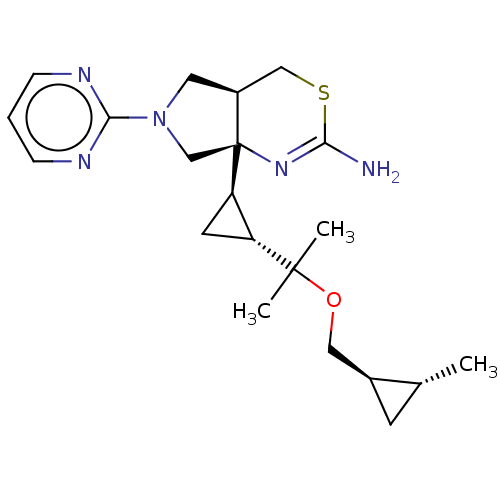
(CHEMBL4637426)Show SMILES [H][C@]1(C[C@H]1C(C)(C)OC[C@@H]1C[C@H]1C)[C@]12CN(C[C@@]1([H])CSC(N)=N2)c1ncccn1 |r,c:25| Show InChI InChI=1S/C21H31N5OS/c1-13-7-14(13)10-27-20(2,3)16-8-17(16)21-12-26(19-23-5-4-6-24-19)9-15(21)11-28-18(22)25-21/h4-6,13-17H,7-12H2,1-3H3,(H2,22,25)/t13-,14+,15+,16-,17-,21+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | 5.30 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Inhibition of human BACE2 |
Bioorg Med Chem 28: (2020)
Article DOI: 10.1016/j.bmc.2019.115194
BindingDB Entry DOI: 10.7270/Q2B56P8K |
More data for this
Ligand-Target Pair | |
Beta-secretase 2
(Homo sapiens (Human)) | BDBM50012647
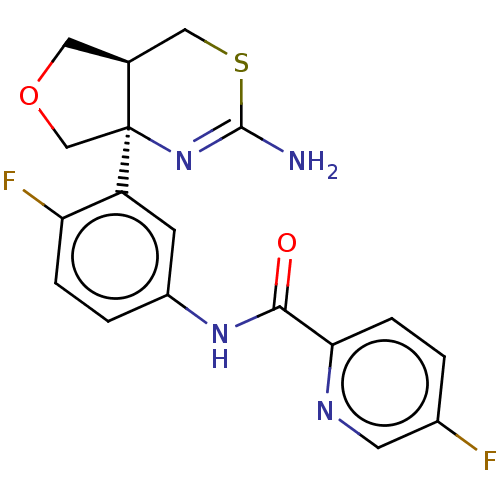
(CHEMBL2396989)Show SMILES [H][C@@]12COC[C@@]1(N=C(N)SC2)c1cc(NC(=O)c2ccc(F)cn2)ccc1F |r,t:7| Show InChI InChI=1S/C18H16F2N4O2S/c19-11-1-4-15(22-6-11)16(25)23-12-2-3-14(20)13(5-12)18-9-26-7-10(18)8-27-17(21)24-18/h1-6,10H,7-9H2,(H2,21,24)(H,23,25)/t10-,18-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Inhibition of human BACE2 |
Bioorg Med Chem 28: (2020)
Article DOI: 10.1016/j.bmc.2019.115194
BindingDB Entry DOI: 10.7270/Q2B56P8K |
More data for this
Ligand-Target Pair | |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM50540172

