 Found 46 hits of Enzyme Inhibition Constant Data
Found 46 hits of Enzyme Inhibition Constant Data Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Vasopressin V1a receptor
(RAT) | BDBM50406623

(CHEMBL2115369)Show SMILES COc1ccc(C[C@@H]2NC(=O)C3(CCCC3)CSSC[C@H](NC(=O)[C@H](CC(N)=O)NC(=O)[C@@H](NC(=O)[C@H](Cc3ccccc3)NC2=O)C(C)C)C(=O)N2CCC[C@H]2C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](Cc2ccc(O)cc2)C(N)=O)cc1 Show InChI InChI=1S/C58H79N11O12S2/c1-34(2)48-55(78)65-44(31-47(60)71)52(75)66-45(56(79)69-27-11-15-46(69)54(77)62-40(14-7-10-26-59)50(73)63-41(49(61)72)28-36-16-20-38(70)21-17-36)32-82-83-33-58(24-8-9-25-58)57(80)67-43(30-37-18-22-39(81-3)23-19-37)51(74)64-42(53(76)68-48)29-35-12-5-4-6-13-35/h4-6,12-13,16-23,34,40-46,48,70H,7-11,14-15,24-33,59H2,1-3H3,(H2,60,71)(H2,61,72)(H,62,77)(H,63,73)(H,64,74)(H,65,78)(H,66,75)(H,67,80)(H,68,76)/t40-,41-,42-,43-,44-,45-,46-,48-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 1.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Sherbrooke
Curated by ChEMBL
| Assay Description
Negative log of Kd for Vasopressin V1 receptor |
J Med Chem 35: 151-7 (1992)
BindingDB Entry DOI: 10.7270/Q2668DT9 |
More data for this
Ligand-Target Pair | |
Vasopressin V1a receptor
(RAT) | BDBM50406622
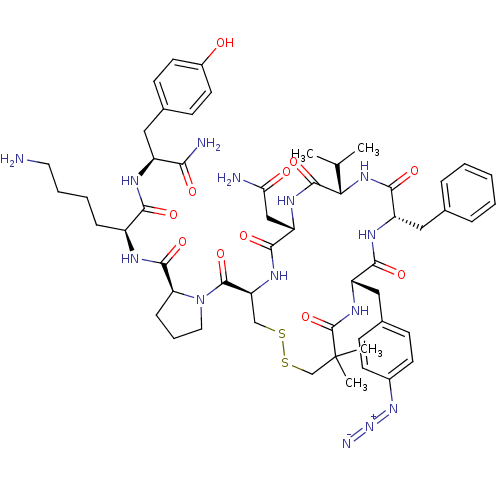
(CHEMBL2115364)Show SMILES CC(C)[C@@H]1NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@H](Cc2ccc(cc2)N=[N+]=[N-])NC(=O)C(C)(C)CSSC[C@H](NC(=O)[C@H](CC(N)=O)NC1=O)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](Cc1ccc(O)cc1)C(N)=O Show InChI InChI=1S/C55H74N14O11S2/c1-31(2)45-52(78)63-41(28-44(57)71)49(75)64-42(53(79)69-24-10-14-43(69)51(77)60-37(13-8-9-23-56)47(73)61-38(46(58)72)25-34-17-21-36(70)22-18-34)29-81-82-30-55(3,4)54(80)65-40(27-33-15-19-35(20-16-33)67-68-59)48(74)62-39(50(76)66-45)26-32-11-6-5-7-12-32/h5-7,11-12,15-22,31,37-43,45,70H,8-10,13-14,23-30,56H2,1-4H3,(H2,57,71)(H2,58,72)(H,60,77)(H,61,73)(H,62,74)(H,63,78)(H,64,75)(H,65,80)(H,66,76)/t37-,38-,39-,40-,41-,42-,43-,45-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 1.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Sherbrooke
Curated by ChEMBL
| Assay Description
Inhibition of the 3 nM VP stimulation of phospholipase-C in cultured WRK cells was used as a V1-mediated assay |
J Med Chem 35: 151-7 (1992)
BindingDB Entry DOI: 10.7270/Q2668DT9 |
More data for this
Ligand-Target Pair | |
