 Found 31 hits Enz. Inhib. hit(s) with all data for entry = 50039687
Found 31 hits Enz. Inhib. hit(s) with all data for entry = 50039687 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Renin
(Homo sapiens (Human)) | BDBM50382333

(CHEMBL2023123)Show SMILES CNC[C@@H](NC(=O)N1CCC[C@H](C1)[C@@H](OCCNC(=O)OC)c1cccc(Cl)c1)[C@@H](O)C1CCCCC1 |r| Show InChI InChI=1S/C27H43ClN4O5/c1-29-17-23(24(33)19-8-4-3-5-9-19)31-26(34)32-14-7-11-21(18-32)25(20-10-6-12-22(28)16-20)37-15-13-30-27(35)36-2/h6,10,12,16,19,21,23-25,29,33H,3-5,7-9,11,13-15,17-18H2,1-2H3,(H,30,35)(H,31,34)/t21-,23-,24+,25+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.150 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant renin using H-Asp-Arg-Val-Tyr-Ile-His-Pro-Phe-His-Leu-Val-Ile-His-Asn-OH as substrate assessed as formation of angiot... |
ACS Med Chem Lett 2: 747-751 (2011)
Article DOI: 10.1021/ml200137x
BindingDB Entry DOI: 10.7270/Q2FT8N2Q |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Renin
(Homo sapiens (Human)) | BDBM50382335
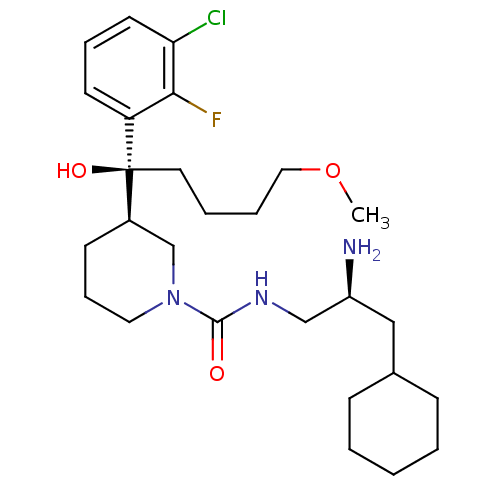
(CHEMBL2024249)Show SMILES COCCCC[C@](O)([C@@H]1CCCN(C1)C(=O)NC[C@@H](N)CC1CCCCC1)c1cccc(Cl)c1F |r| Show InChI InChI=1S/C27H43ClFN3O3/c1-35-16-6-5-14-27(34,23-12-7-13-24(28)25(23)29)21-11-8-15-32(19-21)26(33)31-18-22(30)17-20-9-3-2-4-10-20/h7,12-13,20-22,34H,2-6,8-11,14-19,30H2,1H3,(H,31,33)/t21-,22+,27+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.270 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant renin using H-Asp-Arg-Val-Tyr-Ile-His-Pro-Phe-His-Leu-Val-Ile-His-Asn-OH as substrate assessed as formation of angiot... |
ACS Med Chem Lett 2: 747-751 (2011)
Article DOI: 10.1021/ml200137x
BindingDB Entry DOI: 10.7270/Q2FT8N2Q |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Renin
(Homo sapiens (Human)) | BDBM50382334
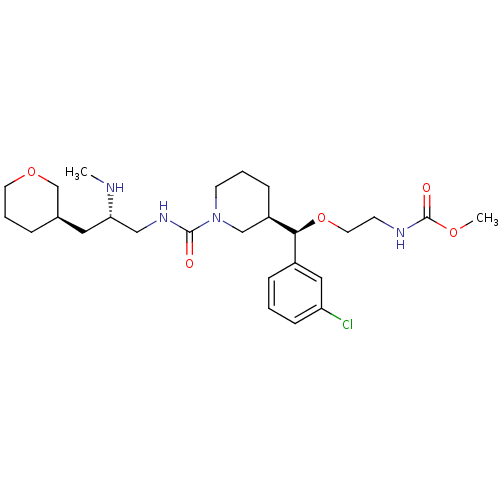
(CHEMBL1276678)Show SMILES CN[C@H](CNC(=O)N1CCC[C@H](C1)[C@@H](OCCNC(=O)OC)c1cccc(Cl)c1)C[C@H]1CCCOC1 |r| Show InChI InChI=1S/C26H41ClN4O5/c1-28-23(14-19-6-5-12-35-18-19)16-30-25(32)31-11-4-8-21(17-31)24(20-7-3-9-22(27)15-20)36-13-10-29-26(33)34-2/h3,7,9,15,19,21,23-24,28H,4-6,8,10-14,16-18H2,1-2H3,(H,29,33)(H,30,32)/t19-,21-,23+,24+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 0.300 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant renin using H-Asp-Arg-Val-Tyr-Ile-His-Pro-Phe-His-Leu-Val-Ile-His-Asn-OH as substrate assessed as formation of angiot... |
ACS Med Chem Lett 2: 747-751 (2011)
Article DOI: 10.1021/ml200137x
BindingDB Entry DOI: 10.7270/Q2FT8N2Q |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Renin
(Homo sapiens (Human)) | BDBM50382333