(CHEMBL4637426)Show SMILES [H][C@]1(C[C@H]1C(C)(C)OC[C@@H]1C[C@H]1C)[C@]12CN(C[C@@]1([H])CSC(N)=N2)c1ncccn1 |r,c:25| Show InChI InChI=1S/C21H31N5OS/c1-13-7-14(13)10-27-20(2,3)16-8-17(16)21-12-26(19-23-5-4-6-24-19)9-15(21)11-28-18(22)25-21/h4-6,13-17H,7-12H2,1-3H3,(H2,22,25)/t13-,14+,15+,16-,17-,21+/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Inhibition of human BACE1 (1 to 460 residues) expressed in HEK293 cells using mcaFRET peptide as substrate after 20 hrs by FRET assay |
Bioorg Med Chem 28: (2020)
Article DOI: 10.1016/j.bmc.2019.115194
BindingDB Entry DOI: 10.7270/Q2B56P8K |
More data for this
Ligand-Target Pair | |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM50540171
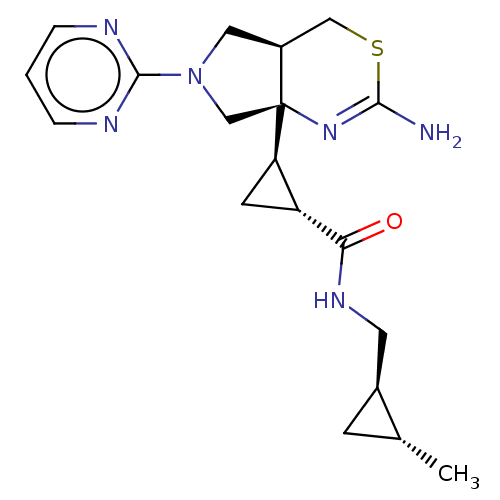
(CHEMBL4643727)Show SMILES [H][C@]1(C[C@H]1C(=O)NC[C@@H]1C[C@H]1C)[C@]12CN(C[C@@]1([H])CSC(N)=N2)c1ncccn1 |r,c:24| Show InChI InChI=1S/C19H26N6OS/c1-11-5-12(11)7-23-16(26)14-6-15(14)19-10-25(18-21-3-2-4-22-18)8-13(19)9-27-17(20)24-19/h2-4,11-15H,5-10H2,1H3,(H2,20,24)(H,23,26)/t11-,12+,13+,14-,15-,19+/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | 18 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Inhibition of BACE1 (unknown origin) by cell based assay |
Bioorg Med Chem 28: (2020)
Article DOI: 10.1016/j.bmc.2019.115194
BindingDB Entry DOI: 10.7270/Q2B56P8K |
More data for this
Ligand-Target Pair | |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM50012647

(CHEMBL2396989)Show SMILES [H][C@@]12COC[C@@]1(N=C(N)SC2)c1cc(NC(=O)c2ccc(F)cn2)ccc1F |r,t:7| Show InChI InChI=1S/C18H16F2N4O2S/c19-11-1-4-15(22-6-11)16(25)23-12-2-3-14(20)13(5-12)18-9-26-7-10(18)8-27-17(21)24-18/h1-6,10H,7-9H2,(H2,21,24)(H,23,25)/t10-,18-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 20 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Inhibition of human BACE1 (1 to 460 residues) expressed in HEK293 cells using mcaFRET peptide as substrate after 20 hrs by FRET assay |
Bioorg Med Chem 28: (2020)
Article DOI: 10.1016/j.bmc.2019.115194
BindingDB Entry DOI: 10.7270/Q2B56P8K |
More data for this
Ligand-Target Pair | |
Beta-secretase 2
(Homo sapiens (Human)) | BDBM50540171

(CHEMBL4643727)Show SMILES [H][C@]1(C[C@H]1C(=O)NC[C@@H]1C[C@H]1C)[C@]12CN(C[C@@]1([H])CSC(N)=N2)c1ncccn1 |r,c:24| Show InChI InChI=1S/C19H26N6OS/c1-11-5-12(11)7-23-16(26)14-6-15(14)19-10-25(18-21-3-2-4-22-18)8-13(19)9-27-17(20)24-19/h2-4,11-15H,5-10H2,1H3,(H2,20,24)(H,23,26)/t11-,12+,13+,14-,15-,19+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | 22 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Inhibition of human BACE2 |
Bioorg Med Chem 28: (2020)
Article DOI: 10.1016/j.bmc.2019.115194
BindingDB Entry DOI: 10.7270/Q2B56P8K |
More data for this
Ligand-Target Pair | |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM50540171

(CHEMBL4643727)Show SMILES [H][C@]1(C[C@H]1C(=O)NC[C@@H]1C[C@H]1C)[C@]12CN(C[C@@]1([H])CSC(N)=N2)c1ncccn1 |r,c:24| Show InChI InChI=1S/C19H26N6OS/c1-11-5-12(11)7-23-16(26)14-6-15(14)19-10-25(18-21-3-2-4-22-18)8-13(19)9-27-17(20)24-19/h2-4,11-15H,5-10H2,1H3,(H2,20,24)(H,23,26)/t11-,12+,13+,14-,15-,19+/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | 28 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Inhibition of human BACE1 (1 to 460 residues) expressed in HEK293 cells using mcaFRET peptide as substrate after 20 hrs by FRET assay |
Bioorg Med Chem 28: (2020)
Article DOI: 10.1016/j.bmc.2019.115194
BindingDB Entry DOI: 10.7270/Q2B56P8K |
More data for this
Ligand-Target Pair | |
Beta-secretase 2
(Homo sapiens (Human)) | BDBM50540170
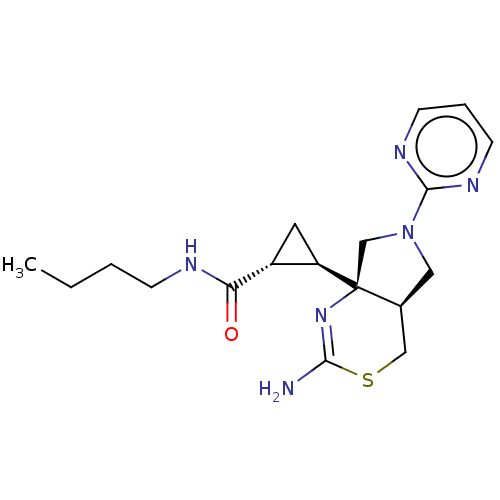
(CHEMBL4636286)Show SMILES [H][C@]1(C[C@H]1C(=O)NCCCC)[C@]12CN(C[C@@]1([H])CSC(N)=N2)c1ncccn1 |r,c:22| Show InChI InChI=1S/C18H26N6OS/c1-2-3-5-20-15(25)13-8-14(13)18-11-24(17-21-6-4-7-22-17)9-12(18)10-26-16(19)23-18/h4,6-7,12-14H,2-3,5,8-11H2,1H3,(H2,19,23)(H,20,25)/t12-,13+,14+,18-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 44 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Inhibition of human BACE2 |
Bioorg Med Chem 28: (2020)
Article DOI: 10.1016/j.bmc.2019.115194
BindingDB Entry DOI: 10.7270/Q2B56P8K |
More data for this
Ligand-Target Pair | |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM50540170