Vasopressin V1a receptor
(RAT) | BDBM50008092

(CHEMBL2372415 | cyclo(Ppa-Phe(NO2)-Phe-Val-Asn-Cys...)Show SMILES CC(C)[C@@H]1NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@H](Cc2ccc(cc2)[N+]([O-])=O)NC(=O)C2(CCCC2)CSSC[C@H](NC(=O)[C@H](CC(N)=O)NC1=O)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCCN)C(=O)N[C@H](Cc1ccc(O)cc1)C(N)=O Show InChI InChI=1S/C57H76N12O13S2/c1-33(2)47-54(78)64-43(30-46(59)71)51(75)65-44(55(79)68-26-10-14-45(68)53(77)61-39(13-6-9-25-58)49(73)62-40(48(60)72)27-36-17-21-38(70)22-18-36)31-83-84-32-57(23-7-8-24-57)56(80)66-42(29-35-15-19-37(20-16-35)69(81)82)50(74)63-41(52(76)67-47)28-34-11-4-3-5-12-34/h3-5,11-12,15-22,33,39-45,47,70H,6-10,13-14,23-32,58H2,1-2H3,(H2,59,71)(H2,60,72)(H,61,77)(H,62,73)(H,63,74)(H,64,78)(H,65,75)(H,66,80)(H,67,76)/t39-,40+,41-,42-,43-,44-,45-,47-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | 3.20 | n/a | n/a | n/a | n/a | n/a |
University of Sherbrooke
Curated by ChEMBL
| Assay Description
Binding potency against Vasopressin V1 receptor in rat liver cells. |
J Med Chem 35: 151-7 (1992)
BindingDB Entry DOI: 10.7270/Q2668DT9 |
More data for this
Ligand-Target Pair | |
Vasopressin V1a receptor
(RAT) | BDBM50406623

(CHEMBL2115369)Show SMILES COc1ccc(C[C@@H]2NC(=O)C3(CCCC3)CSSC[C@H](NC(=O)[C@H](CC(N)=O)NC(=O)[C@@H](NC(=O)[C@H](Cc3ccccc3)NC2=O)C(C)C)C(=O)N2CCC[C@H]2C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](Cc2ccc(O)cc2)C(N)=O)cc1 Show InChI InChI=1S/C58H79N11O12S2/c1-34(2)48-55(78)65-44(31-47(60)71)52(75)66-45(56(79)69-27-11-15-46(69)54(77)62-40(14-7-10-26-59)50(73)63-41(49(61)72)28-36-16-20-38(70)21-17-36)32-82-83-33-58(24-8-9-25-58)57(80)67-43(30-37-18-22-39(81-3)23-19-37)51(74)64-42(53(76)68-48)29-35-12-5-4-6-13-35/h4-6,12-13,16-23,34,40-46,48,70H,7-11,14-15,24-33,59H2,1-3H3,(H2,60,71)(H2,61,72)(H,62,77)(H,63,73)(H,64,74)(H,65,78)(H,66,75)(H,67,80)(H,68,76)/t40-,41-,42-,43-,44-,45-,46-,48-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | 0.180 | n/a | n/a | n/a | n/a | n/a |
University of Sherbrooke
Curated by ChEMBL
| Assay Description
Binding potency against Vasopressin V1 receptor in rat liver cells. |
J Med Chem 35: 151-7 (1992)
BindingDB Entry DOI: 10.7270/Q2668DT9 |
More data for this
Ligand-Target Pair | |
Vasopressin V1a receptor
(RAT) | BDBM50008091
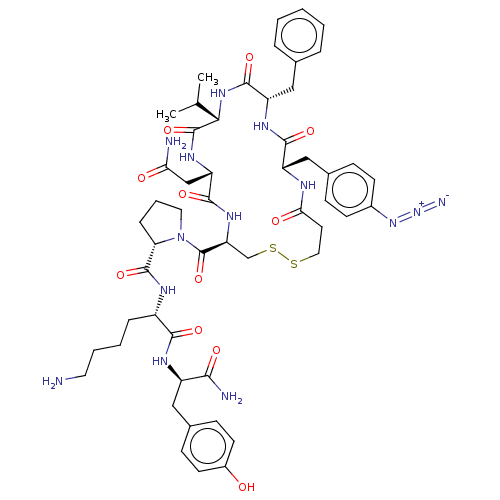
