 Found 59 hits Enz. Inhib. hit(s) with all data for entry = 50011377
Found 59 hits Enz. Inhib. hit(s) with all data for entry = 50011377 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Acetyl-CoA carboxylase 2
(Homo sapiens (Human)) | BDBM50546612

(CHEMBL4753907)Show SMILES CC(C)n1ncc2CC3(CCN(CC3)C(=O)c3cc(nc(c3)-c3ccc(cc3)C(O)=O)N(C)C)CC(=O)c12 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 5.60 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human ACC2 using acetyl coA as substrate incubated for 1 hr by transcreener fluorescence polarization assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00640
BindingDB Entry DOI: 10.7270/Q2RR22VT |
More data for this
Ligand-Target Pair | |
Acetyl-CoA carboxylase 2
(Homo sapiens (Human)) | BDBM50546612

(CHEMBL4753907)Show SMILES CC(C)n1ncc2CC3(CCN(CC3)C(=O)c3cc(nc(c3)-c3ccc(cc3)C(O)=O)N(C)C)CC(=O)c12 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 5.60 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human ACC2 using acetyl coA as substrate incubated for 1 hr by transcreener fluorescence polarization assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00640
BindingDB Entry DOI: 10.7270/Q2RR22VT |
More data for this
Ligand-Target Pair | |
Acetyl-CoA carboxylase 1
(Rattus norvegicus (Rat)) | BDBM50511112

(CHEMBL4567446)Show SMILES COc1cc(cc(n1)-c1ccc(cc1)C(O)=O)C(=O)N1CCC2(CC1)Cc1cnn(C(C)C)c1C(=O)C2 Show InChI InChI=1S/C28H30N4O5/c1-17(2)32-25-21(16-29-32)14-28(15-23(25)33)8-10-31(11-9-28)26(34)20-12-22(30-24(13-20)37-3)18-4-6-19(7-5-18)27(35)36/h4-7,12-13,16-17H,8-11,14-15H2,1-3H3,(H,35,36) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 7.5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of rat ACC1 using acetyl-CoA as substrate incubated for 20 mins in presence of [14C]O3 by liquid scintillation counting method |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00640
BindingDB Entry DOI: 10.7270/Q2RR22VT |
More data for this
Ligand-Target Pair | |
Acetyl-CoA carboxylase 1
(Homo sapiens (Human)) | BDBM50546612

(CHEMBL4753907)Show SMILES CC(C)n1ncc2CC3(CCN(CC3)C(=O)c3cc(nc(c3)-c3ccc(cc3)C(O)=O)N(C)C)CC(=O)c12 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 7.70 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human ACC1 using acetyl coA as substrate incubated for 1 hr by transcreener fluorescence polarization assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00640
BindingDB Entry DOI: 10.7270/Q2RR22VT |
More data for this
Ligand-Target Pair | |
Acetyl-CoA carboxylase 1
(Homo sapiens (Human)) | BDBM50546612

(CHEMBL4753907)Show SMILES CC(C)n1ncc2CC3(CCN(CC3)C(=O)c3cc(nc(c3)-c3ccc(cc3)C(O)=O)N(C)C)CC(=O)c12 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 7.90 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human ACC1 using acetyl coA as substrate incubated for 1 hr by transcreener fluorescence polarization assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00640
BindingDB Entry DOI: 10.7270/Q2RR22VT |
More data for this
Ligand-Target Pair | |
Acetyl-CoA carboxylase 2
(Homo sapiens (Human)) | BDBM50511112

(CHEMBL4567446)Show SMILES COc1cc(cc(n1)-c1ccc(cc1)C(O)=O)C(=O)N1CCC2(CC1)Cc1cnn(C(C)C)c1C(=O)C2 Show InChI InChI=1S/C28H30N4O5/c1-17(2)32-25-21(16-29-32)14-28(15-23(25)33)8-10-31(11-9-28)26(34)20-12-22(30-24(13-20)37-3)18-4-6-19(7-5-18)27(35)36/h4-7,12-13,16-17H,8-11,14-15H2,1-3H3,(H,35,36) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 8.70 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Uncompetitive inhibition of human ACC2 incubated for 15 mins in presence of ATP by Line weaver-Burk plot analysis |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00640
BindingDB Entry DOI: 10.7270/Q2RR22VT |
More data for this
Ligand-Target Pair | |
Acetyl-CoA carboxylase 2
(Homo sapiens (Human)) | BDBM50546611