(CHEMBL4636286)Show SMILES [H][C@]1(C[C@H]1C(=O)NCCCC)[C@]12CN(C[C@@]1([H])CSC(N)=N2)c1ncccn1 |r,c:22| Show InChI InChI=1S/C18H26N6OS/c1-2-3-5-20-15(25)13-8-14(13)18-11-24(17-21-6-4-7-22-17)9-12(18)10-26-16(19)23-18/h4,6-7,12-14H,2-3,5,8-11H2,1H3,(H2,19,23)(H,20,25)/t12-,13+,14+,18-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 67 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Inhibition of human BACE1 (1 to 460 residues) expressed in HEK293 cells using mcaFRET peptide as substrate after 20 hrs by FRET assay |
Bioorg Med Chem 28: (2020)
Article DOI: 10.1016/j.bmc.2019.115194
BindingDB Entry DOI: 10.7270/Q2B56P8K |
More data for this
Ligand-Target Pair | |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM50012632
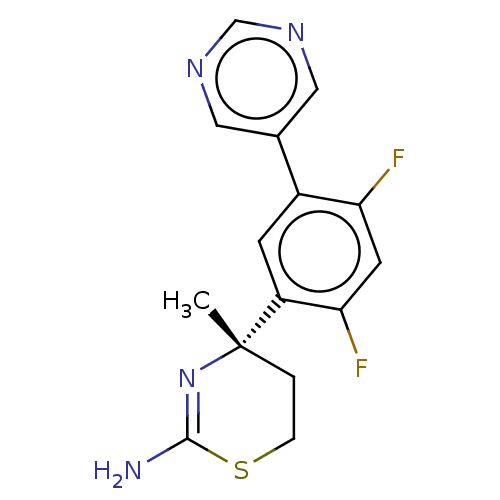
(CHEMBL2333941)Show SMILES C[C@]1(CCSC(N)=N1)c1cc(c(F)cc1F)-c1cncnc1 |r,c:6| Show InChI InChI=1S/C15H14F2N4S/c1-15(2-3-22-14(18)21-15)11-4-10(12(16)5-13(11)17)9-6-19-8-20-7-9/h4-8H,2-3H2,1H3,(H2,18,21)/t15-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 239 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Inhibition of human BACE1 (1 to 460 residues) expressed in HEK293 cells using mcaFRET peptide as substrate after 20 hrs by FRET assay |
Bioorg Med Chem 28: (2020)
Article DOI: 10.1016/j.bmc.2019.115194
BindingDB Entry DOI: 10.7270/Q2B56P8K |
More data for this
Ligand-Target Pair | |
Beta-secretase 2
(Homo sapiens (Human)) | BDBM50012632