(CHEMBL2023123)Show SMILES CNC[C@@H](NC(=O)N1CCC[C@H](C1)[C@@H](OCCNC(=O)OC)c1cccc(Cl)c1)[C@@H](O)C1CCCCC1 |r| Show InChI InChI=1S/C27H43ClN4O5/c1-29-17-23(24(33)19-8-4-3-5-9-19)31-26(34)32-14-7-11-21(18-32)25(20-10-6-12-22(28)16-20)37-15-13-30-27(35)36-2/h6,10,12,16,19,21,23-25,29,33H,3-5,7-9,11,13-15,17-18H2,1-2H3,(H,30,35)(H,31,34)/t21-,23-,24+,25+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.330 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant renin using DABCYL-gamma-Abu-Ile-His-Pro-Phe-His-Leu-Val-Ile-His-Thr-EDANS as substrate for 60 mins by fluorimetry |
ACS Med Chem Lett 2: 747-751 (2011)
Article DOI: 10.1021/ml200137x
BindingDB Entry DOI: 10.7270/Q2FT8N2Q |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Renin
(Homo sapiens (Human)) | BDBM17950
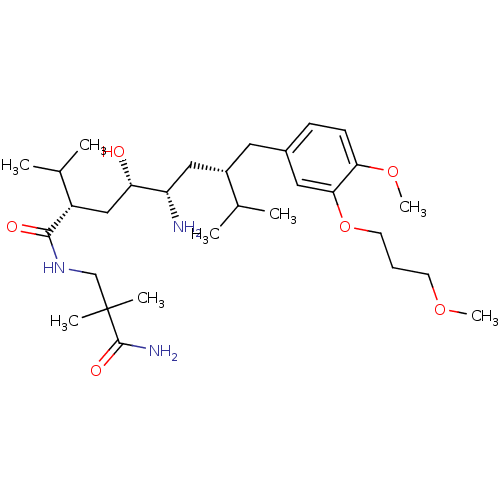
((2S,4S,5S,7S)-5-amino-N-(2-carbamoyl-2,2-dimethyle...)Show SMILES COCCCOc1cc(C[C@@H](C[C@H](N)[C@@H](O)C[C@@H](C(C)C)C(=O)NCC(C)(C)C(N)=O)C(C)C)ccc1OC |r| Show InChI InChI=1S/C30H53N3O6/c1-19(2)22(14-21-10-11-26(38-8)27(15-21)39-13-9-12-37-7)16-24(31)25(34)17-23(20(3)4)28(35)33-18-30(5,6)29(32)36/h10-11,15,19-20,22-25,34H,9,12-14,16-18,31H2,1-8H3,(H2,32,36)(H,33,35)/t22-,23-,24-,25-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
MMDB
PDB
Article
PubMed
| n/a | n/a | 0.400 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant renin using DABCYL-gamma-Abu-Ile-His-Pro-Phe-His-Leu-Val-Ile-His-Thr-EDANS as substrate for 60 mins by fluorimetry |
ACS Med Chem Lett 2: 747-751 (2011)
Article DOI: 10.1021/ml200137x
BindingDB Entry DOI: 10.7270/Q2FT8N2Q |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Renin
(Homo sapiens (Human)) | BDBM50382331
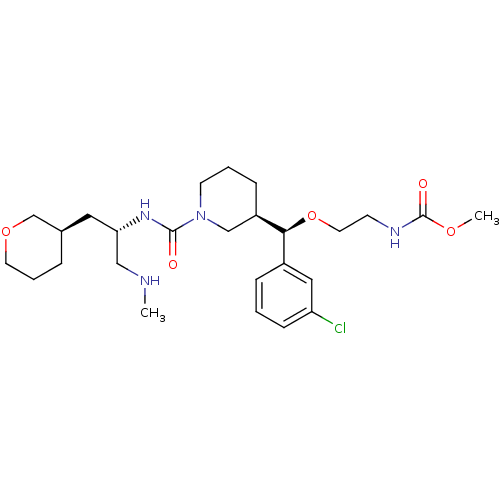
(CHEMBL2024248)Show SMILES CNC[C@H](C[C@H]1CCCOC1)NC(=O)N1CCC[C@H](C1)[C@@H](OCCNC(=O)OC)c1cccc(Cl)c1 |r| Show InChI InChI=1S/C26H41ClN4O5/c1-28-16-23(14-19-6-5-12-35-18-19)30-25(32)31-11-4-8-21(17-31)24(20-7-3-9-22(27)15-20)36-13-10-29-26(33)34-2/h3,7,9,15,19,21,23-24,28H,4-6,8,10-14,16-18H2,1-2H3,(H,29,33)(H,30,32)/t19-,21-,23+,24+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 0.420 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant renin using H-Asp-Arg-Val-Tyr-Ile-His-Pro-Phe-His-Leu-Val-Ile-His-Asn-OH as substrate assessed as formation of angiot... |
ACS Med Chem Lett 2: 747-751 (2011)
Article DOI: 10.1021/ml200137x
BindingDB Entry DOI: 10.7270/Q2FT8N2Q |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Renin
(Homo sapiens (Human)) | BDBM50382334