(1-[16-(4-Azido-benzyl)-13-benzyl-7-carbamoylmethyl...)Show SMILES CC(C)[C@@H]1NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@H](Cc2ccc(cc2)N=[N+]=[N-])NC(=O)CCSSC[C@H](NC(=O)[C@H](CC(N)=O)NC1=O)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCCN)C(=O)N[C@H](Cc1ccc(O)cc1)C(N)=O Show InChI InChI=1S/C53H70N14O11S2/c1-30(2)45-52(77)62-40(28-43(55)69)49(74)63-41(53(78)67-23-8-12-42(67)51(76)59-36(11-6-7-22-54)47(72)60-37(46(56)71)25-33-15-19-35(68)20-16-33)29-80-79-24-21-44(70)58-38(27-32-13-17-34(18-14-32)65-66-57)48(73)61-39(50(75)64-45)26-31-9-4-3-5-10-31/h3-5,9-10,13-20,30,36-42,45,68H,6-8,11-12,21-29,54H2,1-2H3,(H2,55,69)(H2,56,71)(H,58,70)(H,59,76)(H,60,72)(H,61,73)(H,62,77)(H,63,74)(H,64,75)/t36-,37+,38-,39-,40-,41-,42-,45-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a |
University of Sherbrooke
Curated by ChEMBL
| Assay Description
Binding potency against Vasopressin V1 receptor in rat liver cells. |
J Med Chem 35: 151-7 (1992)
BindingDB Entry DOI: 10.7270/Q2668DT9 |
More data for this
Ligand-Target Pair | |
Vasopressin V1a receptor
(RAT) | BDBM50406623

(CHEMBL2115369)Show SMILES COc1ccc(C[C@@H]2NC(=O)C3(CCCC3)CSSC[C@H](NC(=O)[C@H](CC(N)=O)NC(=O)[C@@H](NC(=O)[C@H](Cc3ccccc3)NC2=O)C(C)C)C(=O)N2CCC[C@H]2C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](Cc2ccc(O)cc2)C(N)=O)cc1 Show InChI InChI=1S/C58H79N11O12S2/c1-34(2)48-55(78)65-44(31-47(60)71)52(75)66-45(56(79)69-27-11-15-46(69)54(77)62-40(14-7-10-26-59)50(73)63-41(49(61)72)28-36-16-20-38(70)21-17-36)32-82-83-33-58(24-8-9-25-58)57(80)67-43(30-37-18-22-39(81-3)23-19-37)51(74)64-42(53(76)68-48)29-35-12-5-4-6-13-35/h4-6,12-13,16-23,34,40-46,48,70H,7-11,14-15,24-33,59H2,1-3H3,(H2,60,71)(H2,61,72)(H,62,77)(H,63,73)(H,64,74)(H,65,78)(H,66,75)(H,67,80)(H,68,76)/t40-,41-,42-,43-,44-,45-,46-,48-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | 0.182 | n/a | n/a | n/a | n/a | n/a |
University of Sherbrooke
Curated by ChEMBL
| Assay Description
Negative log of Kd for Vasopressin V1 receptor |
J Med Chem 35: 151-7 (1992)
BindingDB Entry DOI: 10.7270/Q2668DT9 |
More data for this
Ligand-Target Pair | |
Vasopressin V1a receptor
(RAT) | BDBM50008088

