 Found 15 hits Enzyme Inhibition Constant Data
Found 15 hits Enzyme Inhibition Constant Data Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Cyclin-T1/Cyclin-dependent kinase 9
(255/258 = 99%)†
(Homo sapiens (Human)) | BDBM5655

(2-(2-chlorophenyl)-5,7-dihydroxy-8-[(3S,4R)-3-hydr...)Show SMILES CN1CC[C@@H]([C@H](O)C1)c1c(O)cc(O)c2c1oc(cc2=O)-c1ccccc1Cl |r| Show InChI InChI=1S/C21H20ClNO5/c1-23-7-6-12(17(27)10-23)19-14(24)8-15(25)20-16(26)9-18(28-21(19)20)11-4-2-3-5-13(11)22/h2-5,8-9,12,17,24-25,27H,6-7,10H2,1H3/t12-,17+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
MMDB
PDB
Article
PubMed
| 3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Oxford
Curated by ChEMBL
| Assay Description
Inhibition of CDK9/Cyclin T (1 to 330 amino acid residues) (unknown origin) by differential scanning fluorimetry assay |
J Med Chem 56: 660-70 (2013)
Article DOI: 10.1021/jm301495v
BindingDB Entry DOI: 10.7270/Q28G8N18 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Cyclin-dependent kinase 9
(329/329 = 100%)†
(Homo sapiens (Human)) | BDBM5655

(2-(2-chlorophenyl)-5,7-dihydroxy-8-[(3S,4R)-3-hydr...)Show SMILES CN1CC[C@@H]([C@H](O)C1)c1c(O)cc(O)c2c1oc(cc2=O)-c1ccccc1Cl |r| Show InChI InChI=1S/C21H20ClNO5/c1-23-7-6-12(17(27)10-23)19-14(24)8-15(25)20-16(26)9-18(28-21(19)20)11-4-2-3-5-13(11)22/h2-5,8-9,12,17,24-25,27H,6-7,10H2,1H3/t12-,17+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
MMDB
PDB
Article
PubMed
| 3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Oxford
Curated by ChEMBL
| Assay Description
Inhibition of CDK9 (unknown origin) |
J Med Chem 56: 660-70 (2013)
Article DOI: 10.1021/jm301495v
BindingDB Entry DOI: 10.7270/Q28G8N18 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Cyclin-T1/Cyclin-dependent kinase 9
(255/258 = 99%)†
(Homo sapiens (Human)) | BDBM5655

(2-(2-chlorophenyl)-5,7-dihydroxy-8-[(3S,4R)-3-hydr...)Show SMILES CN1CC[C@@H]([C@H](O)C1)c1c(O)cc(O)c2c1oc(cc2=O)-c1ccccc1Cl |r| Show InChI InChI=1S/C21H20ClNO5/c1-23-7-6-12(17(27)10-23)19-14(24)8-15(25)20-16(26)9-18(28-21(19)20)11-4-2-3-5-13(11)22/h2-5,8-9,12,17,24-25,27H,6-7,10H2,1H3/t12-,17+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
MMDB
PDB
Article
PubMed
| n/a | n/a | 2.5 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Nebraska Medical Center
Curated by ChEMBL
| Assay Description
Inhibition of human His6-tagged CDK9/cyclin T1 expressed in baculovirus infected sf9 cells using GST-CTD as substrate after 10 mins in presence of [g... |
J Med Chem 59: 8667-8684 (2016)
Article DOI: 10.1021/acs.jmedchem.6b00150
BindingDB Entry DOI: 10.7270/Q2G73GP8 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Cyclin-T1/Cyclin-dependent kinase 9
(255/258 = 99%)†
(Homo sapiens (Human)) | BDBM5655

(2-(2-chlorophenyl)-5,7-dihydroxy-8-[(3S,4R)-3-hydr...)Show SMILES CN1CC[C@@H]([C@H](O)C1)c1c(O)cc(O)c2c1oc(cc2=O)-c1ccccc1Cl |r| Show InChI InChI=1S/C21H20ClNO5/c1-23-7-6-12(17(27)10-23)19-14(24)8-15(25)20-16(26)9-18(28-21(19)20)11-4-2-3-5-13(11)22/h2-5,8-9,12,17,24-25,27H,6-7,10H2,1H3/t12-,17+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
MMDB
PDB
Article
PubMed
| n/a | n/a | 3.70 | n/a | n/a | n/a | n/a | n/a | n/a |
CSIR-Indian Institute of Integrative Medicine
Curated by ChEMBL
| Assay Description
Inhibition of CDK9/cyclin T (unknown origin) using YSPTSPSYSPTSPSYSPTSPKKK as substrate after 30 mins in presence of [33P]-gamma-ATP |
J Med Chem 60: 9470-9489 (2017)
Article DOI: 10.1021/acs.jmedchem.7b00663
BindingDB Entry DOI: 10.7270/Q2WH2SDH |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Cyclin-T1/Cyclin-dependent kinase 9
(255/258 = 99%)†
(Homo sapiens (Human)) | BDBM5655

