 Found 16 hits of Enzyme Inhibition Constant Data
Found 16 hits of Enzyme Inhibition Constant Data Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Receptor-type tyrosine-protein phosphatase C
(Homo sapiens (Human)) | BDBM50094938

(5-Isopropoxy-2-(4-methylsulfanyl-pyridin-2-ylmetha...)Show InChI InChI=1S/C17H19N3O2S2/c1-11(2)22-13-4-5-15-16(9-13)20-17(19-15)24(21)10-12-8-14(23-3)6-7-18-12/h4-9,11H,10H2,1-3H3,(H,19,20) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 280 | n/a | n/a | n/a | n/a | n/a | n/a |
Taisho Pharmaceutical Co., Ltd
Curated by ChEMBL
| Assay Description
The compound was evaluated for its inhibitory activity towards Protein tyrosine phosphatase of CD45 |
Bioorg Med Chem Lett 10: 2657-60 (2000)
BindingDB Entry DOI: 10.7270/Q2MK6C54 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 1
(Homo sapiens (Human)) | BDBM50094938

(5-Isopropoxy-2-(4-methylsulfanyl-pyridin-2-ylmetha...)Show InChI InChI=1S/C17H19N3O2S2/c1-11(2)22-13-4-5-15-16(9-13)20-17(19-15)24(21)10-12-8-14(23-3)6-7-18-12/h4-9,11H,10H2,1-3H3,(H,19,20) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 1.82E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Taisho Pharmaceutical Co., Ltd
Curated by ChEMBL
| Assay Description
The compound was evaluated for its inhibitory activity towards Protein tyrosine phosphatase of PTP1B |
Bioorg Med Chem Lett 10: 2657-60 (2000)
BindingDB Entry DOI: 10.7270/Q2MK6C54 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 1
(Homo sapiens (Human)) | BDBM50094936

(CHEMBL86473 | Vanadate) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 3.02E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Taisho Pharmaceutical Co., Ltd
Curated by ChEMBL
| Assay Description
The compound was evaluated for its inhibitory activity towards Protein tyrosine phosphatase of PP2A |
Bioorg Med Chem Lett 10: 2657-60 (2000)
BindingDB Entry DOI: 10.7270/Q2MK6C54 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 1
(Homo sapiens (Human)) | BDBM50094936

(CHEMBL86473 | Vanadate) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 3.08E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Taisho Pharmaceutical Co., Ltd
Curated by ChEMBL
| Assay Description
The compound was evaluated for its inhibitory activity towards Protein tyrosine phosphatase of PP1 |
Bioorg Med Chem Lett 10: 2657-60 (2000)
BindingDB Entry DOI: 10.7270/Q2MK6C54 |
More data for this
Ligand-Target Pair | |
Receptor-type tyrosine-protein phosphatase C
(Homo sapiens (Human)) | BDBM50094936

(CHEMBL86473 | Vanadate) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 3.42E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Taisho Pharmaceutical Co., Ltd
Curated by ChEMBL
| Assay Description
The compound was evaluated for its inhibitory activity towards Protein tyrosine phosphatase of CD45 |
Bioorg Med Chem Lett 10: 2657-60 (2000)
BindingDB Entry DOI: 10.7270/Q2MK6C54 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 1
(Homo sapiens (Human)) | BDBM50094936

(CHEMBL86473 | Vanadate) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 3.81E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Taisho Pharmaceutical Co., Ltd
Curated by ChEMBL
| Assay Description
The compound was evaluated for its inhibitory activity towards Protein tyrosine phosphatase of PTP-S2 |
Bioorg Med Chem Lett 10: 2657-60 (2000)
BindingDB Entry DOI: 10.7270/Q2MK6C54 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 1
(Homo sapiens (Human)) | BDBM50094938

(5-Isopropoxy-2-(4-methylsulfanyl-pyridin-2-ylmetha...)Show InChI InChI=1S/C17H19N3O2S2/c1-11(2)22-13-4-5-15-16(9-13)20-17(19-15)24(21)10-12-8-14(23-3)6-7-18-12/h4-9,11H,10H2,1-3H3,(H,19,20) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 5.52E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Taisho Pharmaceutical Co., Ltd
Curated by ChEMBL
| Assay Description
The compound was evaluated for its inhibitory activity towards Protein tyrosine phosphatase of PTP-S2 |
Bioorg Med Chem Lett 10: 2657-60 (2000)
BindingDB Entry DOI: 10.7270/Q2MK6C54 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 1
(Homo sapiens (Human)) | BDBM50094938