(CHEMBL2372416 | cyclo(Depa-Phe(N3)-Phe-Val-Asn-Cys...)Show SMILES CCC1(CC)CSSC[C@H](NC(=O)[C@H](CC(N)=O)NC(=O)[C@@H](NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@H](Cc2ccc(cc2)N=[N+]=[N-])NC1=O)C(C)C)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCCN)C(=O)N[C@H](Cc1ccc(O)cc1)C(N)=O Show InChI InChI=1S/C57H78N14O11S2/c1-5-57(6-2)32-84-83-31-44(55(81)71-26-12-16-45(71)53(79)62-39(15-10-11-25-58)49(75)63-40(48(60)74)27-36-19-23-38(72)24-20-36)66-51(77)43(30-46(59)73)65-54(80)47(33(3)4)68-52(78)41(28-34-13-8-7-9-14-34)64-50(76)42(67-56(57)82)29-35-17-21-37(22-18-35)69-70-61/h7-9,13-14,17-24,33,39-45,47,72H,5-6,10-12,15-16,25-32,58H2,1-4H3,(H2,59,73)(H2,60,74)(H,62,79)(H,63,75)(H,64,76)(H,65,80)(H,66,77)(H,67,82)(H,68,78)/t39-,40+,41-,42-,43-,44-,45-,47-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | 2.60 | n/a | n/a | n/a | n/a | n/a |
University of Sherbrooke
Curated by ChEMBL
| Assay Description
Negative log of Kd for Vasopressin V1 receptor |
J Med Chem 35: 151-7 (1992)
BindingDB Entry DOI: 10.7270/Q2668DT9 |
More data for this
Ligand-Target Pair | |
Vasopressin V1a receptor
(RAT) | BDBM50008083
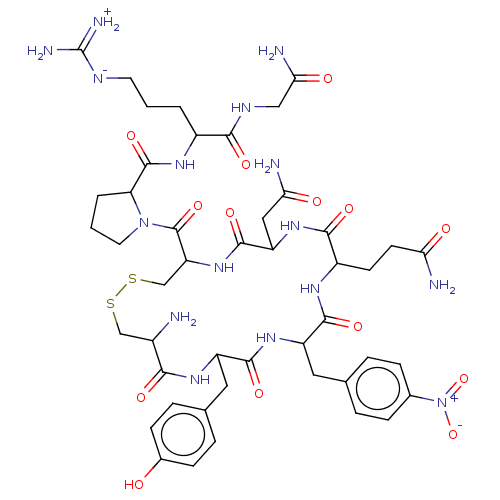
(CHEMBL2372418 | [2-({1-[19-Amino-10-(2-carbamoyl-e...)Show SMILES [#7]-[#6@H]-1-[#6]-[#16]-[#16]-[#6]-[#6@H](-[#7]-[#6](=O)-[#6@H](-[#6]-[#6](-[#7])=O)-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6](-[#7])=O)-[#7]-[#6](=O)-[#6@H](-[#6]-c2ccc(cc2)-[#7+](-[#8-])=O)-[#7]-[#6](=O)-[#6@@H](-[#6]-c2ccc(-[#8])cc2)-[#7]-[#6]-1=O)-[#6](=O)-[#7]-1-[#6]-[#6]-[#6]-[#6@H]-1-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](=O)-[#7]-[#6]-[#6](-[#7])=O Show InChI InChI=1S/C46H64N16O14S2/c47-27-21-77-78-22-33(45(74)61-16-2-4-34(61)44(73)56-28(3-1-15-53-46(51)52)39(68)54-20-37(50)66)60-43(72)32(19-36(49)65)59-40(69)29(13-14-35(48)64)55-41(70)31(17-23-5-9-25(10-6-23)62(75)76)58-42(71)30(57-38(27)67)18-24-7-11-26(63)12-8-24/h5-12,27-34,63H,1-4,13-22,47H2,(H2,48,64)(H2,49,65)(H2,50,66)(H,54,68)(H,55,70)(H,56,73)(H,57,67)(H,58,71)(H,59,69)(H,60,72)(H4,51,52,53)/t27-,28-,29-,30+,31-,32-,33-,34-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | 54 | n/a | n/a | n/a | n/a | n/a |
University of Sherbrooke
Curated by ChEMBL
| Assay Description
Negative log of Kd for Vasopressin V1 receptor |
J Med Chem 35: 151-7 (1992)
BindingDB Entry DOI: 10.7270/Q2668DT9 |
More data for this
Ligand-Target Pair | |
Vasopressin V1a receptor
(RAT) | BDBM50406623