(CHEMBL4762472)Show SMILES CN(C)c1cc(cc(n1)-c1ccc(cc1)C(O)=O)C(=O)N1CCC2(CC1)CC(=O)c1nn(cc1O2)C(C)(C)C | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 9.40 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human ACC2 using acetyl coA as substrate incubated for 1 hr by transcreener fluorescence polarization assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00640
BindingDB Entry DOI: 10.7270/Q2RR22VT |
More data for this
Ligand-Target Pair | |
Acetyl-CoA carboxylase 2
(Homo sapiens (Human)) | BDBM50546611

(CHEMBL4762472)Show SMILES CN(C)c1cc(cc(n1)-c1ccc(cc1)C(O)=O)C(=O)N1CCC2(CC1)CC(=O)c1nn(cc1O2)C(C)(C)C | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human ACC2 using acetyl coA as substrate incubated for 1 hr by transcreener fluorescence polarization assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00640
BindingDB Entry DOI: 10.7270/Q2RR22VT |
More data for this
Ligand-Target Pair | |
Acetyl-CoA carboxylase 1
(Homo sapiens (Human)) | BDBM50511112

(CHEMBL4567446)Show SMILES COc1cc(cc(n1)-c1ccc(cc1)C(O)=O)C(=O)N1CCC2(CC1)Cc1cnn(C(C)C)c1C(=O)C2 Show InChI InChI=1S/C28H30N4O5/c1-17(2)32-25-21(16-29-32)14-28(15-23(25)33)8-10-31(11-9-28)26(34)20-12-22(30-24(13-20)37-3)18-4-6-19(7-5-18)27(35)36/h4-7,12-13,16-17H,8-11,14-15H2,1-3H3,(H,35,36) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 12 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Uncompetitive inhibition of human ACC1 incubated for 15 mins in presence of ATP by Line weaver-Burk plot analysis |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00640
BindingDB Entry DOI: 10.7270/Q2RR22VT |
More data for this
Ligand-Target Pair | |
Acetyl-CoA carboxylase 2
(Homo sapiens (Human)) | BDBM50546609

(CHEMBL4743101)Show SMILES COc1cc(cc(n1)-c1ccc(cc1)C(O)=O)C(=O)N1CCC2(CC1)CC(=O)c1nn(cc1O2)C(C)(C)C | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 13 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human ACC2 using acetyl coA as substrate incubated for 1 hr by transcreener fluorescence polarization assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00640
BindingDB Entry DOI: 10.7270/Q2RR22VT |
More data for this
Ligand-Target Pair | |
Acetyl-CoA carboxylase 2
(Homo sapiens (Human)) | BDBM50546609

(CHEMBL4743101)Show SMILES COc1cc(cc(n1)-c1ccc(cc1)C(O)=O)C(=O)N1CCC2(CC1)CC(=O)c1nn(cc1O2)C(C)(C)C | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 13 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human ACC2 using acetyl coA as substrate incubated for 1 hr by transcreener fluorescence polarization assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00640
BindingDB Entry DOI: 10.7270/Q2RR22VT |
More data for this
Ligand-Target Pair | |
Acetyl-CoA carboxylase 1
(Homo sapiens (Human)) | BDBM50546611

(CHEMBL4762472)Show SMILES CN(C)c1cc(cc(n1)-c1ccc(cc1)C(O)=O)C(=O)N1CCC2(CC1)CC(=O)c1nn(cc1O2)C(C)(C)C | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 16 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human ACC1 using acetyl coA as substrate incubated for 1 hr by transcreener fluorescence polarization assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00640
BindingDB Entry DOI: 10.7270/Q2RR22VT |
More data for this
Ligand-Target Pair | |
Acetyl-CoA carboxylase 1
(Homo sapiens (Human)) | BDBM50546611

(CHEMBL4762472)Show SMILES CN(C)c1cc(cc(n1)-c1ccc(cc1)C(O)=O)C(=O)N1CCC2(CC1)CC(=O)c1nn(cc1O2)C(C)(C)C | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 17 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human ACC1 using acetyl coA as substrate incubated for 1 hr by transcreener fluorescence polarization assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00640
BindingDB Entry DOI: 10.7270/Q2RR22VT |
More data for this
Ligand-Target Pair | |
Acetyl-CoA carboxylase 1
(Homo sapiens (Human)) | BDBM50546609