(CHEMBL2333941)Show SMILES C[C@]1(CCSC(N)=N1)c1cc(c(F)cc1F)-c1cncnc1 |r,c:6| Show InChI InChI=1S/C15H14F2N4S/c1-15(2-3-22-14(18)21-15)11-4-10(12(16)5-13(11)17)9-6-19-8-20-7-9/h4-8H,2-3H2,1H3,(H2,18,21)/t15-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 288 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Inhibition of human BACE2 |
Bioorg Med Chem 28: (2020)
Article DOI: 10.1016/j.bmc.2019.115194
BindingDB Entry DOI: 10.7270/Q2B56P8K |
More data for this
Ligand-Target Pair | |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM50540168
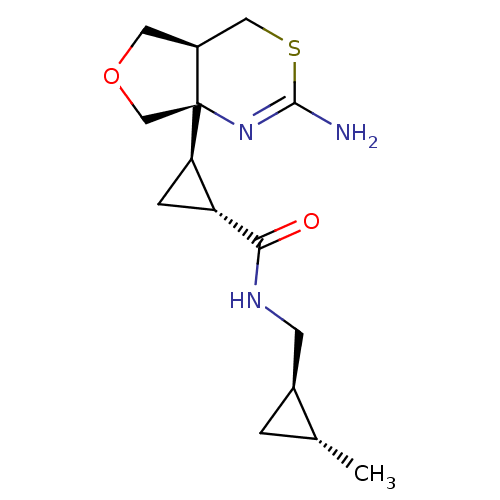
(CHEMBL4644249)Show SMILES [H][C@]1(C[C@H]1C(=O)NC[C@@H]1C[C@H]1C)[C@]12COC[C@@]1([H])CSC(N)=N2 |r,c:24| Show InChI InChI=1S/C15H23N3O2S/c1-8-2-9(8)4-17-13(19)11-3-12(11)15-7-20-5-10(15)6-21-14(16)18-15/h8-12H,2-7H2,1H3,(H2,16,18)(H,17,19)/t8-,9+,10+,11-,12-,15+/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | 990 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Inhibition of human BACE1 (1 to 460 residues) expressed in HEK293 cells using mcaFRET peptide as substrate after 20 hrs by FRET assay |
Bioorg Med Chem 28: (2020)
Article DOI: 10.1016/j.bmc.2019.115194
BindingDB Entry DOI: 10.7270/Q2B56P8K |
More data for this
Ligand-Target Pair | |
Beta-secretase 2
(Homo sapiens (Human)) | BDBM50540168

(CHEMBL4644249)Show SMILES [H][C@]1(C[C@H]1C(=O)NC[C@@H]1C[C@H]1C)[C@]12COC[C@@]1([H])CSC(N)=N2 |r,c:24| Show InChI InChI=1S/C15H23N3O2S/c1-8-2-9(8)4-17-13(19)11-3-12(11)15-7-20-5-10(15)6-21-14(16)18-15/h8-12H,2-7H2,1H3,(H2,16,18)(H,17,19)/t8-,9+,10+,11-,12-,15+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | 1.58E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Inhibition of human BACE2 |
Bioorg Med Chem 28: (2020)
Article DOI: 10.1016/j.bmc.2019.115194
BindingDB Entry DOI: 10.7270/Q2B56P8K |
More data for this
Ligand-Target Pair | |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM50540167

(CHEMBL4642463)Show SMILES [H][C@]1(C[C@H]1C(=O)NCCCC)[C@]12COC[C@@]1([H])CSC(N)=N2 |r,c:22| Show InChI InChI=1S/C14H23N3O2S/c1-2-3-4-16-12(18)10-5-11(10)14-8-19-6-9(14)7-20-13(15)17-14/h9-11H,2-8H2,1H3,(H2,15,17)(H,16,18)/t9-,10+,11+,14-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | 6.64E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Inhibition of human BACE1 (1 to 460 residues) expressed in HEK293 cells using mcaFRET peptide as substrate after 20 hrs by FRET assay |
Bioorg Med Chem 28: (2020)
Article DOI: 10.1016/j.bmc.2019.115194
BindingDB Entry DOI: 10.7270/Q2B56P8K |
More data for this
Ligand-Target Pair | |
Beta-secretase 2
(Homo sapiens (Human)) | BDBM50540167

(CHEMBL4642463)Show SMILES [H][C@]1(C[C@H]1C(=O)NCCCC)[C@]12COC[C@@]1([H])CSC(N)=N2 |r,c:22| Show InChI InChI=1S/C14H23N3O2S/c1-2-3-4-16-12(18)10-5-11(10)14-8-19-6-9(14)7-20-13(15)17-14/h9-11H,2-8H2,1H3,(H2,15,17)(H,16,18)/t9-,10+,11+,14-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | 7.33E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Inhibition of human BACE2 |
Bioorg Med Chem 28: (2020)
Article DOI: 10.1016/j.bmc.2019.115194
BindingDB Entry DOI: 10.7270/Q2B56P8K |
More data for this
Ligand-Target Pair | |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM50540166