(2-(2-chlorophenyl)-5,7-dihydroxy-8-[(3S,4R)-3-hydr...)Show SMILES CN1CC[C@@H]([C@H](O)C1)c1c(O)cc(O)c2c1oc(cc2=O)-c1ccccc1Cl |r| Show InChI InChI=1S/C21H20ClNO5/c1-23-7-6-12(17(27)10-23)19-14(24)8-15(25)20-16(26)9-18(28-21(19)20)11-4-2-3-5-13(11)22/h2-5,8-9,12,17,24-25,27H,6-7,10H2,1H3/t12-,17+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
MMDB
PDB
PubMed
| n/a | n/a | 4.60 | n/a | n/a | n/a | n/a | n/a | n/a |
Chinese Academy of Medical Sciences and Peking Union Medical College
Curated by ChEMBL
| Assay Description
Inhibition of CDK9/Cyclin-T1 (unknown origin) using UlightCFFKNIVTPRTPPPSQGK-amide substrate after 90 mins by autoradiography |
Medchemcomm 6: 444-454 (2015)
BindingDB Entry DOI: 10.7270/Q2CJ8G9D |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Cyclin-T1/Cyclin-dependent kinase 9
(255/258 = 99%)†
(Homo sapiens (Human)) | BDBM5655

(2-(2-chlorophenyl)-5,7-dihydroxy-8-[(3S,4R)-3-hydr...)Show SMILES CN1CC[C@@H]([C@H](O)C1)c1c(O)cc(O)c2c1oc(cc2=O)-c1ccccc1Cl |r| Show InChI InChI=1S/C21H20ClNO5/c1-23-7-6-12(17(27)10-23)19-14(24)8-15(25)20-16(26)9-18(28-21(19)20)11-4-2-3-5-13(11)22/h2-5,8-9,12,17,24-25,27H,6-7,10H2,1H3/t12-,17+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
MMDB
PDB
Article
PubMed
| n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
Vichem Chemie Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of CDK9/CycT1 (unknown origin) |
J Med Chem 57: 3939-65 (2014)
Article DOI: 10.1021/jm401742r
BindingDB Entry DOI: 10.7270/Q28C9XRB |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Cyclin-T1/Cyclin-dependent kinase 9
(255/258 = 99%)†
(Homo sapiens (Human)) | BDBM5655

(2-(2-chlorophenyl)-5,7-dihydroxy-8-[(3S,4R)-3-hydr...)Show SMILES CN1CC[C@@H]([C@H](O)C1)c1c(O)cc(O)c2c1oc(cc2=O)-c1ccccc1Cl |r| Show InChI InChI=1S/C21H20ClNO5/c1-23-7-6-12(17(27)10-23)19-14(24)8-15(25)20-16(26)9-18(28-21(19)20)11-4-2-3-5-13(11)22/h2-5,8-9,12,17,24-25,27H,6-7,10H2,1H3/t12-,17+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
MMDB
PDB
Article
PubMed
| n/a | n/a | 11 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Nebraska Medical Center
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human full length C-terminal His6-tagged CDK9/cyclin T1 expressed in baculovirus infected Sf21 insect cells using PDKtide a... |
J Med Chem 59: 8667-8684 (2016)
Article DOI: 10.1021/acs.jmedchem.6b00150
BindingDB Entry DOI: 10.7270/Q2G73GP8 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Cyclin-T1/Cyclin-dependent kinase 9
(255/258 = 99%)†
(Homo sapiens (Human)) | BDBM5655

(2-(2-chlorophenyl)-5,7-dihydroxy-8-[(3S,4R)-3-hydr...)Show SMILES CN1CC[C@@H]([C@H](O)C1)c1c(O)cc(O)c2c1oc(cc2=O)-c1ccccc1Cl |r| Show InChI InChI=1S/C21H20ClNO5/c1-23-7-6-12(17(27)10-23)19-14(24)8-15(25)20-16(26)9-18(28-21(19)20)11-4-2-3-5-13(11)22/h2-5,8-9,12,17,24-25,27H,6-7,10H2,1H3/t12-,17+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
MMDB
PDB
Article
PubMed
| n/a | n/a | 20 | n/a | n/a | n/a | n/a | n/a | n/a |
Medicinal Chemistry Division, CSIR-Indian Institute of Integrative Medicine , Canal Road, Jammu-180001, India.
Curated by ChEMBL
| Assay Description
Inhibition of CDK9/cyclin T1 (unknown origin) |
J Med Chem 61: 1664-1687 (2018)
Article DOI: 10.1021/acs.jmedchem.7b01765
BindingDB Entry DOI: 10.7270/Q2DV1NC4 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Cyclin-dependent kinase 9
(329/329 = 100%)†
(Homo sapiens (Human)) | BDBM5655