(CHEMBL4743101)Show SMILES COc1cc(cc(n1)-c1ccc(cc1)C(O)=O)C(=O)N1CCC2(CC1)CC(=O)c1nn(cc1O2)C(C)(C)C | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 20 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human ACC1 using acetyl coA as substrate incubated for 1 hr by transcreener fluorescence polarization assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00640
BindingDB Entry DOI: 10.7270/Q2RR22VT |
More data for this
Ligand-Target Pair | |
Acetyl-CoA carboxylase 1
(Homo sapiens (Human)) | BDBM50546609

(CHEMBL4743101)Show SMILES COc1cc(cc(n1)-c1ccc(cc1)C(O)=O)C(=O)N1CCC2(CC1)CC(=O)c1nn(cc1O2)C(C)(C)C | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 22 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human ACC1 using acetyl coA as substrate incubated for 1 hr by transcreener fluorescence polarization assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00640
BindingDB Entry DOI: 10.7270/Q2RR22VT |
More data for this
Ligand-Target Pair | |
Acetyl-CoA carboxylase 2
(Homo sapiens (Human)) | BDBM50511112

(CHEMBL4567446)Show SMILES COc1cc(cc(n1)-c1ccc(cc1)C(O)=O)C(=O)N1CCC2(CC1)Cc1cnn(C(C)C)c1C(=O)C2 Show InChI InChI=1S/C28H30N4O5/c1-17(2)32-25-21(16-29-32)14-28(15-23(25)33)8-10-31(11-9-28)26(34)20-12-22(30-24(13-20)37-3)18-4-6-19(7-5-18)27(35)36/h4-7,12-13,16-17H,8-11,14-15H2,1-3H3,(H,35,36) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 25 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human ACC2 using acetyl coA as substrate incubated for 1 hr by transcreener fluorescence polarization assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00640
BindingDB Entry DOI: 10.7270/Q2RR22VT |
More data for this
Ligand-Target Pair | |
Acetyl-CoA carboxylase 2
(Homo sapiens (Human)) | BDBM50511112

(CHEMBL4567446)Show SMILES COc1cc(cc(n1)-c1ccc(cc1)C(O)=O)C(=O)N1CCC2(CC1)Cc1cnn(C(C)C)c1C(=O)C2 Show InChI InChI=1S/C28H30N4O5/c1-17(2)32-25-21(16-29-32)14-28(15-23(25)33)8-10-31(11-9-28)26(34)20-12-22(30-24(13-20)37-3)18-4-6-19(7-5-18)27(35)36/h4-7,12-13,16-17H,8-11,14-15H2,1-3H3,(H,35,36) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 26 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human ACC2 using acetyl coA as substrate incubated for 1 hr by transcreener fluorescence polarization assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00640
BindingDB Entry DOI: 10.7270/Q2RR22VT |
More data for this
Ligand-Target Pair | |
Acetyl-CoA carboxylase 1
(Homo sapiens (Human)) | BDBM50511112

(CHEMBL4567446)Show SMILES COc1cc(cc(n1)-c1ccc(cc1)C(O)=O)C(=O)N1CCC2(CC1)Cc1cnn(C(C)C)c1C(=O)C2 Show InChI InChI=1S/C28H30N4O5/c1-17(2)32-25-21(16-29-32)14-28(15-23(25)33)8-10-31(11-9-28)26(34)20-12-22(30-24(13-20)37-3)18-4-6-19(7-5-18)27(35)36/h4-7,12-13,16-17H,8-11,14-15H2,1-3H3,(H,35,36) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 32 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human ACC1 using acetyl coA as substrate incubated for 1 hr by transcreener fluorescence polarization assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00640
BindingDB Entry DOI: 10.7270/Q2RR22VT |
More data for this
Ligand-Target Pair | |
Acetyl-CoA carboxylase 2
(Homo sapiens (Human)) | BDBM50546606

(CHEMBL4759617)Show SMILES CC(C)(C)n1cc2OC3(CCN(CC3)C(=O)c3ccc4[nH]nc(C(O)=O)c4c3)CC(=O)c2n1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 32 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human ACC2 using acetyl coA as substrate incubated for 1 hr by transcreener fluorescence polarization assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00640
BindingDB Entry DOI: 10.7270/Q2RR22VT |
More data for this
Ligand-Target Pair | |
Acetyl-CoA carboxylase 1
(Homo sapiens (Human)) | BDBM50511112

