

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Dimer of Gag-Pol polyprotein [489-587] (Human immunodeficiency virus type 1) | BDBM590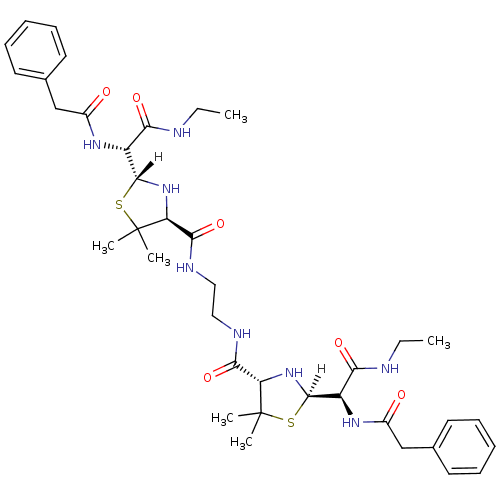 ((2R,4S)-2-[(R)-(ethylcarbamoyl)(1-phenylacetamido)...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 4.80 | n/a | n/a | n/a | n/a | 6.0 | 37 |
Glaxo Group Research Ltd. | Assay Description IC50 values were obtained by assaying the enzyme against the synthetic substrate peptide KQGTVSFNFPQIT, which was tritiated at the proline residue, a... | J Med Chem 36: 3120-8 (1993) Article DOI: 10.1021/jm00073a011 BindingDB Entry DOI: 10.7270/Q24F1NXP | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gag-Pol polyprotein [489-587] (Human immunodeficiency virus type 1) | BDBM590 ((2R,4S)-2-[(R)-(ethylcarbamoyl)(1-phenylacetamido)...) | PDB MMDB UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article | n/a | n/a | 4.80 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description The compound was tested in vitro for its inhibitory activity against HIV-1 Proteinase activity | Bioorg Med Chem Lett 3: 503-508 (1993) Article DOI: 10.1016/S0960-894X(01)81216-6 BindingDB Entry DOI: 10.7270/Q2GH9HXV | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gag-Pol polyprotein [489-587] (Human immunodeficiency virus type 1) | BDBM590 ((2R,4S)-2-[(R)-(ethylcarbamoyl)(1-phenylacetamido)...) | PDB MMDB UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 4.80 | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Group Research Limited Curated by ChEMBL | Assay Description Inhibitory activity towards HIV-1 protease | J Med Chem 35: 3080-1 (1992) BindingDB Entry DOI: 10.7270/Q27H1HJQ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||