(CHEMBL1276678)Show SMILES CN[C@H](CNC(=O)N1CCC[C@H](C1)[C@@H](OCCNC(=O)OC)c1cccc(Cl)c1)C[C@H]1CCCOC1 |r| Show InChI InChI=1S/C26H41ClN4O5/c1-28-23(14-19-6-5-12-35-18-19)16-30-25(32)31-11-4-8-21(17-31)24(20-7-3-9-22(27)15-20)36-13-10-29-26(33)34-2/h3,7,9,15,19,21,23-24,28H,4-6,8,10-14,16-18H2,1-2H3,(H,29,33)(H,30,32)/t19-,21-,23+,24+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 0.470 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant renin using DABCYL-gamma-Abu-Ile-His-Pro-Phe-His-Leu-Val-Ile-His-Thr-EDANS as substrate for 60 mins by fluorimetry |
ACS Med Chem Lett 2: 747-751 (2011)
Article DOI: 10.1021/ml200137x
BindingDB Entry DOI: 10.7270/Q2FT8N2Q |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Renin
(Homo sapiens (Human)) | BDBM50305452
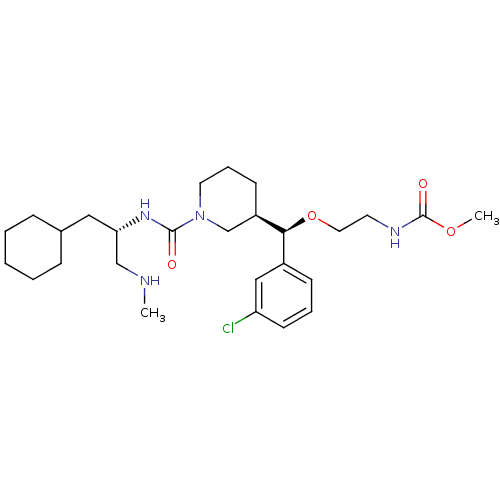
(CHEMBL591578 | methyl 2-((R)-(3-chlorophenyl)((R)-...)Show SMILES CNC[C@H](CC1CCCCC1)NC(=O)N1CCC[C@H](C1)[C@@H](OCCNC(=O)OC)c1cccc(Cl)c1 |r| Show InChI InChI=1S/C27H43ClN4O4/c1-29-18-24(16-20-8-4-3-5-9-20)31-26(33)32-14-7-11-22(19-32)25(21-10-6-12-23(28)17-21)36-15-13-30-27(34)35-2/h6,10,12,17,20,22,24-25,29H,3-5,7-9,11,13-16,18-19H2,1-2H3,(H,30,34)(H,31,33)/t22-,24+,25+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 0.480 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant renin using DABCYL-gamma-Abu-Ile-His-Pro-Phe-His-Leu-Val-Ile-His-Thr-EDANS as substrate for 60 mins by fluorimetry |
ACS Med Chem Lett 2: 747-751 (2011)
Article DOI: 10.1021/ml200137x
BindingDB Entry DOI: 10.7270/Q2FT8N2Q |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Renin
(Homo sapiens (Human)) | BDBM50382331

(CHEMBL2024248)Show SMILES CNC[C@H](C[C@H]1CCCOC1)NC(=O)N1CCC[C@H](C1)[C@@H](OCCNC(=O)OC)c1cccc(Cl)c1 |r| Show InChI InChI=1S/C26H41ClN4O5/c1-28-16-23(14-19-6-5-12-35-18-19)30-25(32)31-11-4-8-21(17-31)24(20-7-3-9-22(27)15-20)36-13-10-29-26(33)34-2/h3,7,9,15,19,21,23-24,28H,4-6,8,10-14,16-18H2,1-2H3,(H,29,33)(H,30,32)/t19-,21-,23+,24+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 0.480 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of renin in human plasma assessed as formation of angiotensin1 product after 90 mins by competitive radioimmunoassay |
ACS Med Chem Lett 2: 747-751 (2011)
Article DOI: 10.1021/ml200137x
BindingDB Entry DOI: 10.7270/Q2FT8N2Q |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Renin
(Homo sapiens (Human)) | BDBM17950