(CHEMBL4567446)Show SMILES COc1cc(cc(n1)-c1ccc(cc1)C(O)=O)C(=O)N1CCC2(CC1)Cc1cnn(C(C)C)c1C(=O)C2 Show InChI InChI=1S/C28H30N4O5/c1-17(2)32-25-21(16-29-32)14-28(15-23(25)33)8-10-31(11-9-28)26(34)20-12-22(30-24(13-20)37-3)18-4-6-19(7-5-18)27(35)36/h4-7,12-13,16-17H,8-11,14-15H2,1-3H3,(H,35,36) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 33 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human ACC1 using acetyl coA as substrate incubated for 1 hr by transcreener fluorescence polarization assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00640
BindingDB Entry DOI: 10.7270/Q2RR22VT |
More data for this
Ligand-Target Pair | |
Acetyl-CoA carboxylase 2
(Homo sapiens (Human)) | BDBM50546606

(CHEMBL4759617)Show SMILES CC(C)(C)n1cc2OC3(CCN(CC3)C(=O)c3ccc4[nH]nc(C(O)=O)c4c3)CC(=O)c2n1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 33 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human ACC2 using acetyl coA as substrate incubated for 1 hr by transcreener fluorescence polarization assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00640
BindingDB Entry DOI: 10.7270/Q2RR22VT |
More data for this
Ligand-Target Pair | |
Acetyl-CoA carboxylase 2
(Homo sapiens (Human)) | BDBM50546607
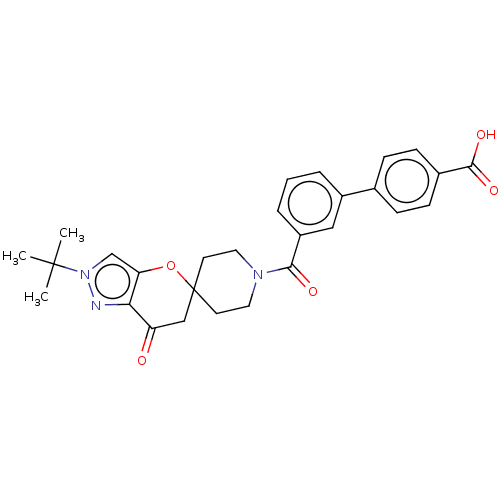
(CHEMBL4777931)Show SMILES CC(C)(C)n1cc2OC3(CCN(CC3)C(=O)c3cccc(c3)-c3ccc(cc3)C(O)=O)CC(=O)c2n1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 92 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human ACC2 using acetyl coA as substrate incubated for 1 hr by transcreener fluorescence polarization assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00640
BindingDB Entry DOI: 10.7270/Q2RR22VT |
More data for this
Ligand-Target Pair | |
Acetyl-CoA carboxylase 2
(Homo sapiens (Human)) | BDBM50546607

(CHEMBL4777931)Show SMILES CC(C)(C)n1cc2OC3(CCN(CC3)C(=O)c3cccc(c3)-c3ccc(cc3)C(O)=O)CC(=O)c2n1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 100 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human ACC2 using acetyl coA as substrate incubated for 1 hr by transcreener fluorescence polarization assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00640
BindingDB Entry DOI: 10.7270/Q2RR22VT |
More data for this
Ligand-Target Pair | |
Acetyl-CoA carboxylase 1
(Homo sapiens (Human)) | BDBM50546606

(CHEMBL4759617)Show SMILES CC(C)(C)n1cc2OC3(CCN(CC3)C(=O)c3ccc4[nH]nc(C(O)=O)c4c3)CC(=O)c2n1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 126 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human ACC1 using acetyl coA as substrate incubated for 1 hr by transcreener fluorescence polarization assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00640
BindingDB Entry DOI: 10.7270/Q2RR22VT |
More data for this
Ligand-Target Pair | |
Acetyl-CoA carboxylase 1
(Homo sapiens (Human)) | BDBM50546606

(CHEMBL4759617)Show SMILES CC(C)(C)n1cc2OC3(CCN(CC3)C(=O)c3ccc4[nH]nc(C(O)=O)c4c3)CC(=O)c2n1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 130 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human ACC1 using acetyl coA as substrate incubated for 1 hr by transcreener fluorescence polarization assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00640
BindingDB Entry DOI: 10.7270/Q2RR22VT |
More data for this
Ligand-Target Pair | |
Acetyl-CoA carboxylase 2
(Homo sapiens (Human)) | BDBM50546610