(CHEMBL4636311)Show SMILES [H][C@]1(C[C@H]1C(=O)NCCC)[C@]12COC[C@@]1([H])CSC(N)=N2 |r,c:21| Show InChI InChI=1S/C13H21N3O2S/c1-2-3-15-11(17)9-4-10(9)13-7-18-5-8(13)6-19-12(14)16-13/h8-10H,2-7H2,1H3,(H2,14,16)(H,15,17)/t8-,9+,10+,13-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.40E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Inhibition of human BACE1 (1 to 460 residues) expressed in HEK293 cells using mcaFRET peptide as substrate after 20 hrs by FRET assay |
Bioorg Med Chem 28: (2020)
Article DOI: 10.1016/j.bmc.2019.115194
BindingDB Entry DOI: 10.7270/Q2B56P8K |
More data for this
Ligand-Target Pair | |
Cathepsin D
(Homo sapiens (Human)) | BDBM50012632

(CHEMBL2333941)Show SMILES C[C@]1(CCSC(N)=N1)c1cc(c(F)cc1F)-c1cncnc1 |r,c:6| Show InChI InChI=1S/C15H14F2N4S/c1-15(2-3-22-14(18)21-15)11-4-10(12(16)5-13(11)17)9-6-19-8-20-7-9/h4-8H,2-3H2,1H3,(H2,18,21)/t15-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.57E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Inhibition of human cathepsin D |
Bioorg Med Chem 28: (2020)
Article DOI: 10.1016/j.bmc.2019.115194
BindingDB Entry DOI: 10.7270/Q2B56P8K |
More data for this
Ligand-Target Pair | |
Beta-secretase 2
(Homo sapiens (Human)) | BDBM50540166

(CHEMBL4636311)Show SMILES [H][C@]1(C[C@H]1C(=O)NCCC)[C@]12COC[C@@]1([H])CSC(N)=N2 |r,c:21| Show InChI InChI=1S/C13H21N3O2S/c1-2-3-15-11(17)9-4-10(9)13-7-18-5-8(13)6-19-12(14)16-13/h8-10H,2-7H2,1H3,(H2,14,16)(H,15,17)/t8-,9+,10+,13-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 2.46E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Inhibition of human BACE2 |
Bioorg Med Chem 28: (2020)
Article DOI: 10.1016/j.bmc.2019.115194
BindingDB Entry DOI: 10.7270/Q2B56P8K |
More data for this
Ligand-Target Pair | |
Cathepsin D
(Homo sapiens (Human)) | BDBM50540170

(CHEMBL4636286)Show SMILES [H][C@]1(C[C@H]1C(=O)NCCCC)[C@]12CN(C[C@@]1([H])CSC(N)=N2)c1ncccn1 |r,c:22| Show InChI InChI=1S/C18H26N6OS/c1-2-3-5-20-15(25)13-8-14(13)18-11-24(17-21-6-4-7-22-17)9-12(18)10-26-16(19)23-18/h4,6-7,12-14H,2-3,5,8-11H2,1H3,(H2,19,23)(H,20,25)/t12-,13+,14+,18-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 5.70E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Inhibition of human cathepsin D |
Bioorg Med Chem 28: (2020)
Article DOI: 10.1016/j.bmc.2019.115194
BindingDB Entry DOI: 10.7270/Q2B56P8K |
More data for this
Ligand-Target Pair | |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM50540165

(CHEMBL4637913)Show SMILES [H][C@]1(C[C@H]1C(=O)NCC)[C@]12COC[C@@]1([H])CSC(N)=N2 |r,c:20| Show InChI InChI=1S/C12H19N3O2S/c1-2-14-10(16)8-3-9(8)12-6-17-4-7(12)5-18-11(13)15-12/h7-9H,2-6H2,1H3,(H2,13,15)(H,14,16)/t7-,8+,9+,12-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 5.80E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Inhibition of human BACE1 (1 to 460 residues) expressed in HEK293 cells using mcaFRET peptide as substrate after 20 hrs by FRET assay |
Bioorg Med Chem 28: (2020)
Article DOI: 10.1016/j.bmc.2019.115194
BindingDB Entry DOI: 10.7270/Q2B56P8K |
More data for this
Ligand-Target Pair | |
Beta-secretase 2
(Homo sapiens (Human)) | BDBM50540164

(CHEMBL4640948)Show SMILES [H][C@]1(C[C@H]1C(=O)Nc1ccc(F)cn1)[C@]12COC[C@@]1([H])CSC(N)=N2 |r,c:26| Show InChI InChI=1S/C15H17FN4O2S/c16-9-1-2-12(18-4-9)19-13(21)10-3-11(10)15-7-22-5-8(15)6-23-14(17)20-15/h1-2,4,8,10-11H,3,5-7H2,(H2,17,20)(H,18,19,21)/t8-,10+,11+,15-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 7.36E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Inhibition of human BACE2 |
Bioorg Med Chem 28: (2020)
Article DOI: 10.1016/j.bmc.2019.115194
BindingDB Entry DOI: 10.7270/Q2B56P8K |
More data for this
Ligand-Target Pair | |
Cathepsin D
(Homo sapiens (Human)) | BDBM50540171