((2S,4S,5S,7S)-5-amino-N-(2-carbamoyl-2,2-dimethyle...)Show SMILES COCCCOc1cc(C[C@@H](C[C@H](N)[C@@H](O)C[C@@H](C(C)C)C(=O)NCC(C)(C)C(N)=O)C(C)C)ccc1OC |r| Show InChI InChI=1S/C30H53N3O6/c1-19(2)22(14-21-10-11-26(38-8)27(15-21)39-13-9-12-37-7)16-24(31)25(34)17-23(20(3)4)28(35)33-18-30(5,6)29(32)36/h10-11,15,19-20,22-25,34H,9,12-14,16-18,31H2,1-8H3,(H2,32,36)(H,33,35)/t22-,23-,24-,25-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
MMDB
PDB
Article
PubMed
| n/a | n/a | 0.530 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant renin using H-Asp-Arg-Val-Tyr-Ile-His-Pro-Phe-His-Leu-Val-Ile-His-Asn-OH as substrate assessed as formation of angiot... |
ACS Med Chem Lett 2: 747-751 (2011)
Article DOI: 10.1021/ml200137x
BindingDB Entry DOI: 10.7270/Q2FT8N2Q |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Renin
(Homo sapiens (Human)) | BDBM50382331

(CHEMBL2024248)Show SMILES CNC[C@H](C[C@H]1CCCOC1)NC(=O)N1CCC[C@H](C1)[C@@H](OCCNC(=O)OC)c1cccc(Cl)c1 |r| Show InChI InChI=1S/C26H41ClN4O5/c1-28-16-23(14-19-6-5-12-35-18-19)30-25(32)31-11-4-8-21(17-31)24(20-7-3-9-22(27)15-20)36-13-10-29-26(33)34-2/h3,7,9,15,19,21,23-24,28H,4-6,8,10-14,16-18H2,1-2H3,(H,29,33)(H,30,32)/t19-,21-,23+,24+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 0.560 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant renin using DABCYL-gamma-Abu-Ile-His-Pro-Phe-His-Leu-Val-Ile-His-Thr-EDANS as substrate for 60 mins by fluorimetry |
ACS Med Chem Lett 2: 747-751 (2011)
Article DOI: 10.1021/ml200137x
BindingDB Entry DOI: 10.7270/Q2FT8N2Q |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Renin
(Homo sapiens (Human)) | BDBM17950

((2S,4S,5S,7S)-5-amino-N-(2-carbamoyl-2,2-dimethyle...)Show SMILES COCCCOc1cc(C[C@@H](C[C@H](N)[C@@H](O)C[C@@H](C(C)C)C(=O)NCC(C)(C)C(N)=O)C(C)C)ccc1OC |r| Show InChI InChI=1S/C30H53N3O6/c1-19(2)22(14-21-10-11-26(38-8)27(15-21)39-13-9-12-37-7)16-24(31)25(34)17-23(20(3)4)28(35)33-18-30(5,6)29(32)36/h10-11,15,19-20,22-25,34H,9,12-14,16-18,31H2,1-8H3,(H2,32,36)(H,33,35)/t22-,23-,24-,25-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
MMDB
PDB
Article
PubMed
| n/a | n/a | 0.650 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of renin in human plasma assessed as formation of angiotensin1 product after 90 mins by competitive radioimmunoassay |
ACS Med Chem Lett 2: 747-751 (2011)
Article DOI: 10.1021/ml200137x
BindingDB Entry DOI: 10.7270/Q2FT8N2Q |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Renin
(Homo sapiens (Human)) | BDBM29957
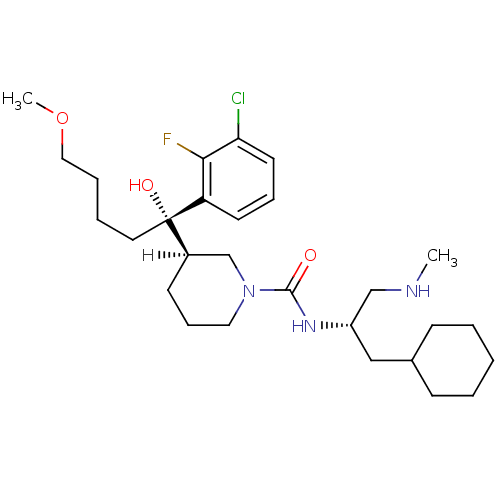
(piperidine-1-carboxamide, 21t)Show SMILES [H][C@]1(CCCN(C1)C(=O)N[C@H](CNC)CC1CCCCC1)[C@@](O)(CCCCOC)c1cccc(Cl)c1F |r| Show InChI InChI=1S/C28H45ClFN3O3/c1-31-19-23(18-21-10-4-3-5-11-21)32-27(34)33-16-9-12-22(20-33)28(35,15-6-7-17-36-2)24-13-8-14-25(29)26(24)30/h8,13-14,21-23,31,35H,3-7,9-12,15-20H2,1-2H3,(H,32,34)/t22-,23+,28+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 0.730 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant renin using DABCYL-gamma-Abu-Ile-His-Pro-Phe-His-Leu-Val-Ile-His-Thr-EDANS as substrate for 60 mins by fluorimetry |
ACS Med Chem Lett 2: 747-751 (2011)
Article DOI: 10.1021/ml200137x
BindingDB Entry DOI: 10.7270/Q2FT8N2Q |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Renin
(Homo sapiens (Human)) | BDBM50305452