(CHEMBL4799861)Show SMILES CC(C)(C)n1cc2OC3(CCN(CC3)C(=O)c3ccnc(c3)-c3ccc(cc3)C(O)=O)CC(=O)c2n1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 190 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human ACC2 using acetyl coA as substrate incubated for 1 hr by transcreener fluorescence polarization assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00640
BindingDB Entry DOI: 10.7270/Q2RR22VT |
More data for this
Ligand-Target Pair | |
Acetyl-CoA carboxylase 2
(Homo sapiens (Human)) | BDBM50546608

(CHEMBL4785069)Show SMILES CC(C)(C)n1cc2OC3(CCN(CC3)C(=O)c3cccc(c3)-c3cccc(c3)C(O)=O)CC(=O)c2n1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 200 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human ACC2 using acetyl coA as substrate incubated for 1 hr by transcreener fluorescence polarization assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00640
BindingDB Entry DOI: 10.7270/Q2RR22VT |
More data for this
Ligand-Target Pair | |
Acetyl-CoA carboxylase 2
(Homo sapiens (Human)) | BDBM50546610

(CHEMBL4799861)Show SMILES CC(C)(C)n1cc2OC3(CCN(CC3)C(=O)c3ccnc(c3)-c3ccc(cc3)C(O)=O)CC(=O)c2n1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 200 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human ACC2 using acetyl coA as substrate incubated for 1 hr by transcreener fluorescence polarization assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00640
BindingDB Entry DOI: 10.7270/Q2RR22VT |
More data for this
Ligand-Target Pair | |
Acetyl-CoA carboxylase 2
(Homo sapiens (Human)) | BDBM50546608

(CHEMBL4785069)Show SMILES CC(C)(C)n1cc2OC3(CCN(CC3)C(=O)c3cccc(c3)-c3cccc(c3)C(O)=O)CC(=O)c2n1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 200 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human ACC2 using acetyl coA as substrate incubated for 1 hr by transcreener fluorescence polarization assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00640
BindingDB Entry DOI: 10.7270/Q2RR22VT |
More data for this
Ligand-Target Pair | |
Acetyl-CoA carboxylase 1
(Homo sapiens (Human)) | BDBM50546607

(CHEMBL4777931)Show SMILES CC(C)(C)n1cc2OC3(CCN(CC3)C(=O)c3cccc(c3)-c3ccc(cc3)C(O)=O)CC(=O)c2n1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 440 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human ACC1 using acetyl coA as substrate incubated for 1 hr by transcreener fluorescence polarization assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00640
BindingDB Entry DOI: 10.7270/Q2RR22VT |
More data for this
Ligand-Target Pair | |
Acetyl-CoA carboxylase 1
(Homo sapiens (Human)) | BDBM50546607

(CHEMBL4777931)Show SMILES CC(C)(C)n1cc2OC3(CCN(CC3)C(=O)c3cccc(c3)-c3ccc(cc3)C(O)=O)CC(=O)c2n1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 440 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human ACC1 using acetyl coA as substrate incubated for 1 hr by transcreener fluorescence polarization assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00640
BindingDB Entry DOI: 10.7270/Q2RR22VT |
More data for this
Ligand-Target Pair | |
Acetyl-CoA carboxylase 1
(Homo sapiens (Human)) | BDBM50546610

(CHEMBL4799861)Show SMILES CC(C)(C)n1cc2OC3(CCN(CC3)C(=O)c3ccnc(c3)-c3ccc(cc3)C(O)=O)CC(=O)c2n1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human ACC1 using acetyl coA as substrate incubated for 1 hr by transcreener fluorescence polarization assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00640
BindingDB Entry DOI: 10.7270/Q2RR22VT |
More data for this
Ligand-Target Pair | |
Acetyl-CoA carboxylase 1
(Homo sapiens (Human)) | BDBM50546610

(CHEMBL4799861)Show SMILES CC(C)(C)n1cc2OC3(CCN(CC3)C(=O)c3ccnc(c3)-c3ccc(cc3)C(O)=O)CC(=O)c2n1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human ACC1 using acetyl coA as substrate incubated for 1 hr by transcreener fluorescence polarization assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00640
BindingDB Entry DOI: 10.7270/Q2RR22VT |
More data for this
Ligand-Target Pair | |
Acetyl-CoA carboxylase 2
(Homo sapiens (Human)) | BDBM50546605

