

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Aromatase (Homo sapiens (Human)) | BDBM9461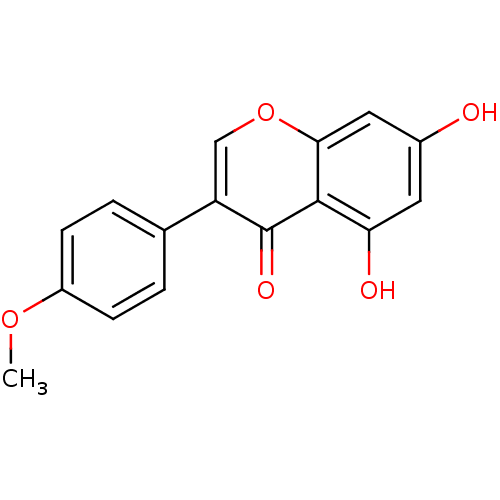 (5,7-dihydroxy-3-(4-methoxyphenyl)-4H-chromen-4-one...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 1.20E+4 | -29.2 | 3.40E+4 | n/a | n/a | n/a | n/a | 7.0 | 37 |
Ohio State University | Assay Description Inhibition of human placental aromatase was determined by monitoring the amount of 3H2O released as the enzyme converts [1beta-3H]androst-4-ene-3,17-... | J Med Chem 47: 4032-40 (2004) Article DOI: 10.1021/jm0306024 BindingDB Entry DOI: 10.7270/Q2RJ4GP7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aromatase (Homo sapiens (Human)) | BDBM9461 (5,7-dihydroxy-3-(4-methoxyphenyl)-4H-chromen-4-one...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 1.20E+4 | -29.2 | 3.40E+4 | n/a | n/a | n/a | n/a | 7.0 | 37 |
Ohio State University | Assay Description Inhibition of human placental aromatase was determined by monitoring the amount of 3H2O released as the enzyme converts [1beta-3H]androst-4-ene-3,17-... | Bioorg Med Chem 13: 4063-70 (2005) Article DOI: 10.1016/j.bmc.2005.03.050 BindingDB Entry DOI: 10.7270/Q2TB154R | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aromatase (Homo sapiens (Human)) | BDBM9461 (5,7-dihydroxy-3-(4-methoxyphenyl)-4H-chromen-4-one...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 1.20E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Indian Institute of Technology (BHU) Curated by ChEMBL | Assay Description Inhibition of human placental microsome aromatase using [1beta,2beta3H]androstenedione as substrate after 15 mins in presence of NADPH by liquid scin... | Eur J Med Chem 177: 116-143 (2019) Article DOI: 10.1016/j.ejmech.2019.05.023 BindingDB Entry DOI: 10.7270/Q2W099B9 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aromatase (Homo sapiens (Human)) | BDBM9461 (5,7-dihydroxy-3-(4-methoxyphenyl)-4H-chromen-4-one...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 10.2 | n/a | n/a | n/a | n/a | n/a | n/a |
Johns Hopkins University Curated by ChEMBL | Assay Description Inhibition of human placental microsome CYP19 | Bioorg Med Chem Lett 20: 3050-64 (2010) Article DOI: 10.1016/j.bmcl.2010.03.113 BindingDB Entry DOI: 10.7270/Q2CJ8FFS | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aromatase (Homo sapiens (Human)) | BDBM9461 (5,7-dihydroxy-3-(4-methoxyphenyl)-4H-chromen-4-one...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 1.13E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Northern Kentucky University Curated by ChEMBL | Assay Description Inhibition of aromatase | Bioorg Med Chem 20: 2603-13 (2012) Article DOI: 10.1016/j.bmc.2012.02.042 BindingDB Entry DOI: 10.7270/Q29S1S24 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aromatase (Homo sapiens (Human)) | BDBM9461 (5,7-dihydroxy-3-(4-methoxyphenyl)-4H-chromen-4-one...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 1.13E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
American University of Ras Al Khaimah Curated by ChEMBL | Assay Description Inhibition of aromatase (unknown origin) | Eur J Med Chem 102: 375-86 (2015) Article DOI: 10.1016/j.ejmech.2015.08.010 BindingDB Entry DOI: 10.7270/Q2ZC84Q6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||