(CHEMBL591578 | methyl 2-((R)-(3-chlorophenyl)((R)-...)Show SMILES CNC[C@H](CC1CCCCC1)NC(=O)N1CCC[C@H](C1)[C@@H](OCCNC(=O)OC)c1cccc(Cl)c1 |r| Show InChI InChI=1S/C27H43ClN4O4/c1-29-18-24(16-20-8-4-3-5-9-20)31-26(33)32-14-7-11-22(19-32)25(21-10-6-12-23(28)17-21)36-15-13-30-27(34)35-2/h6,10,12,17,20,22,24-25,29H,3-5,7-9,11,13-16,18-19H2,1-2H3,(H,30,34)(H,31,33)/t22-,24+,25+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 0.820 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of renin in human plasma assessed as formation of angiotensin1 product after 90 mins by competitive radioimmunoassay |
ACS Med Chem Lett 2: 747-751 (2011)
Article DOI: 10.1021/ml200137x
BindingDB Entry DOI: 10.7270/Q2FT8N2Q |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Renin
(Homo sapiens (Human)) | BDBM50382333

(CHEMBL2023123)Show SMILES CNC[C@@H](NC(=O)N1CCC[C@H](C1)[C@@H](OCCNC(=O)OC)c1cccc(Cl)c1)[C@@H](O)C1CCCCC1 |r| Show InChI InChI=1S/C27H43ClN4O5/c1-29-17-23(24(33)19-8-4-3-5-9-19)31-26(34)32-14-7-11-21(18-32)25(20-10-6-12-22(28)16-20)37-15-13-30-27(35)36-2/h6,10,12,16,19,21,23-25,29,33H,3-5,7-9,11,13-15,17-18H2,1-2H3,(H,30,35)(H,31,34)/t21-,23-,24+,25+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.06 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of renin in human plasma assessed as formation of angiotensin1 product after 90 mins by competitive radioimmunoassay |
ACS Med Chem Lett 2: 747-751 (2011)
Article DOI: 10.1021/ml200137x
BindingDB Entry DOI: 10.7270/Q2FT8N2Q |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Renin
(Homo sapiens (Human)) | BDBM50382334

(CHEMBL1276678)Show SMILES CN[C@H](CNC(=O)N1CCC[C@H](C1)[C@@H](OCCNC(=O)OC)c1cccc(Cl)c1)C[C@H]1CCCOC1 |r| Show InChI InChI=1S/C26H41ClN4O5/c1-28-23(14-19-6-5-12-35-18-19)16-30-25(32)31-11-4-8-21(17-31)24(20-7-3-9-22(27)15-20)36-13-10-29-26(33)34-2/h3,7,9,15,19,21,23-24,28H,4-6,8,10-14,16-18H2,1-2H3,(H,29,33)(H,30,32)/t19-,21-,23+,24+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 1.10 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of renin in human plasma assessed as formation of angiotensin1 product after 90 mins by competitive radioimmunoassay |
ACS Med Chem Lett 2: 747-751 (2011)
Article DOI: 10.1021/ml200137x
BindingDB Entry DOI: 10.7270/Q2FT8N2Q |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Renin
(Homo sapiens (Human)) | BDBM50382332
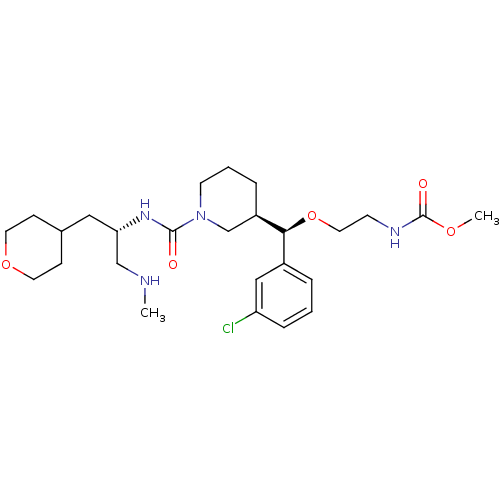
(CHEMBL2023124)Show SMILES CNC[C@H](CC1CCOCC1)NC(=O)N1CCC[C@H](C1)[C@@H](OCCNC(=O)OC)c1cccc(Cl)c1 |r| Show InChI InChI=1S/C26H41ClN4O5/c1-28-17-23(15-19-8-12-35-13-9-19)30-25(32)31-11-4-6-21(18-31)24(20-5-3-7-22(27)16-20)36-14-10-29-26(33)34-2/h3,5,7,16,19,21,23-24,28H,4,6,8-15,17-18H2,1-2H3,(H,29,33)(H,30,32)/t21-,23+,24+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.70 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant renin using H-Asp-Arg-Val-Tyr-Ile-His-Pro-Phe-His-Leu-Val-Ile-His-Asn-OH as substrate assessed as formation of angiot... |
ACS Med Chem Lett 2: 747-751 (2011)
Article DOI: 10.1021/ml200137x
BindingDB Entry DOI: 10.7270/Q2FT8N2Q |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Renin
(Homo sapiens (Human)) | BDBM50382335