(CHEMBL4744660)Show SMILES CC(C)n1nc(C(O)=O)c2CC3(CCN(CC3)C(=O)c3ccc4[nH]c(C)nc4c3)CC(=O)c12 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human ACC2 using acetyl coA as substrate incubated for 1 hr by transcreener fluorescence polarization assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00640
BindingDB Entry DOI: 10.7270/Q2RR22VT |
More data for this
Ligand-Target Pair | |
Acetyl-CoA carboxylase 2
(Homo sapiens (Human)) | BDBM50546605

(CHEMBL4744660)Show SMILES CC(C)n1nc(C(O)=O)c2CC3(CCN(CC3)C(=O)c3ccc4[nH]c(C)nc4c3)CC(=O)c12 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.26E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human ACC2 using acetyl coA as substrate incubated for 1 hr by transcreener fluorescence polarization assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00640
BindingDB Entry DOI: 10.7270/Q2RR22VT |
More data for this
Ligand-Target Pair | |
Acetyl-CoA carboxylase 1
(Homo sapiens (Human)) | BDBM50546608

(CHEMBL4785069)Show SMILES CC(C)(C)n1cc2OC3(CCN(CC3)C(=O)c3cccc(c3)-c3cccc(c3)C(O)=O)CC(=O)c2n1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human ACC1 using acetyl coA as substrate incubated for 1 hr by transcreener fluorescence polarization assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00640
BindingDB Entry DOI: 10.7270/Q2RR22VT |
More data for this
Ligand-Target Pair | |
Acetyl-CoA carboxylase 1
(Homo sapiens (Human)) | BDBM50546608

(CHEMBL4785069)Show SMILES CC(C)(C)n1cc2OC3(CCN(CC3)C(=O)c3cccc(c3)-c3cccc(c3)C(O)=O)CC(=O)c2n1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.59E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human ACC1 using acetyl coA as substrate incubated for 1 hr by transcreener fluorescence polarization assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00640
BindingDB Entry DOI: 10.7270/Q2RR22VT |
More data for this
Ligand-Target Pair | |
Solute carrier organic anion transporter family member 1B1
(Homo sapiens (Human)) | BDBM50511112

(CHEMBL4567446)Show SMILES COc1cc(cc(n1)-c1ccc(cc1)C(O)=O)C(=O)N1CCC2(CC1)Cc1cnn(C(C)C)c1C(=O)C2 Show InChI InChI=1S/C28H30N4O5/c1-17(2)32-25-21(16-29-32)14-28(15-23(25)33)8-10-31(11-9-28)26(34)20-12-22(30-24(13-20)37-3)18-4-6-19(7-5-18)27(35)36/h4-7,12-13,16-17H,8-11,14-15H2,1-3H3,(H,35,36) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.17E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human OATP1B1 expressed in HEK293 cells n presence of rosuvastatin |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00640
BindingDB Entry DOI: 10.7270/Q2RR22VT |
More data for this
Ligand-Target Pair | |
Acetyl-CoA carboxylase 1
(Homo sapiens (Human)) | BDBM50546605

(CHEMBL4744660)Show SMILES CC(C)n1nc(C(O)=O)c2CC3(CCN(CC3)C(=O)c3ccc4[nH]c(C)nc4c3)CC(=O)c12 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.20E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human ACC1 using acetyl coA as substrate incubated for 1 hr by transcreener fluorescence polarization assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00640
BindingDB Entry DOI: 10.7270/Q2RR22VT |
More data for this
Ligand-Target Pair | |
Acetyl-CoA carboxylase 1
(Homo sapiens (Human)) | BDBM50546605

(CHEMBL4744660)Show SMILES CC(C)n1nc(C(O)=O)c2CC3(CCN(CC3)C(=O)c3ccc4[nH]c(C)nc4c3)CC(=O)c12 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.26E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human ACC1 using acetyl coA as substrate incubated for 1 hr by transcreener fluorescence polarization assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00640
BindingDB Entry DOI: 10.7270/Q2RR22VT |
More data for this
Ligand-Target Pair | |
UDP-glucuronosyltransferase 1A4
(Homo sapiens (Human)) | BDBM50511112