(CHEMBL2115369)Show SMILES COc1ccc(C[C@@H]2NC(=O)C3(CCCC3)CSSC[C@H](NC(=O)[C@H](CC(N)=O)NC(=O)[C@@H](NC(=O)[C@H](Cc3ccccc3)NC2=O)C(C)C)C(=O)N2CCC[C@H]2C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](Cc2ccc(O)cc2)C(N)=O)cc1 Show InChI InChI=1S/C58H79N11O12S2/c1-34(2)48-55(78)65-44(31-47(60)71)52(75)66-45(56(79)69-27-11-15-46(69)54(77)62-40(14-7-10-26-59)50(73)63-41(49(61)72)28-36-16-20-38(70)21-17-36)32-82-83-33-58(24-8-9-25-58)57(80)67-43(30-37-18-22-39(81-3)23-19-37)51(74)64-42(53(76)68-48)29-35-12-5-4-6-13-35/h4-6,12-13,16-23,34,40-46,48,70H,7-11,14-15,24-33,59H2,1-3H3,(H2,60,71)(H2,61,72)(H,62,77)(H,63,73)(H,64,74)(H,65,78)(H,66,75)(H,67,80)(H,68,76)/t40-,41-,42-,43-,44-,45-,46-,48-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | 0.180 | n/a | n/a | n/a | n/a | n/a |
University of Sherbrooke
Curated by ChEMBL
| Assay Description
Binding potency against Vasopressin V1 receptor in rat liver cells. |
J Med Chem 35: 151-7 (1992)
BindingDB Entry DOI: 10.7270/Q2668DT9 |
More data for this
Ligand-Target Pair | |
Vasopressin V1a receptor
(RAT) | BDBM50406622

(CHEMBL2115364)Show SMILES CC(C)[C@@H]1NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@H](Cc2ccc(cc2)N=[N+]=[N-])NC(=O)C(C)(C)CSSC[C@H](NC(=O)[C@H](CC(N)=O)NC1=O)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](Cc1ccc(O)cc1)C(N)=O Show InChI InChI=1S/C55H74N14O11S2/c1-31(2)45-52(78)63-41(28-44(57)71)49(75)64-42(53(79)69-24-10-14-43(69)51(77)60-37(13-8-9-23-56)47(73)61-38(46(58)72)25-34-17-21-36(70)22-18-34)29-81-82-30-55(3,4)54(80)65-40(27-33-15-19-35(20-16-33)67-68-59)48(74)62-39(50(76)66-45)26-32-11-6-5-7-12-32/h5-7,11-12,15-22,31,37-43,45,70H,8-10,13-14,23-30,56H2,1-4H3,(H2,57,71)(H2,58,72)(H,60,77)(H,61,73)(H,62,74)(H,63,78)(H,64,75)(H,65,80)(H,66,76)/t37-,38-,39-,40-,41-,42-,43-,45-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | 0.603 | n/a | n/a | n/a | n/a | n/a |
University of Sherbrooke
Curated by ChEMBL
| Assay Description
Negative log of Kd for Vasopressin V1 receptor |
J Med Chem 35: 151-7 (1992)
BindingDB Entry DOI: 10.7270/Q2668DT9 |
More data for this
Ligand-Target Pair | |
Vasopressin V1a receptor
(RAT) | BDBM50453152

(CHEMBL2115365)Show SMILES CCC1(CC)CSSC[C@H](NC(=O)[C@H](CC(N)=O)NC(=O)[C@@H](NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@H](Cc2ccc(cc2)[N+]([O-])=O)NC1=O)C(C)C)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](Cc1ccc(O)cc1)C(N)=O Show InChI InChI=1S/C57H78N12O13S2/c1-5-57(6-2)32-84-83-31-44(55(79)68-26-12-16-45(68)53(77)61-39(15-10-11-25-58)49(73)62-40(48(60)72)27-36-19-23-38(70)24-20-36)65-51(75)43(30-46(59)71)64-54(78)47(33(3)4)67-52(76)41(28-34-13-8-7-9-14-34)63-50(74)42(66-56(57)80)29-35-17-21-37(22-18-35)69(81)82/h7-9,13-14,17-24,33,39-45,47,70H,5-6,10-12,15-16,25-32,58H2,1-4H3,(H2,59,71)(H2,60,72)(H,61,77)(H,62,73)(H,63,74)(H,64,78)(H,65,75)(H,66,80)(H,67,76)/t39-,40-,41-,42-,43-,44-,45-,47-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | 2.63 | n/a | n/a | n/a | n/a | n/a |
