 Found 6 hits Enz. Inhib. hit(s) with Target = 'Acetylcholinesterase' and Ligand = 'BDBM50079362'
Found 6 hits Enz. Inhib. hit(s) with Target = 'Acetylcholinesterase' and Ligand = 'BDBM50079362' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50079362
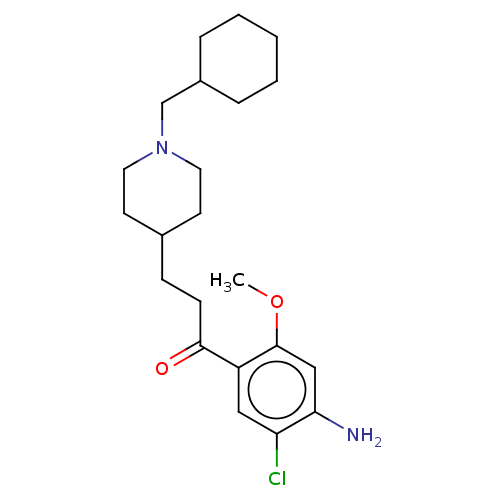
(CHEMBL3417009 | US9663465, 9)Show SMILES COc1cc(N)c(Cl)cc1C(=O)CCC1CCN(CC2CCCCC2)CC1 Show InChI InChI=1S/C22H33ClN2O2/c1-27-22-14-20(24)19(23)13-18(22)21(26)8-7-16-9-11-25(12-10-16)15-17-5-3-2-4-6-17/h13-14,16-17H,2-12,15,24H2,1H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 16 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Normandie Univ
Curated by ChEMBL
| Assay Description
Inhibition of AChE (unknown origin) |
Eur J Med Chem 162: 234-248 (2019)
Article DOI: 10.1016/j.ejmech.2018.10.064
BindingDB Entry DOI: 10.7270/Q2T72MRJ |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50079362

(CHEMBL3417009 | US9663465, 9)Show SMILES COc1cc(N)c(Cl)cc1C(=O)CCC1CCN(CC2CCCCC2)CC1 Show InChI InChI=1S/C22H33ClN2O2/c1-27-22-14-20(24)19(23)13-18(22)21(26)8-7-16-9-11-25(12-10-16)15-17-5-3-2-4-6-17/h13-14,16-17H,2-12,15,24H2,1H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 16 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.bmcl.2022.128873
BindingDB Entry DOI: 10.7270/Q2TT4VX9 |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50079362

(CHEMBL3417009 | US9663465, 9)Show SMILES COc1cc(N)c(Cl)cc1C(=O)CCC1CCN(CC2CCCCC2)CC1 Show InChI InChI=1S/C22H33ClN2O2/c1-27-22-14-20(24)19(23)13-18(22)21(26)8-7-16-9-11-25(12-10-16)15-17-5-3-2-4-6-17/h13-14,16-17H,2-12,15,24H2,1H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 16 | n/a | n/a | n/a | n/a | n/a | n/a |
Jining Medical University
Curated by ChEMBL
| Assay Description
Inhibition of human erythrocyte AChE using acetylthiocholine iodide as substrate incubated for 5 mins followed by substrate addition by Ellman's meth... |
Eur J Med Chem 169: 200-223 (2019)
Article DOI: 10.1016/j.ejmech.2019.02.076
BindingDB Entry DOI: 10.7270/Q2MS3X47 |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50079362

(CHEMBL3417009 | US9663465, 9)Show SMILES COc1cc(N)c(Cl)cc1C(=O)CCC1CCN(CC2CCCCC2)CC1 Show InChI InChI=1S/C22H33ClN2O2/c1-27-22-14-20(24)19(23)13-18(22)21(26)8-7-16-9-11-25(12-10-16)15-17-5-3-2-4-6-17/h13-14,16-17H,2-12,15,24H2,1H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 16 | n/a | n/a | n/a | n/a | n/a | n/a |
CERMN
Curated by ChEMBL
| Assay Description
Inhibition of human erythrocyte AChE pre-incubated for 5 mins before acetylthiocholine iodide substrate by Ellman' method |
J Med Chem 58: 3172-87 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00115
BindingDB Entry DOI: 10.7270/Q2HQ41MH |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50079362

(CHEMBL3417009 | US9663465, 9)Show SMILES COc1cc(N)c(Cl)cc1C(=O)CCC1CCN(CC2CCCCC2)CC1 Show InChI InChI=1S/C22H33ClN2O2/c1-27-22-14-20(24)19(23)13-18(22)21(26)8-7-16-9-11-25(12-10-16)15-17-5-3-2-4-6-17/h13-14,16-17H,2-12,15,24H2,1H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 16 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University
Curated by ChEMBL
| Assay Description
Mixed type competitive inhibition of human AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition me... |
Eur J Med Chem 158: 463-477 (2018)
Article DOI: 10.1016/j.ejmech.2018.09.031
BindingDB Entry DOI: 10.7270/Q26H4M3Z |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50079362

(CHEMBL3417009 | US9663465, 9)Show SMILES COc1cc(N)c(Cl)cc1C(=O)CCC1CCN(CC2CCCCC2)CC1 Show InChI InChI=1S/C22H33ClN2O2/c1-27-22-14-20(24)19(23)13-18(22)21(26)8-7-16-9-11-25(12-10-16)15-17-5-3-2-4-6-17/h13-14,16-17H,2-12,15,24H2,1H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| US Patent
| n/a | n/a | 63 | n/a | n/a | n/a | n/a | n/a | n/a |
UNIVERSITE DE CAEN
US Patent
| Assay Description
Acetylcholinesterase extracted from human erythrocytes (buffered aqueous solution, ≧500 units/mg, Sigma Aldrich) is diluted in a 20 mM HEPES bu... |
US Patent US9663465 (2017)
BindingDB Entry DOI: 10.7270/Q23J3G2W |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data