 Found 6 hits of ic50 for monomerid = 288473
Found 6 hits of ic50 for monomerid = 288473 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Tyrosine-protein kinase BTK
(Homo sapiens (Human)) | BDBM288473
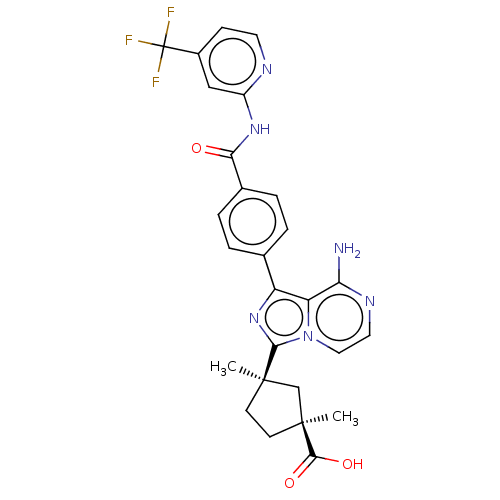
((1S,3R)-3-[8-amino-1- (4-{[4- (trifluoromethyl) py...)Show SMILES C[C@@]1(CC[C@](C)(C1)c1nc(-c2ccc(cc2)C(=O)Nc2cc(ccn2)C(F)(F)F)c2c(N)nccn12)C(O)=O |r| Show InChI InChI=1S/C27H25F3N6O3/c1-25(8-9-26(2,14-25)24(38)39)23-35-19(20-21(31)33-11-12-36(20)23)15-3-5-16(6-4-15)22(37)34-18-13-17(7-10-32-18)27(28,29)30/h3-7,10-13H,8-9,14H2,1-2H3,(H2,31,33)(H,38,39)(H,32,34,37)/t25-,26+/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck & Co Inc
Curated by ChEMBL
| Assay Description
Inhibition of 6-His-tagged recombinant full length BTK (unknown origin) expressed in baculovirus-transfected Sf9 cells using Biotin-EQEDEPEGDYFEWLE-N... |
Bioorg Med Chem Lett 27: 1471-1477 (2017)
Article DOI: 10.1016/j.bmcl.2016.11.079
BindingDB Entry DOI: 10.7270/Q20867K5 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK
(Homo sapiens (Human)) | BDBM288473

((1S,3R)-3-[8-amino-1- (4-{[4- (trifluoromethyl) py...)Show SMILES C[C@@]1(CC[C@](C)(C1)c1nc(-c2ccc(cc2)C(=O)Nc2cc(ccn2)C(F)(F)F)c2c(N)nccn12)C(O)=O |r| Show InChI InChI=1S/C27H25F3N6O3/c1-25(8-9-26(2,14-25)24(38)39)23-35-19(20-21(31)33-11-12-36(20)23)15-3-5-16(6-4-15)22(37)34-18-13-17(7-10-32-18)27(28,29)30/h3-7,10-13H,8-9,14H2,1-2H3,(H2,31,33)(H,38,39)(H,32,34,37)/t25-,26+/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 2.40 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Sharp & Dohme Corp.
US Patent
| Assay Description
BTK enzymatic activity was determined with the LANCE (Lanthanide Chelate Excite) TR-FRET (Time-resolved fluorescence resonance energy transfer) assay... |
US Patent US10087188 (2018)
BindingDB Entry DOI: 10.7270/Q2GF0WJC |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK
(Homo sapiens (Human)) | BDBM288473