(CHEMBL4567446)Show SMILES COc1cc(cc(n1)-c1ccc(cc1)C(O)=O)C(=O)N1CCC2(CC1)Cc1cnn(C(C)C)c1C(=O)C2 Show InChI InChI=1S/C28H30N4O5/c1-17(2)32-25-21(16-29-32)14-28(15-23(25)33)8-10-31(11-9-28)26(34)20-12-22(30-24(13-20)37-3)18-4-6-19(7-5-18)27(35)36/h4-7,12-13,16-17H,8-11,14-15H2,1-3H3,(H,35,36) | UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of UGT1A4 in human liver microsomes incubated for 30 mins in presence of UDPGA by LC-MS/MS analysis |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00640
BindingDB Entry DOI: 10.7270/Q2RR22VT |
More data for this
Ligand-Target Pair | |
UDP-glucuronosyltransferase 1-6
(Homo sapiens (Human)) | BDBM50511112

(CHEMBL4567446)Show SMILES COc1cc(cc(n1)-c1ccc(cc1)C(O)=O)C(=O)N1CCC2(CC1)Cc1cnn(C(C)C)c1C(=O)C2 Show InChI InChI=1S/C28H30N4O5/c1-17(2)32-25-21(16-29-32)14-28(15-23(25)33)8-10-31(11-9-28)26(34)20-12-22(30-24(13-20)37-3)18-4-6-19(7-5-18)27(35)36/h4-7,12-13,16-17H,8-11,14-15H2,1-3H3,(H,35,36) | UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of UGT1A6 in human liver microsomes incubated for 90 mins in presence of UDPGA by LC-MS/MS analysis |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00640
BindingDB Entry DOI: 10.7270/Q2RR22VT |
More data for this
Ligand-Target Pair | |
UDP-glucuronosyltransferase 1A9
(Homo sapiens (Human)) | BDBM50511112

(CHEMBL4567446)Show SMILES COc1cc(cc(n1)-c1ccc(cc1)C(O)=O)C(=O)N1CCC2(CC1)Cc1cnn(C(C)C)c1C(=O)C2 Show InChI InChI=1S/C28H30N4O5/c1-17(2)32-25-21(16-29-32)14-28(15-23(25)33)8-10-31(11-9-28)26(34)20-12-22(30-24(13-20)37-3)18-4-6-19(7-5-18)27(35)36/h4-7,12-13,16-17H,8-11,14-15H2,1-3H3,(H,35,36) | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of UGT1A9 in human liver microsomes incubated for 30 mins in presence of UDPGA by LC-MS/MS analysis |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00640
BindingDB Entry DOI: 10.7270/Q2RR22VT |
More data for this
Ligand-Target Pair | |
UDP-glucuronosyltransferase 2B7
(Homo sapiens (Human)) | BDBM50511112

(CHEMBL4567446)Show SMILES COc1cc(cc(n1)-c1ccc(cc1)C(O)=O)C(=O)N1CCC2(CC1)Cc1cnn(C(C)C)c1C(=O)C2 Show InChI InChI=1S/C28H30N4O5/c1-17(2)32-25-21(16-29-32)14-28(15-23(25)33)8-10-31(11-9-28)26(34)20-12-22(30-24(13-20)37-3)18-4-6-19(7-5-18)27(35)36/h4-7,12-13,16-17H,8-11,14-15H2,1-3H3,(H,35,36) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of UGT2B7 in human liver microsomes incubated for 60 mins in presence of UDPGA by LC-MS/MS analysis |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00640
BindingDB Entry DOI: 10.7270/Q2RR22VT |
More data for this
Ligand-Target Pair | |
UDP-glucuronosyltransferase 2B15
(Homo sapiens) | BDBM50511112

(CHEMBL4567446)Show SMILES COc1cc(cc(n1)-c1ccc(cc1)C(O)=O)C(=O)N1CCC2(CC1)Cc1cnn(C(C)C)c1C(=O)C2 Show InChI InChI=1S/C28H30N4O5/c1-17(2)32-25-21(16-29-32)14-28(15-23(25)33)8-10-31(11-9-28)26(34)20-12-22(30-24(13-20)37-3)18-4-6-19(7-5-18)27(35)36/h4-7,12-13,16-17H,8-11,14-15H2,1-3H3,(H,35,36) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of UGT2B15 in human liver microsomes in presence of UDPGA by LC-MS/MS analysis |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00640
BindingDB Entry DOI: 10.7270/Q2RR22VT |
More data for this
Ligand-Target Pair | |
Solute carrier family 22 member 6
(Homo sapiens (Human)) | BDBM50511112