(5-Isopropoxy-2-(4-methylsulfanyl-pyridin-2-ylmetha...)Show InChI InChI=1S/C17H19N3O2S2/c1-11(2)22-13-4-5-15-16(9-13)20-17(19-15)24(21)10-12-8-14(23-3)6-7-18-12/h4-9,11H,10H2,1-3H3,(H,19,20) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 5.54E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Taisho Pharmaceutical Co., Ltd
Curated by ChEMBL
| Assay Description
The compound was evaluated for its inhibitory activity towards Protein tyrosine phosphatase of PP1 |
Bioorg Med Chem Lett 10: 2657-60 (2000)
BindingDB Entry DOI: 10.7270/Q2MK6C54 |
More data for this
Ligand-Target Pair | |
Receptor-type tyrosine-protein phosphatase C
(Homo sapiens (Human)) | BDBM50094940
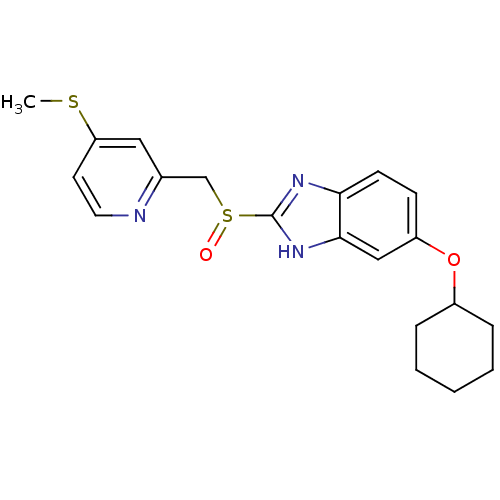
(5-Cyclohexyloxy-2-(4-methylsulfanyl-pyridin-2-ylme...)Show SMILES CSc1ccnc(CS(=O)c2nc3ccc(OC4CCCCC4)cc3[nH]2)c1 Show InChI InChI=1S/C20H23N3O2S2/c1-26-17-9-10-21-14(11-17)13-27(24)20-22-18-8-7-16(12-19(18)23-20)25-15-5-3-2-4-6-15/h7-12,15H,2-6,13H2,1H3,(H,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 8.05E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Taisho Pharmaceutical Co., Ltd
Curated by ChEMBL
| Assay Description
The compound was evaluated for its inhibitory activity towards Protein tyrosine phosphatase of CD45 |
Bioorg Med Chem Lett 10: 2657-60 (2000)
BindingDB Entry DOI: 10.7270/Q2MK6C54 |
More data for this
Ligand-Target Pair | |
Receptor-type tyrosine-protein phosphatase C
(Homo sapiens (Human)) | BDBM50094937
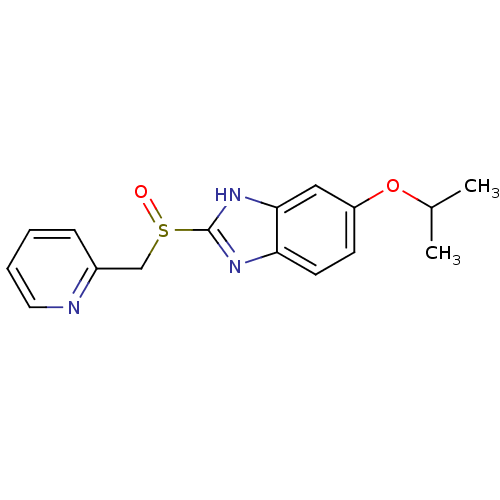
(5-Isopropoxy-2-(pyridin-2-ylmethanesulfinyl)-1H-be...)Show InChI InChI=1S/C16H17N3O2S/c1-11(2)21-13-6-7-14-15(9-13)19-16(18-14)22(20)10-12-5-3-4-8-17-12/h3-9,11H,10H2,1-2H3,(H,18,19) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 1.20E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Taisho Pharmaceutical Co., Ltd
Curated by ChEMBL
| Assay Description
The compound was evaluated for its inhibitory activity towards Protein tyrosine phosphatase of CD45 |
Bioorg Med Chem Lett 10: 2657-60 (2000)
BindingDB Entry DOI: 10.7270/Q2MK6C54 |
More data for this
Ligand-Target Pair | |
Receptor-type tyrosine-protein phosphatase C
(Homo sapiens (Human)) | BDBM50094935
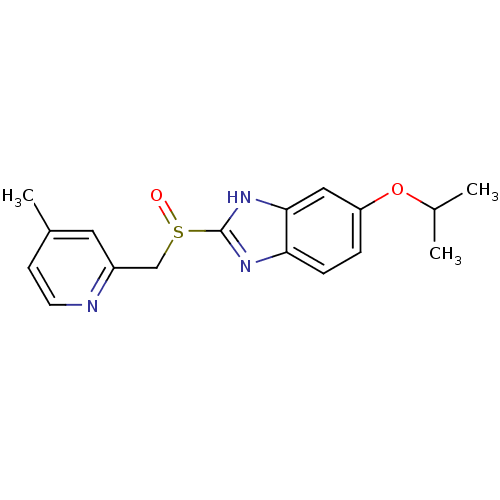
(5-Isopropoxy-2-(4-methyl-pyridin-2-ylmethanesulfin...)Show InChI InChI=1S/C17H19N3O2S/c1-11(2)22-14-4-5-15-16(9-14)20-17(19-15)23(21)10-13-8-12(3)6-7-18-13/h4-9,11H,10H2,1-3H3,(H,19,20) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 3.80E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Taisho Pharmaceutical Co., Ltd
Curated by ChEMBL
| Assay Description
The compound was evaluated for its inhibitory activity towards Protein tyrosine phosphatase of CD45 |
Bioorg Med Chem Lett 10: 2657-60 (2000)
BindingDB Entry DOI: 10.7270/Q2MK6C54 |
More data for this
Ligand-Target Pair | |
Receptor-type tyrosine-protein phosphatase C
(Homo sapiens (Human)) | BDBM50094939
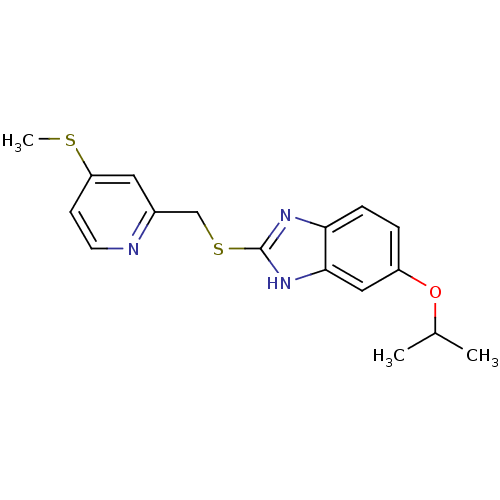
(5-Isopropoxy-2-(4-methylsulfanyl-pyridin-2-ylmethy...)Show InChI InChI=1S/C17H19N3OS2/c1-11(2)21-13-4-5-15-16(9-13)20-17(19-15)23-10-12-8-14(22-3)6-7-18-12/h4-9,11H,10H2,1-3H3,(H,19,20) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | >1.00E+6 | n/a | n/a | n/a | n/a | n/a | n/a |
Taisho Pharmaceutical Co., Ltd
Curated by ChEMBL
| Assay Description
The compound was evaluated for its inhibitory activity towards Protein tyrosine phosphatase of CD45 |
Bioorg Med Chem Lett 10: 2657-60 (2000)
BindingDB Entry DOI: 10.7270/Q2MK6C54 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 1
(Homo sapiens (Human)) | BDBM50094936