(CHEMBL2024249)Show SMILES COCCCC[C@](O)([C@@H]1CCCN(C1)C(=O)NC[C@@H](N)CC1CCCCC1)c1cccc(Cl)c1F |r| Show InChI InChI=1S/C27H43ClFN3O3/c1-35-16-6-5-14-27(34,23-12-7-13-24(28)25(23)29)21-11-8-15-32(19-21)26(33)31-18-22(30)17-20-9-3-2-4-10-20/h7,12-13,20-22,34H,2-6,8-11,14-19,30H2,1H3,(H,31,33)/t21-,22+,27+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.20 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant renin using DABCYL-gamma-Abu-Ile-His-Pro-Phe-His-Leu-Val-Ile-His-Thr-EDANS as substrate for 60 mins by fluorimetry |
ACS Med Chem Lett 2: 747-751 (2011)
Article DOI: 10.1021/ml200137x
BindingDB Entry DOI: 10.7270/Q2FT8N2Q |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Renin
(Homo sapiens (Human)) | BDBM50382336
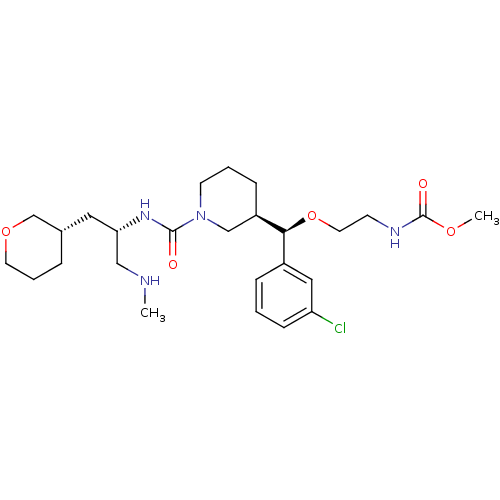
(CHEMBL2023122)Show SMILES CNC[C@H](C[C@@H]1CCCOC1)NC(=O)N1CCC[C@H](C1)[C@@H](OCCNC(=O)OC)c1cccc(Cl)c1 |r| Show InChI InChI=1S/C26H41ClN4O5/c1-28-16-23(14-19-6-5-12-35-18-19)30-25(32)31-11-4-8-21(17-31)24(20-7-3-9-22(27)15-20)36-13-10-29-26(33)34-2/h3,7,9,15,19,21,23-24,28H,4-6,8,10-14,16-18H2,1-2H3,(H,29,33)(H,30,32)/t19-,21+,23-,24-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.70 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant renin using H-Asp-Arg-Val-Tyr-Ile-His-Pro-Phe-His-Leu-Val-Ile-His-Asn-OH as substrate assessed as formation of angiot... |
ACS Med Chem Lett 2: 747-751 (2011)
Article DOI: 10.1021/ml200137x
BindingDB Entry DOI: 10.7270/Q2FT8N2Q |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Renin
(Homo sapiens (Human)) | BDBM50382336

(CHEMBL2023122)Show SMILES CNC[C@H](C[C@@H]1CCCOC1)NC(=O)N1CCC[C@H](C1)[C@@H](OCCNC(=O)OC)c1cccc(Cl)c1 |r| Show InChI InChI=1S/C26H41ClN4O5/c1-28-16-23(14-19-6-5-12-35-18-19)30-25(32)31-11-4-8-21(17-31)24(20-7-3-9-22(27)15-20)36-13-10-29-26(33)34-2/h3,7,9,15,19,21,23-24,28H,4-6,8,10-14,16-18H2,1-2H3,(H,29,33)(H,30,32)/t19-,21+,23-,24-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4.5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of renin in human plasma assessed as formation of angiotensin1 product after 90 mins by competitive radioimmunoassay |
ACS Med Chem Lett 2: 747-751 (2011)
Article DOI: 10.1021/ml200137x
BindingDB Entry DOI: 10.7270/Q2FT8N2Q |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Renin
(Homo sapiens (Human)) | BDBM50382336

(CHEMBL2023122)Show SMILES CNC[C@H](C[C@@H]1CCCOC1)NC(=O)N1CCC[C@H](C1)[C@@H](OCCNC(=O)OC)c1cccc(Cl)c1 |r| Show InChI InChI=1S/C26H41ClN4O5/c1-28-16-23(14-19-6-5-12-35-18-19)30-25(32)31-11-4-8-21(17-31)24(20-7-3-9-22(27)15-20)36-13-10-29-26(33)34-2/h3,7,9,15,19,21,23-24,28H,4-6,8,10-14,16-18H2,1-2H3,(H,29,33)(H,30,32)/t19-,21+,23-,24-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5.5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant renin using DABCYL-gamma-Abu-Ile-His-Pro-Phe-His-Leu-Val-Ile-His-Thr-EDANS as substrate for 60 mins by fluorimetry |
ACS Med Chem Lett 2: 747-751 (2011)
Article DOI: 10.1021/ml200137x
BindingDB Entry DOI: 10.7270/Q2FT8N2Q |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Renin
(Homo sapiens (Human)) | BDBM50382332