University of Sherbrooke
Curated by ChEMBL
| Assay Description
Negative log of Kd for Vasopressin V1 receptor |
J Med Chem 35: 151-7 (1992)
BindingDB Entry DOI: 10.7270/Q2668DT9 |
More data for this
Ligand-Target Pair | |
Vasopressin V1a receptor
(RAT) | BDBM50453152

(CHEMBL2115365)Show SMILES CCC1(CC)CSSC[C@H](NC(=O)[C@H](CC(N)=O)NC(=O)[C@@H](NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@H](Cc2ccc(cc2)[N+]([O-])=O)NC1=O)C(C)C)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](Cc1ccc(O)cc1)C(N)=O Show InChI InChI=1S/C57H78N12O13S2/c1-5-57(6-2)32-84-83-31-44(55(79)68-26-12-16-45(68)53(77)61-39(15-10-11-25-58)49(73)62-40(48(60)72)27-36-19-23-38(70)24-20-36)65-51(75)43(30-46(59)71)64-54(78)47(33(3)4)67-52(76)41(28-34-13-8-7-9-14-34)63-50(74)42(66-56(57)80)29-35-17-21-37(22-18-35)69(81)82/h7-9,13-14,17-24,33,39-45,47,70H,5-6,10-12,15-16,25-32,58H2,1-4H3,(H2,59,71)(H2,60,72)(H,61,77)(H,62,73)(H,63,74)(H,64,78)(H,65,75)(H,66,80)(H,67,76)/t39-,40-,41-,42-,43-,44-,45-,47-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | 2.60 | n/a | n/a | n/a | n/a | n/a |
University of Sherbrooke
Curated by ChEMBL
| Assay Description
Binding potency against Vasopressin V1 receptor in rat liver cells. |
J Med Chem 35: 151-7 (1992)
BindingDB Entry DOI: 10.7270/Q2668DT9 |
More data for this
Ligand-Target Pair | |
Vasopressin V2 receptor
(Sus scrofa) | BDBM50406622

(CHEMBL2115364)Show SMILES CC(C)[C@@H]1NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@H](Cc2ccc(cc2)N=[N+]=[N-])NC(=O)C(C)(C)CSSC[C@H](NC(=O)[C@H](CC(N)=O)NC1=O)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](Cc1ccc(O)cc1)C(N)=O Show InChI InChI=1S/C55H74N14O11S2/c1-31(2)45-52(78)63-41(28-44(57)71)49(75)64-42(53(79)69-24-10-14-43(69)51(77)60-37(13-8-9-23-56)47(73)61-38(46(58)72)25-34-17-21-36(70)22-18-34)29-81-82-30-55(3,4)54(80)65-40(27-33-15-19-35(20-16-33)67-68-59)48(74)62-39(50(76)66-45)26-32-11-6-5-7-12-32/h5-7,11-12,15-22,31,37-43,45,70H,8-10,13-14,23-30,56H2,1-4H3,(H2,57,71)(H2,58,72)(H,60,77)(H,61,73)(H,62,74)(H,63,78)(H,64,75)(H,65,80)(H,66,76)/t37-,38-,39-,40-,41-,42-,43-,45-/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | 100 | n/a | n/a | n/a | n/a | n/a |