(CHEMBL4643727)Show SMILES [H][C@]1(C[C@H]1C(=O)NC[C@@H]1C[C@H]1C)[C@]12CN(C[C@@]1([H])CSC(N)=N2)c1ncccn1 |r,c:24| Show InChI InChI=1S/C19H26N6OS/c1-11-5-12(11)7-23-16(26)14-6-15(14)19-10-25(18-21-3-2-4-22-18)8-13(19)9-27-17(20)24-19/h2-4,11-15H,5-10H2,1H3,(H2,20,24)(H,23,26)/t11-,12+,13+,14-,15-,19+/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | 9.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Inhibition of human cathepsin D |
Bioorg Med Chem 28: (2020)
Article DOI: 10.1016/j.bmc.2019.115194
BindingDB Entry DOI: 10.7270/Q2B56P8K |
More data for this
Ligand-Target Pair | |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM50540164

(CHEMBL4640948)Show SMILES [H][C@]1(C[C@H]1C(=O)Nc1ccc(F)cn1)[C@]12COC[C@@]1([H])CSC(N)=N2 |r,c:26| Show InChI InChI=1S/C15H17FN4O2S/c16-9-1-2-12(18-4-9)19-13(21)10-3-11(10)15-7-22-5-8(15)6-23-14(17)20-15/h1-2,4,8,10-11H,3,5-7H2,(H2,17,20)(H,18,19,21)/t8-,10+,11+,15-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 9.24E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Inhibition of human BACE1 (1 to 460 residues) expressed in HEK293 cells using mcaFRET peptide as substrate after 20 hrs by FRET assay |
Bioorg Med Chem 28: (2020)
Article DOI: 10.1016/j.bmc.2019.115194
BindingDB Entry DOI: 10.7270/Q2B56P8K |
More data for this
Ligand-Target Pair | |
Beta-secretase 2
(Homo sapiens (Human)) | BDBM50540169

(CHEMBL4648644)Show SMILES [H][C@]1(C[C@H]1NC(=O)CCCC)[C@]12COC[C@@]1([H])CSC(N)=N2 |r,c:22| Show InChI InChI=1S/C14H23N3O2S/c1-2-3-4-12(18)16-11-5-10(11)14-8-19-6-9(14)7-20-13(15)17-14/h9-11H,2-8H2,1H3,(H2,15,17)(H,16,18)/t9-,10+,11+,14-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Inhibition of human BACE2 |
Bioorg Med Chem 28: (2020)
Article DOI: 10.1016/j.bmc.2019.115194
BindingDB Entry DOI: 10.7270/Q2B56P8K |
More data for this
Ligand-Target Pair | |
Cathepsin D
(Homo sapiens (Human)) | BDBM50540169

(CHEMBL4648644)Show SMILES [H][C@]1(C[C@H]1NC(=O)CCCC)[C@]12COC[C@@]1([H])CSC(N)=N2 |r,c:22| Show InChI InChI=1S/C14H23N3O2S/c1-2-3-4-12(18)16-11-5-10(11)14-8-19-6-9(14)7-20-13(15)17-14/h9-11H,2-8H2,1H3,(H2,15,17)(H,16,18)/t9-,10+,11+,14-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Inhibition of human cathepsin D |
Bioorg Med Chem 28: (2020)
Article DOI: 10.1016/j.bmc.2019.115194
BindingDB Entry DOI: 10.7270/Q2B56P8K |
More data for this
Ligand-Target Pair | |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM50540163

(CHEMBL4637568)Show SMILES [H][C@@]1(C[C@@H]1NC(=O)c1ccc(F)cn1)[C@]12COC[C@@]1([H])CSC(N)=N2 |r,c:26| Show InChI InChI=1S/C15H17FN4O2S/c16-9-1-2-11(18-4-9)13(21)19-12-3-10(12)15-7-22-5-8(15)6-23-14(17)20-15/h1-2,4,8,10,12H,3,5-7H2,(H2,17,20)(H,19,21)/t8-,10-,12-,15-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Inhibition of human BACE1 (1 to 460 residues) expressed in HEK293 cells using mcaFRET peptide as substrate after 20 hrs by FRET assay |
Bioorg Med Chem 28: (2020)
Article DOI: 10.1016/j.bmc.2019.115194
BindingDB Entry DOI: 10.7270/Q2B56P8K |
More data for this
Ligand-Target Pair | |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM50540169