(CHEMBL2023124)Show SMILES CNC[C@H](CC1CCOCC1)NC(=O)N1CCC[C@H](C1)[C@@H](OCCNC(=O)OC)c1cccc(Cl)c1 |r| Show InChI InChI=1S/C26H41ClN4O5/c1-28-17-23(15-19-8-12-35-13-9-19)30-25(32)31-11-4-6-21(18-31)24(20-5-3-7-22(27)16-20)36-14-10-29-26(33)34-2/h3,5,7,16,19,21,23-24,28H,4,6,8-15,17-18H2,1-2H3,(H,29,33)(H,30,32)/t21-,23+,24+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6.20 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant renin using DABCYL-gamma-Abu-Ile-His-Pro-Phe-His-Leu-Val-Ile-His-Thr-EDANS as substrate for 60 mins by fluorimetry |
ACS Med Chem Lett 2: 747-751 (2011)
Article DOI: 10.1021/ml200137x
BindingDB Entry DOI: 10.7270/Q2FT8N2Q |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Renin
(Homo sapiens (Human)) | BDBM29957

(piperidine-1-carboxamide, 21t)Show SMILES [H][C@]1(CCCN(C1)C(=O)N[C@H](CNC)CC1CCCCC1)[C@@](O)(CCCCOC)c1cccc(Cl)c1F |r| Show InChI InChI=1S/C28H45ClFN3O3/c1-31-19-23(18-21-10-4-3-5-11-21)32-27(34)33-16-9-12-22(20-33)28(35,15-6-7-17-36-2)24-13-8-14-25(29)26(24)30/h8,13-14,21-23,31,35H,3-7,9-12,15-20H2,1-2H3,(H,32,34)/t22-,23+,28+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 9.40 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of renin in human plasma assessed as formation of angiotensin1 product after 90 mins by competitive radioimmunoassay |
ACS Med Chem Lett 2: 747-751 (2011)
Article DOI: 10.1021/ml200137x
BindingDB Entry DOI: 10.7270/Q2FT8N2Q |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Renin
(Homo sapiens (Human)) | BDBM50382332

(CHEMBL2023124)Show SMILES CNC[C@H](CC1CCOCC1)NC(=O)N1CCC[C@H](C1)[C@@H](OCCNC(=O)OC)c1cccc(Cl)c1 |r| Show InChI InChI=1S/C26H41ClN4O5/c1-28-17-23(15-19-8-12-35-13-9-19)30-25(32)31-11-4-6-21(18-31)24(20-5-3-7-22(27)16-20)36-14-10-29-26(33)34-2/h3,5,7,16,19,21,23-24,28H,4,6,8-15,17-18H2,1-2H3,(H,29,33)(H,30,32)/t21-,23+,24+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 13.1 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of renin in human plasma assessed as formation of angiotensin1 product after 90 mins by competitive radioimmunoassay |
ACS Med Chem Lett 2: 747-751 (2011)
Article DOI: 10.1021/ml200137x
BindingDB Entry DOI: 10.7270/Q2FT8N2Q |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Renin
(Homo sapiens (Human)) | BDBM50382335

(CHEMBL2024249)Show SMILES COCCCC[C@](O)([C@@H]1CCCN(C1)C(=O)NC[C@@H](N)CC1CCCCC1)c1cccc(Cl)c1F |r| Show InChI InChI=1S/C27H43ClFN3O3/c1-35-16-6-5-14-27(34,23-12-7-13-24(28)25(23)29)21-11-8-15-32(19-21)26(33)31-18-22(30)17-20-9-3-2-4-10-20/h7,12-13,20-22,34H,2-6,8-11,14-19,30H2,1H3,(H,31,33)/t21-,22+,27+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 69 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of renin in human plasma assessed as formation of angiotensin1 product after 90 mins by competitive radioimmunoassay |
ACS Med Chem Lett 2: 747-751 (2011)
Article DOI: 10.1021/ml200137x
BindingDB Entry DOI: 10.7270/Q2FT8N2Q |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50382335

(CHEMBL2024249)Show SMILES COCCCC[C@](O)([C@@H]1CCCN(C1)C(=O)NC[C@@H](N)CC1CCCCC1)c1cccc(Cl)c1F |r| Show InChI InChI=1S/C27H43ClFN3O3/c1-35-16-6-5-14-27(34,23-12-7-13-24(28)25(23)29)21-11-8-15-32(19-21)26(33)31-18-22(30)17-20-9-3-2-4-10-20/h7,12-13,20-22,34H,2-6,8-11,14-19,30H2,1H3,(H,31,33)/t21-,22+,27+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.91E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 using testosterone as substrate in human liver microsome for 20 mins by HPLC analysis |
ACS Med Chem Lett 2: 747-751 (2011)
Article DOI: 10.1021/ml200137x
BindingDB Entry DOI: 10.7270/Q2FT8N2Q |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50305452