(CHEMBL4567446)Show SMILES COc1cc(cc(n1)-c1ccc(cc1)C(O)=O)C(=O)N1CCC2(CC1)Cc1cnn(C(C)C)c1C(=O)C2 Show InChI InChI=1S/C28H30N4O5/c1-17(2)32-25-21(16-29-32)14-28(15-23(25)33)8-10-31(11-9-28)26(34)20-12-22(30-24(13-20)37-3)18-4-6-19(7-5-18)27(35)36/h4-7,12-13,16-17H,8-11,14-15H2,1-3H3,(H,35,36) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human SLC22A6 expressed in HEK293 cells in presence of para-aminohippuric acid/metformin |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00640
BindingDB Entry DOI: 10.7270/Q2RR22VT |
More data for this
Ligand-Target Pair | |
Solute carrier family 22 member 2
(Homo sapiens (Human)) | BDBM50511112

(CHEMBL4567446)Show SMILES COc1cc(cc(n1)-c1ccc(cc1)C(O)=O)C(=O)N1CCC2(CC1)Cc1cnn(C(C)C)c1C(=O)C2 Show InChI InChI=1S/C28H30N4O5/c1-17(2)32-25-21(16-29-32)14-28(15-23(25)33)8-10-31(11-9-28)26(34)20-12-22(30-24(13-20)37-3)18-4-6-19(7-5-18)27(35)36/h4-7,12-13,16-17H,8-11,14-15H2,1-3H3,(H,35,36) | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human SLC22A2 expressed in HEK293 cells in presence of para-aminohippuric acid/metformin |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00640
BindingDB Entry DOI: 10.7270/Q2RR22VT |
More data for this
Ligand-Target Pair | |
Solute carrier family 22 member 8
(Homo sapiens (Human)) | BDBM50511112

(CHEMBL4567446)Show SMILES COc1cc(cc(n1)-c1ccc(cc1)C(O)=O)C(=O)N1CCC2(CC1)Cc1cnn(C(C)C)c1C(=O)C2 Show InChI InChI=1S/C28H30N4O5/c1-17(2)32-25-21(16-29-32)14-28(15-23(25)33)8-10-31(11-9-28)26(34)20-12-22(30-24(13-20)37-3)18-4-6-19(7-5-18)27(35)36/h4-7,12-13,16-17H,8-11,14-15H2,1-3H3,(H,35,36) | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human SLC22A8 expressed in HEK293 cells in presence of para-aminohippuric acid/metformin |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00640
BindingDB Entry DOI: 10.7270/Q2RR22VT |
More data for this
Ligand-Target Pair | |
Multidrug resistance-associated protein 1
(Homo sapiens (Human)) | BDBM50511112

(CHEMBL4567446)Show SMILES COc1cc(cc(n1)-c1ccc(cc1)C(O)=O)C(=O)N1CCC2(CC1)Cc1cnn(C(C)C)c1C(=O)C2 Show InChI InChI=1S/C28H30N4O5/c1-17(2)32-25-21(16-29-32)14-28(15-23(25)33)8-10-31(11-9-28)26(34)20-12-22(30-24(13-20)37-3)18-4-6-19(7-5-18)27(35)36/h4-7,12-13,16-17H,8-11,14-15H2,1-3H3,(H,35,36) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 3.90E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human ABCC1 expressed in MDCK2-LE cells in presence of pitavastatin |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00640
BindingDB Entry DOI: 10.7270/Q2RR22VT |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM50511112

(CHEMBL4567446)Show SMILES COc1cc(cc(n1)-c1ccc(cc1)C(O)=O)C(=O)N1CCC2(CC1)Cc1cnn(C(C)C)c1C(=O)C2 Show InChI InChI=1S/C28H30N4O5/c1-17(2)32-25-21(16-29-32)14-28(15-23(25)33)8-10-31(11-9-28)26(34)20-12-22(30-24(13-20)37-3)18-4-6-19(7-5-18)27(35)36/h4-7,12-13,16-17H,8-11,14-15H2,1-3H3,(H,35,36) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
inhibition of CYP2D6 in human liver microsomes |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00640
BindingDB Entry DOI: 10.7270/Q2RR22VT |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data