(CHEMBL4648644)Show SMILES [H][C@]1(C[C@H]1NC(=O)CCCC)[C@]12COC[C@@]1([H])CSC(N)=N2 |r,c:22| Show InChI InChI=1S/C14H23N3O2S/c1-2-3-4-12(18)16-11-5-10(11)14-8-19-6-9(14)7-20-13(15)17-14/h9-11H,2-8H2,1H3,(H2,15,17)(H,16,18)/t9-,10+,11+,14-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Inhibition of human BACE1 (1 to 460 residues) expressed in HEK293 cells using mcaFRET peptide as substrate after 20 hrs by FRET assay |
Bioorg Med Chem 28: (2020)
Article DOI: 10.1016/j.bmc.2019.115194
BindingDB Entry DOI: 10.7270/Q2B56P8K |
More data for this
Ligand-Target Pair | |
Beta-secretase 2
(Homo sapiens (Human)) | BDBM50540163

(CHEMBL4637568)Show SMILES [H][C@@]1(C[C@@H]1NC(=O)c1ccc(F)cn1)[C@]12COC[C@@]1([H])CSC(N)=N2 |r,c:26| Show InChI InChI=1S/C15H17FN4O2S/c16-9-1-2-11(18-4-9)13(21)19-12-3-10(12)15-7-22-5-8(15)6-23-14(17)20-15/h1-2,4,8,10,12H,3,5-7H2,(H2,17,20)(H,19,21)/t8-,10-,12-,15-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Inhibition of human BACE2 |
Bioorg Med Chem 28: (2020)
Article DOI: 10.1016/j.bmc.2019.115194
BindingDB Entry DOI: 10.7270/Q2B56P8K |
More data for this
Ligand-Target Pair | |
Beta-secretase 2
(Homo sapiens (Human)) | BDBM50540165

(CHEMBL4637913)Show SMILES [H][C@]1(C[C@H]1C(=O)NCC)[C@]12COC[C@@]1([H])CSC(N)=N2 |r,c:20| Show InChI InChI=1S/C12H19N3O2S/c1-2-14-10(16)8-3-9(8)12-6-17-4-7(12)5-18-11(13)15-12/h7-9H,2-6H2,1H3,(H2,13,15)(H,14,16)/t7-,8+,9+,12-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.04E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Inhibition of human BACE2 |
Bioorg Med Chem 28: (2020)
Article DOI: 10.1016/j.bmc.2019.115194
BindingDB Entry DOI: 10.7270/Q2B56P8K |
More data for this
Ligand-Target Pair | |
Cathepsin D
(Homo sapiens (Human)) | BDBM50540172

(CHEMBL4637426)Show SMILES [H][C@]1(C[C@H]1C(C)(C)OC[C@@H]1C[C@H]1C)[C@]12CN(C[C@@]1([H])CSC(N)=N2)c1ncccn1 |r,c:25| Show InChI InChI=1S/C21H31N5OS/c1-13-7-14(13)10-27-20(2,3)16-8-17(16)21-12-26(19-23-5-4-6-24-19)9-15(21)11-28-18(22)25-21/h4-6,13-17H,7-12H2,1-3H3,(H2,22,25)/t13-,14+,15+,16-,17-,21+/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | 1.60E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Inhibition of human cathepsin D |
Bioorg Med Chem 28: (2020)
Article DOI: 10.1016/j.bmc.2019.115194
BindingDB Entry DOI: 10.7270/Q2B56P8K |
More data for this
Ligand-Target Pair | |
Cathepsin D
(Homo sapiens (Human)) | BDBM50540167

(CHEMBL4642463)Show SMILES [H][C@]1(C[C@H]1C(=O)NCCCC)[C@]12COC[C@@]1([H])CSC(N)=N2 |r,c:22| Show InChI InChI=1S/C14H23N3O2S/c1-2-3-4-16-12(18)10-5-11(10)14-8-19-6-9(14)7-20-13(15)17-14/h9-11H,2-8H2,1H3,(H2,15,17)(H,16,18)/t9-,10+,11+,14-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | >3.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Inhibition of human cathepsin D |
Bioorg Med Chem 28: (2020)
Article DOI: 10.1016/j.bmc.2019.115194
BindingDB Entry DOI: 10.7270/Q2B56P8K |
More data for this
Ligand-Target Pair | |
Cathepsin D
(Homo sapiens (Human)) | BDBM50540168