(2-(2-chlorophenyl)-5,7-dihydroxy-8-[(3S,4R)-3-hydr...)Show SMILES CN1CC[C@@H]([C@H](O)C1)c1c(O)cc(O)c2c1oc(cc2=O)-c1ccccc1Cl |r| Show InChI InChI=1S/C21H20ClNO5/c1-23-7-6-12(17(27)10-23)19-14(24)8-15(25)20-16(26)9-18(28-21(19)20)11-4-2-3-5-13(11)22/h2-5,8-9,12,17,24-25,27H,6-7,10H2,1H3/t12-,17+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
MMDB
PDB
Article
PubMed
| n/a | n/a | 20 | n/a | n/a | n/a | n/a | n/a | n/a |
Central University of Punjab
Curated by ChEMBL
| Assay Description
Inhibition of CDK9 (unknown origin) |
Eur J Med Chem 142: 424-458 (2017)
Article DOI: 10.1016/j.ejmech.2017.08.071
BindingDB Entry DOI: 10.7270/Q2S1856W |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Cyclin-dependent kinase 9
(329/329 = 100%)†
(Homo sapiens (Human)) | BDBM5655

(2-(2-chlorophenyl)-5,7-dihydroxy-8-[(3S,4R)-3-hydr...)Show SMILES CN1CC[C@@H]([C@H](O)C1)c1c(O)cc(O)c2c1oc(cc2=O)-c1ccccc1Cl |r| Show InChI InChI=1S/C21H20ClNO5/c1-23-7-6-12(17(27)10-23)19-14(24)8-15(25)20-16(26)9-18(28-21(19)20)11-4-2-3-5-13(11)22/h2-5,8-9,12,17,24-25,27H,6-7,10H2,1H3/t12-,17+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
MMDB
PDB
Article
PubMed
| n/a | n/a | 20 | n/a | n/a | n/a | n/a | n/a | n/a |
Nerviano Medical Sciences Oncology
Curated by ChEMBL
| Assay Description
Inhibition of cdk9 |
Nat Chem Biol 4: 357-65 (2008)
Article DOI: 10.1038/nchembio.90
BindingDB Entry DOI: 10.7270/Q2DV1KVK |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Cyclin-dependent kinase 9
(329/329 = 100%)†
(Homo sapiens (Human)) | BDBM5655

(2-(2-chlorophenyl)-5,7-dihydroxy-8-[(3S,4R)-3-hydr...)Show SMILES CN1CC[C@@H]([C@H](O)C1)c1c(O)cc(O)c2c1oc(cc2=O)-c1ccccc1Cl |r| Show InChI InChI=1S/C21H20ClNO5/c1-23-7-6-12(17(27)10-23)19-14(24)8-15(25)20-16(26)9-18(28-21(19)20)11-4-2-3-5-13(11)22/h2-5,8-9,12,17,24-25,27H,6-7,10H2,1H3/t12-,17+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
MMDB
PDB
US Patent
| n/a | n/a | <100 | n/a | n/a | n/a | n/a | n/a | n/a |
VIROSTATICS SRL
US Patent
| Assay Description
The activity of the compounds described in the present invention can be determined by measuring the phosphorylation of a fluorescently-labeled peptid... |
US Patent US9617225 (2017)
BindingDB Entry DOI: 10.7270/Q29W0HK6 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Cyclin-T1/Cyclin-dependent kinase 9
(255/258 = 99%)†
(Homo sapiens (Human)) | BDBM5655

(2-(2-chlorophenyl)-5,7-dihydroxy-8-[(3S,4R)-3-hydr...)Show SMILES CN1CC[C@@H]([C@H](O)C1)c1c(O)cc(O)c2c1oc(cc2=O)-c1ccccc1Cl |r| Show InChI InChI=1S/C21H20ClNO5/c1-23-7-6-12(17(27)10-23)19-14(24)8-15(25)20-16(26)9-18(28-21(19)20)11-4-2-3-5-13(11)22/h2-5,8-9,12,17,24-25,27H,6-7,10H2,1H3/t12-,17+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
MMDB
PDB
US Patent
| n/a | n/a | <100 | n/a | n/a | n/a | n/a | n/a | n/a |
GSK
| Assay Description
The activity of the compounds described in the present invention can be determined by measuring the phosphorylation of a fluorescently-labeled peptid... |
Bioorg Med Chem Lett 18: 1022-6 (2008)
BindingDB Entry DOI: 10.7270/Q2RB76X4 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Cyclin-dependent kinase 9
(329/329 = 100%)†
(Homo sapiens (Human)) | BDBM5655