(CHEMBL591578 | methyl 2-((R)-(3-chlorophenyl)((R)-...)Show SMILES CNC[C@H](CC1CCCCC1)NC(=O)N1CCC[C@H](C1)[C@@H](OCCNC(=O)OC)c1cccc(Cl)c1 |r| Show InChI InChI=1S/C27H43ClN4O4/c1-29-18-24(16-20-8-4-3-5-9-20)31-26(33)32-14-7-11-22(19-32)25(21-10-6-12-23(28)17-21)36-15-13-30-27(34)35-2/h6,10,12,17,20,22,24-25,29H,3-5,7-9,11,13-16,18-19H2,1-2H3,(H,30,34)(H,31,33)/t22-,24+,25+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 4.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 using testosterone as substrate in human liver microsome for 20 mins by HPLC analysis |
ACS Med Chem Lett 2: 747-751 (2011)
Article DOI: 10.1021/ml200137x
BindingDB Entry DOI: 10.7270/Q2FT8N2Q |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50382333

(CHEMBL2023123)Show SMILES CNC[C@@H](NC(=O)N1CCC[C@H](C1)[C@@H](OCCNC(=O)OC)c1cccc(Cl)c1)[C@@H](O)C1CCCCC1 |r| Show InChI InChI=1S/C27H43ClN4O5/c1-29-17-23(24(33)19-8-4-3-5-9-19)31-26(34)32-14-7-11-21(18-32)25(20-10-6-12-22(28)16-20)37-15-13-30-27(35)36-2/h6,10,12,16,19,21,23-25,29,33H,3-5,7-9,11,13-15,17-18H2,1-2H3,(H,30,35)(H,31,34)/t21-,23-,24+,25+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 7.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 using testosterone as substrate in human liver microsome for 20 mins by HPLC analysis |
ACS Med Chem Lett 2: 747-751 (2011)
Article DOI: 10.1021/ml200137x
BindingDB Entry DOI: 10.7270/Q2FT8N2Q |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50382331

(CHEMBL2024248)Show SMILES CNC[C@H](C[C@H]1CCCOC1)NC(=O)N1CCC[C@H](C1)[C@@H](OCCNC(=O)OC)c1cccc(Cl)c1 |r| Show InChI InChI=1S/C26H41ClN4O5/c1-28-16-23(14-19-6-5-12-35-18-19)30-25(32)31-11-4-8-21(17-31)24(20-7-3-9-22(27)15-20)36-13-10-29-26(33)34-2/h3,7,9,15,19,21,23-24,28H,4-6,8,10-14,16-18H2,1-2H3,(H,29,33)(H,30,32)/t19-,21-,23+,24+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.80E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 using testosterone as substrate in human liver microsome for 20 mins by HPLC analysis |
ACS Med Chem Lett 2: 747-751 (2011)
Article DOI: 10.1021/ml200137x
BindingDB Entry DOI: 10.7270/Q2FT8N2Q |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50382332

(CHEMBL2023124)Show SMILES CNC[C@H](CC1CCOCC1)NC(=O)N1CCC[C@H](C1)[C@@H](OCCNC(=O)OC)c1cccc(Cl)c1 |r| Show InChI InChI=1S/C26H41ClN4O5/c1-28-17-23(15-19-8-12-35-13-9-19)30-25(32)31-11-4-6-21(18-31)24(20-5-3-7-22(27)16-20)36-14-10-29-26(33)34-2/h3,5,7,16,19,21,23-24,28H,4,6,8-15,17-18H2,1-2H3,(H,29,33)(H,30,32)/t21-,23+,24+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 using testosterone as substrate in human liver microsome for 20 mins by HPLC analysis |
ACS Med Chem Lett 2: 747-751 (2011)
Article DOI: 10.1021/ml200137x
BindingDB Entry DOI: 10.7270/Q2FT8N2Q |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50382334

(CHEMBL1276678)Show SMILES CN[C@H](CNC(=O)N1CCC[C@H](C1)[C@@H](OCCNC(=O)OC)c1cccc(Cl)c1)C[C@H]1CCCOC1 |r| Show InChI InChI=1S/C26H41ClN4O5/c1-28-23(14-19-6-5-12-35-18-19)16-30-25(32)31-11-4-8-21(17-31)24(20-7-3-9-22(27)15-20)36-13-10-29-26(33)34-2/h3,7,9,15,19,21,23-24,28H,4-6,8,10-14,16-18H2,1-2H3,(H,29,33)(H,30,32)/t19-,21-,23+,24+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 using testosterone as substrate in human liver microsome for 20 mins by HPLC analysis |
ACS Med Chem Lett 2: 747-751 (2011)
Article DOI: 10.1021/ml200137x
BindingDB Entry DOI: 10.7270/Q2FT8N2Q |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data