(CHEMBL4644249)Show SMILES [H][C@]1(C[C@H]1C(=O)NC[C@@H]1C[C@H]1C)[C@]12COC[C@@]1([H])CSC(N)=N2 |r,c:24| Show InChI InChI=1S/C15H23N3O2S/c1-8-2-9(8)4-17-13(19)11-3-12(11)15-7-20-5-10(15)6-21-14(16)18-15/h8-12H,2-7H2,1H3,(H2,16,18)(H,17,19)/t8-,9+,10+,11-,12-,15+/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | >3.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Inhibition of human cathepsin D |
Bioorg Med Chem 28: (2020)
Article DOI: 10.1016/j.bmc.2019.115194
BindingDB Entry DOI: 10.7270/Q2B56P8K |
More data for this
Ligand-Target Pair | |
Cathepsin D
(Homo sapiens (Human)) | BDBM50540165

(CHEMBL4637913)Show SMILES [H][C@]1(C[C@H]1C(=O)NCC)[C@]12COC[C@@]1([H])CSC(N)=N2 |r,c:20| Show InChI InChI=1S/C12H19N3O2S/c1-2-14-10(16)8-3-9(8)12-6-17-4-7(12)5-18-11(13)15-12/h7-9H,2-6H2,1H3,(H2,13,15)(H,14,16)/t7-,8+,9+,12-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >3.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Inhibition of human cathepsin D |
Bioorg Med Chem 28: (2020)
Article DOI: 10.1016/j.bmc.2019.115194
BindingDB Entry DOI: 10.7270/Q2B56P8K |
More data for this
Ligand-Target Pair | |
Cathepsin D
(Homo sapiens (Human)) | BDBM50540164

(CHEMBL4640948)Show SMILES [H][C@]1(C[C@H]1C(=O)Nc1ccc(F)cn1)[C@]12COC[C@@]1([H])CSC(N)=N2 |r,c:26| Show InChI InChI=1S/C15H17FN4O2S/c16-9-1-2-12(18-4-9)19-13(21)10-3-11(10)15-7-22-5-8(15)6-23-14(17)20-15/h1-2,4,8,10-11H,3,5-7H2,(H2,17,20)(H,18,19,21)/t8-,10+,11+,15-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >3.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Inhibition of human cathepsin D |
Bioorg Med Chem 28: (2020)
Article DOI: 10.1016/j.bmc.2019.115194
BindingDB Entry DOI: 10.7270/Q2B56P8K |
More data for this
Ligand-Target Pair | |
Cathepsin D
(Homo sapiens (Human)) | BDBM50540163

(CHEMBL4637568)Show SMILES [H][C@@]1(C[C@@H]1NC(=O)c1ccc(F)cn1)[C@]12COC[C@@]1([H])CSC(N)=N2 |r,c:26| Show InChI InChI=1S/C15H17FN4O2S/c16-9-1-2-11(18-4-9)13(21)19-12-3-10(12)15-7-22-5-8(15)6-23-14(17)20-15/h1-2,4,8,10,12H,3,5-7H2,(H2,17,20)(H,19,21)/t8-,10-,12-,15-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | >3.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Inhibition of human cathepsin D |
Bioorg Med Chem 28: (2020)
Article DOI: 10.1016/j.bmc.2019.115194
BindingDB Entry DOI: 10.7270/Q2B56P8K |
More data for this
Ligand-Target Pair | |
Cathepsin D
(Homo sapiens (Human)) | BDBM50012647

(CHEMBL2396989)Show SMILES [H][C@@]12COC[C@@]1(N=C(N)SC2)c1cc(NC(=O)c2ccc(F)cn2)ccc1F |r,t:7| Show InChI InChI=1S/C18H16F2N4O2S/c19-11-1-4-15(22-6-11)16(25)23-12-2-3-14(20)13(5-12)18-9-26-7-10(18)8-27-17(21)24-18/h1-6,10H,7-9H2,(H2,21,24)(H,23,25)/t10-,18-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | >3.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Inhibition of human cathepsin D |
Bioorg Med Chem 28: (2020)
Article DOI: 10.1016/j.bmc.2019.115194
BindingDB Entry DOI: 10.7270/Q2B56P8K |
More data for this
Ligand-Target Pair | |
Cathepsin D
(Homo sapiens (Human)) | BDBM50540166

(CHEMBL4636311)Show SMILES [H][C@]1(C[C@H]1C(=O)NCCC)[C@]12COC[C@@]1([H])CSC(N)=N2 |r,c:21| Show InChI InChI=1S/C13H21N3O2S/c1-2-3-15-11(17)9-4-10(9)13-7-18-5-8(13)6-19-12(14)16-13/h8-10H,2-7H2,1H3,(H2,14,16)(H,15,17)/t8-,9+,10+,13-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >3.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Inhibition of human cathepsin D |
Bioorg Med Chem 28: (2020)
Article DOI: 10.1016/j.bmc.2019.115194
BindingDB Entry DOI: 10.7270/Q2B56P8K |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data