((1S,3R)-3-[8-amino-1- (4-{[4- (trifluoromethyl) py...)Show SMILES C[C@@]1(CC[C@](C)(C1)c1nc(-c2ccc(cc2)C(=O)Nc2cc(ccn2)C(F)(F)F)c2c(N)nccn12)C(O)=O |r| Show InChI InChI=1S/C27H25F3N6O3/c1-25(8-9-26(2,14-25)24(38)39)23-35-19(20-21(31)33-11-12-36(20)23)15-3-5-16(6-4-15)22(37)34-18-13-17(7-10-32-18)27(28,29)30/h3-7,10-13H,8-9,14H2,1-2H3,(H2,31,33)(H,38,39)(H,32,34,37)/t25-,26+/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 8 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck & Co Inc
Curated by ChEMBL
| Assay Description
Inhibition of BTK in human PBMC |
Bioorg Med Chem Lett 27: 1471-1477 (2017)
Article DOI: 10.1016/j.bmcl.2016.11.079
BindingDB Entry DOI: 10.7270/Q20867K5 |
More data for this
Ligand-Target Pair | |
Voltage-dependent L-type calcium channel subunit alpha-1C
(Homo sapiens (Human)) | BDBM288473

((1S,3R)-3-[8-amino-1- (4-{[4- (trifluoromethyl) py...)Show SMILES C[C@@]1(CC[C@](C)(C1)c1nc(-c2ccc(cc2)C(=O)Nc2cc(ccn2)C(F)(F)F)c2c(N)nccn12)C(O)=O |r| Show InChI InChI=1S/C27H25F3N6O3/c1-25(8-9-26(2,14-25)24(38)39)23-35-19(20-21(31)33-11-12-36(20)23)15-3-5-16(6-4-15)22(37)34-18-13-17(7-10-32-18)27(28,29)30/h3-7,10-13H,8-9,14H2,1-2H3,(H2,31,33)(H,38,39)(H,32,34,37)/t25-,26+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.90E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck & Co Inc
Curated by ChEMBL
| Assay Description
Inhibition of Cav1.2 (unknown origin) |
Bioorg Med Chem Lett 27: 1471-1477 (2017)
Article DOI: 10.1016/j.bmcl.2016.11.079
BindingDB Entry DOI: 10.7270/Q20867K5 |
More data for this
Ligand-Target Pair | |
Sodium channel protein type 5 subunit alpha
(Homo sapiens (Human)) | BDBM288473

((1S,3R)-3-[8-amino-1- (4-{[4- (trifluoromethyl) py...)Show SMILES C[C@@]1(CC[C@](C)(C1)c1nc(-c2ccc(cc2)C(=O)Nc2cc(ccn2)C(F)(F)F)c2c(N)nccn12)C(O)=O |r| Show InChI InChI=1S/C27H25F3N6O3/c1-25(8-9-26(2,14-25)24(38)39)23-35-19(20-21(31)33-11-12-36(20)23)15-3-5-16(6-4-15)22(37)34-18-13-17(7-10-32-18)27(28,29)30/h3-7,10-13H,8-9,14H2,1-2H3,(H2,31,33)(H,38,39)(H,32,34,37)/t25-,26+/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck & Co Inc
Curated by ChEMBL
| Assay Description
Inhibition of Nav1.5 (unknown origin) |
Bioorg Med Chem Lett 27: 1471-1477 (2017)
Article DOI: 10.1016/j.bmcl.2016.11.079
BindingDB Entry DOI: 10.7270/Q20867K5 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM288473

((1S,3R)-3-[8-amino-1- (4-{[4- (trifluoromethyl) py...)Show SMILES C[C@@]1(CC[C@](C)(C1)c1nc(-c2ccc(cc2)C(=O)Nc2cc(ccn2)C(F)(F)F)c2c(N)nccn12)C(O)=O |r| Show InChI InChI=1S/C27H25F3N6O3/c1-25(8-9-26(2,14-25)24(38)39)23-35-19(20-21(31)33-11-12-36(20)23)15-3-5-16(6-4-15)22(37)34-18-13-17(7-10-32-18)27(28,29)30/h3-7,10-13H,8-9,14H2,1-2H3,(H2,31,33)(H,38,39)(H,32,34,37)/t25-,26+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 5.40E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck & Co Inc
Curated by ChEMBL
| Assay Description
Inhibition of human ERG |
Bioorg Med Chem Lett 27: 1471-1477 (2017)
Article DOI: 10.1016/j.bmcl.2016.11.079
BindingDB Entry DOI: 10.7270/Q20867K5 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data