

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Beta-3 adrenergic receptor (Rattus norvegicus) | BDBM50327860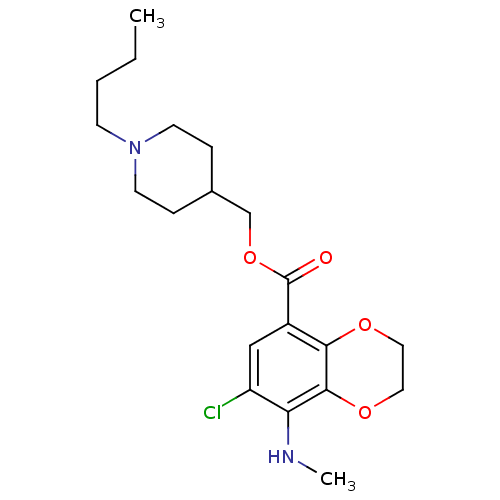 ((1-Butylpiperidin-4-yl)methyl 7-chloro-8-(methylam...) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Mental Health Curated by ChEMBL | Assay Description Binding affinity to rat adrenergic beta3 receptor | J Med Chem 53: 7035-47 (2010) Article DOI: 10.1021/jm100668r BindingDB Entry DOI: 10.7270/Q2BR8SD7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-3 adrenergic receptor (Rattus norvegicus) | BDBM50327863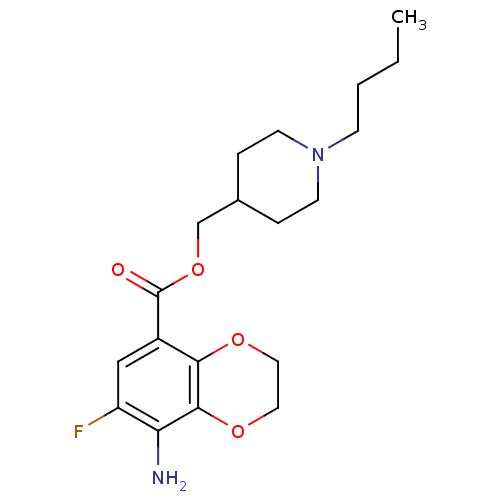 ((1-Butylpiperidin-4-yl)methyl 8-Amino-7-fluoro-2,3...) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Mental Health Curated by ChEMBL | Assay Description Binding affinity to rat adrenergic beta3 receptor | J Med Chem 53: 7035-47 (2010) Article DOI: 10.1021/jm100668r BindingDB Entry DOI: 10.7270/Q2BR8SD7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-3 adrenergic receptor (Rattus norvegicus) | BDBM50327861 ((1-Butylpiperidin-4-yl)methyl 4-Amino-3-methoxyben...) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Mental Health Curated by ChEMBL | Assay Description Binding affinity to rat adrenergic beta3 receptor | J Med Chem 53: 7035-47 (2010) Article DOI: 10.1021/jm100668r BindingDB Entry DOI: 10.7270/Q2BR8SD7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||