(2-(2-chlorophenyl)-5,7-dihydroxy-8-[(3S,4R)-3-hydr...)Show SMILES CN1CC[C@@H]([C@H](O)C1)c1c(O)cc(O)c2c1oc(cc2=O)-c1ccccc1Cl |r| Show InChI InChI=1S/C21H20ClNO5/c1-23-7-6-12(17(27)10-23)19-14(24)8-15(25)20-16(26)9-18(28-21(19)20)11-4-2-3-5-13(11)22/h2-5,8-9,12,17,24-25,27H,6-7,10H2,1H3/t12-,17+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
MMDB
PDB
Article
PubMed
| n/a | n/a | n/a | 6.40 | n/a | n/a | n/a | n/a | n/a |
Ambit Biosciences
Curated by ChEMBL
| Assay Description
Binding constant for full-length CDK9 |
Nat Biotechnol 26: 127-32 (2008)
Article DOI: 10.1038/nbt1358
BindingDB Entry DOI: 10.7270/Q2TT4RX2 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Cyclin-dependent kinase 9
(329/329 = 100%)†
(Homo sapiens (Human)) | BDBM5655

(2-(2-chlorophenyl)-5,7-dihydroxy-8-[(3S,4R)-3-hydr...)Show SMILES CN1CC[C@@H]([C@H](O)C1)c1c(O)cc(O)c2c1oc(cc2=O)-c1ccccc1Cl |r| Show InChI InChI=1S/C21H20ClNO5/c1-23-7-6-12(17(27)10-23)19-14(24)8-15(25)20-16(26)9-18(28-21(19)20)11-4-2-3-5-13(11)22/h2-5,8-9,12,17,24-25,27H,6-7,10H2,1H3/t12-,17+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
MMDB
PDB
Article
PubMed
| n/a | n/a | n/a | 6.40 | n/a | n/a | n/a | n/a | n/a |
Ambit Biosciences
Curated by ChEMBL
| Assay Description
Binding constant for CDK9 kinase domain |
Nat Biotechnol 29: 1046-51 (2011)
Article DOI: 10.1038/nbt.1990
BindingDB Entry DOI: 10.7270/Q25D8S70 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Cyclin-dependent kinase 9
(329/329 = 100%)†
(Homo sapiens (Human)) | BDBM31099
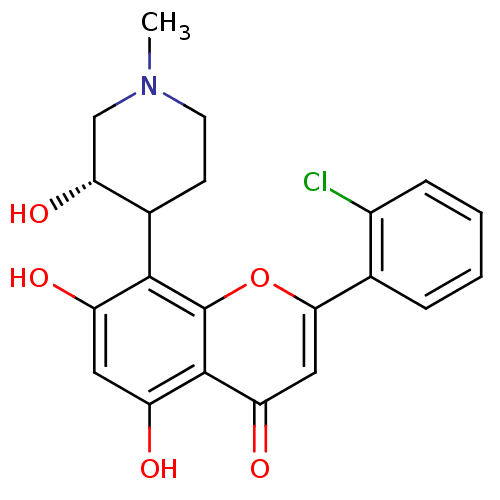
(2-(2-chlorophenyl)-5,7-dihydroxy-8-[(3S)-3-hydroxy...)Show SMILES CN1CCC([C@H](O)C1)c1c(O)cc(O)c2c1oc(cc2=O)-c1ccccc1Cl Show InChI InChI=1S/C21H20ClNO5/c1-23-7-6-12(17(27)10-23)19-14(24)8-15(25)20-16(26)9-18(28-21(19)20)11-4-2-3-5-13(11)22/h2-5,8-9,12,17,24-25,27H,6-7,10H2,1H3/t12?,17-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
PCBioAssay
| n/a | n/a | n/a | 6.40 | n/a | n/a | n/a | 7.4 | 23 |
Ambit Biosciences
Curated by PubChem BioAssay
| Assay Description
Kinase inhibitors are a new class of therapeutics with a propensity to inhibit multiple targets. The biological consequences of multi-kinase activity... |
PubChem Bioassay (2008)
BindingDB Entry DOI: 10.7270/Q2NC5ZHH |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data