University of Sherbrooke
Curated by ChEMBL
| Assay Description
Binding affinity towards V2-receptor in LLCPK cells |
J Med Chem 35: 151-7 (1992)
BindingDB Entry DOI: 10.7270/Q2668DT9 |
More data for this
Ligand-Target Pair | |
Vasopressin V1a receptor
(RAT) | BDBM50406623

(CHEMBL2115369)Show SMILES COc1ccc(C[C@@H]2NC(=O)C3(CCCC3)CSSC[C@H](NC(=O)[C@H](CC(N)=O)NC(=O)[C@@H](NC(=O)[C@H](Cc3ccccc3)NC2=O)C(C)C)C(=O)N2CCC[C@H]2C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](Cc2ccc(O)cc2)C(N)=O)cc1 Show InChI InChI=1S/C58H79N11O12S2/c1-34(2)48-55(78)65-44(31-47(60)71)52(75)66-45(56(79)69-27-11-15-46(69)54(77)62-40(14-7-10-26-59)50(73)63-41(49(61)72)28-36-16-20-38(70)21-17-36)32-82-83-33-58(24-8-9-25-58)57(80)67-43(30-37-18-22-39(81-3)23-19-37)51(74)64-42(53(76)68-48)29-35-12-5-4-6-13-35/h4-6,12-13,16-23,34,40-46,48,70H,7-11,14-15,24-33,59H2,1-3H3,(H2,60,71)(H2,61,72)(H,62,77)(H,63,73)(H,64,74)(H,65,78)(H,66,75)(H,67,80)(H,68,76)/t40-,41-,42-,43-,44-,45-,46-,48-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | 0.182 | n/a | n/a | n/a | n/a | n/a |
University of Sherbrooke
Curated by ChEMBL
| Assay Description
Binding potency against Vasopressin V1 receptor in rat liver cells. |
J Med Chem 35: 151-7 (1992)
BindingDB Entry DOI: 10.7270/Q2668DT9 |
More data for this
Ligand-Target Pair | |
Vasopressin V1a receptor
(RAT) | BDBM35667
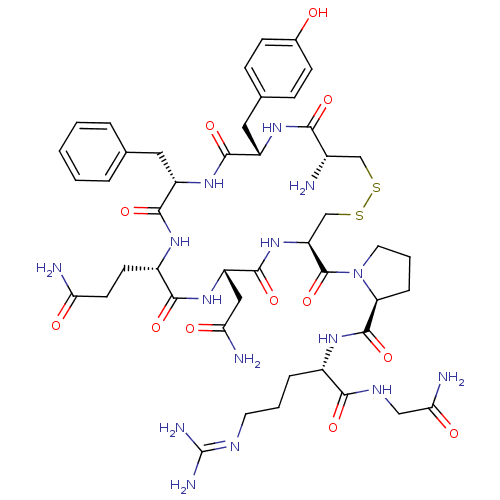
(AVP | CHEMBL373742 | US10131692, 44 (AVP) | [3H]Ar...)Show SMILES [#7]-[#6@H]-1-[#6]-[#16]-[#16]-[#6]-[#6@H](-[#7]-[#6](=O)-[#6@H](-[#6]-[#6](-[#7])=O)-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6](-[#7])=O)-[#7]-[#6](=O)-[#6@H](-[#6]-c2ccccc2)-[#7]-[#6](=O)-[#6@H](-[#6]-c2ccc(-[#8])cc2)-[#7]-[#6]-1=O)-[#6](=O)-[#7]-1-[#6]-[#6]-[#6]-[#6@H]-1-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6](=O)-[#7]-[#6]-[#6](-[#7])=O Show InChI InChI=1S/C46H65N15O12S2/c47-27-22-74-75-23-33(45(73)61-17-5-9-34(61)44(72)56-28(8-4-16-53-46(51)52)39(67)54-21-37(50)65)60-43(71)32(20-36(49)64)59-40(68)29(14-15-35(48)63)55-41(69)31(18-24-6-2-1-3-7-24)58-42(70)30(57-38(27)66)19-25-10-12-26(62)13-11-25/h1-3,6-7,10-13,27-34,62H,4-5,8-9,14-23,47H2,(H2,48,63)(H2,49,64)(H2,50,65)(H,54,67)(H,55,69)(H,56,72)(H,57,66)(H,58,70)(H,59,68)(H,60,71)(H4,51,52,53)/t27-,28-,29-,30-,31-,32-,33-,34-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MMDB