(CHEMBL86473 | Vanadate) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | >1.00E+6 | n/a | n/a | n/a | n/a | n/a | n/a |
Taisho Pharmaceutical Co., Ltd
Curated by ChEMBL
| Assay Description
The compound was evaluated for its inhibitory activity towards Protein tyrosine phosphatase of PTP1B |
Bioorg Med Chem Lett 10: 2657-60 (2000)
BindingDB Entry DOI: 10.7270/Q2MK6C54 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 1
(Homo sapiens (Human)) | BDBM50094938

(5-Isopropoxy-2-(4-methylsulfanyl-pyridin-2-ylmetha...)Show InChI InChI=1S/C17H19N3O2S2/c1-11(2)22-13-4-5-15-16(9-13)20-17(19-15)24(21)10-12-8-14(23-3)6-7-18-12/h4-9,11H,10H2,1-3H3,(H,19,20) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | >1.00E+6 | n/a | n/a | n/a | n/a | n/a | n/a |
Taisho Pharmaceutical Co., Ltd
Curated by ChEMBL
| Assay Description
The compound was evaluated for its inhibitory activity towards Protein tyrosine phosphatase of LAR |
Bioorg Med Chem Lett 10: 2657-60 (2000)
BindingDB Entry DOI: 10.7270/Q2MK6C54 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 1
(Homo sapiens (Human)) | BDBM50094938

(5-Isopropoxy-2-(4-methylsulfanyl-pyridin-2-ylmetha...)Show InChI InChI=1S/C17H19N3O2S2/c1-11(2)22-13-4-5-15-16(9-13)20-17(19-15)24(21)10-12-8-14(23-3)6-7-18-12/h4-9,11H,10H2,1-3H3,(H,19,20) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | >1.00E+6 | n/a | n/a | n/a | n/a | n/a | n/a |
Taisho Pharmaceutical Co., Ltd
Curated by ChEMBL
| Assay Description
The compound was evaluated for its inhibitory activity towards Protein tyrosine phosphatase of PP2A |
Bioorg Med Chem Lett 10: 2657-60 (2000)
BindingDB Entry DOI: 10.7270/Q2MK6C54 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 1
(Homo sapiens (Human)) | BDBM50094936

(CHEMBL86473 | Vanadate) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | >1.00E+6 | n/a | n/a | n/a | n/a | n/a | n/a |
Taisho Pharmaceutical Co., Ltd
Curated by ChEMBL
| Assay Description
The compound was evaluated for its inhibitory activity towards Protein tyrosine phosphatase of LAR |
Bioorg Med Chem Lett 10: 2657-60 (2000)
BindingDB Entry DOI: 10.7270/Q2MK6C54 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data