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | n/a | 0.646 | n/a | n/a | n/a | n/a | n/a |
University of Sherbrooke
Curated by ChEMBL
| Assay Description
Negative log of Kd for Vasopressin V1 receptor |
J Med Chem 35: 151-7 (1992)
BindingDB Entry DOI: 10.7270/Q2668DT9 |
More data for this
Ligand-Target Pair | |
Vasopressin V1a receptor
(RAT) | BDBM50008096
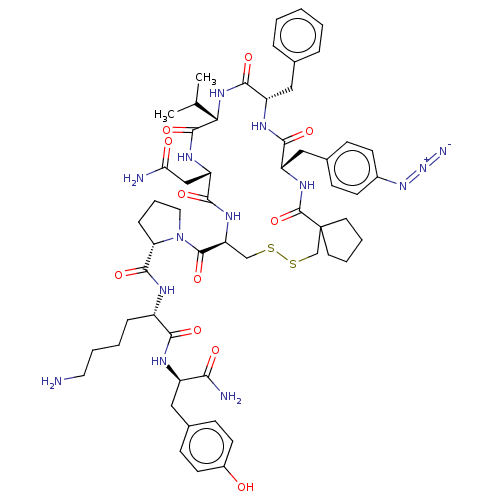
(CHEMBL2372414 | cyclo(Ppa-Phe(N3)-Phe-Val-Asn-Cys)...)Show SMILES CC(C)[C@@H]1NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@H](Cc2ccc(cc2)N=[N+]=[N-])NC(=O)C2(CCCC2)CSSC[C@H](NC(=O)[C@H](CC(N)=O)NC1=O)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCCN)C(=O)N[C@H](Cc1ccc(O)cc1)C(N)=O Show InChI InChI=1S/C57H76N14O11S2/c1-33(2)47-54(80)65-43(30-46(59)73)51(77)66-44(55(81)71-26-10-14-45(71)53(79)62-39(13-6-9-25-58)49(75)63-40(48(60)74)27-36-17-21-38(72)22-18-36)31-83-84-32-57(23-7-8-24-57)56(82)67-42(29-35-15-19-37(20-16-35)69-70-61)50(76)64-41(52(78)68-47)28-34-11-4-3-5-12-34/h3-5,11-12,15-22,33,39-45,47,72H,6-10,13-14,23-32,58H2,1-2H3,(H2,59,73)(H2,60,74)(H,62,79)(H,63,75)(H,64,76)(H,65,80)(H,66,77)(H,67,82)(H,68,78)/t39-,40+,41-,42-,43-,44-,45-,47-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | 36 | n/a | n/a | n/a | n/a | n/a |
University of Sherbrooke
Curated by ChEMBL
| Assay Description
Binding potency against Vasopressin V1 receptor in rat liver cells. |
J Med Chem 35: 151-7 (1992)
BindingDB Entry DOI: 10.7270/Q2668DT9 |
More data for this
Ligand-Target Pair | |
Vasopressin V1a receptor
(RAT) | BDBM50008096

(CHEMBL2372414 | cyclo(Ppa-Phe(N3)-Phe-Val-Asn-Cys)...)Show SMILES CC(C)[C@@H]1NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@H](Cc2ccc(cc2)N=[N+]=[N-])NC(=O)C2(CCCC2)CSSC[C@H](NC(=O)[C@H](CC(N)=O)NC1=O)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCCN)C(=O)N[C@H](Cc1ccc(O)cc1)C(N)=O Show InChI InChI=1S/C57H76N14O11S2/c1-33(2)47-54(80)65-43(30-46(59)73)51(77)66-44(55(81)71-26-10-14-45(71)53(79)62-39(13-6-9-25-58)49(75)63-40(48(60)74)27-36-17-21-38(72)22-18-36)31-83-84-32-57(23-7-8-24-57)56(82)67-42(29-35-15-19-37(20-16-35)69-70-61)50(76)64-41(52(78)68-47)28-34-11-4-3-5-12-34/h3-5,11-12,15-22,33,39-45,47,72H,6-10,13-14,23-32,58H2,1-2H3,(H2,59,73)(H2,60,74)(H,62,79)(H,63,75)(H,64,76)(H,65,80)(H,66,77)(H,67,82)(H,68,78)/t39-,40+,41-,42-,43-,44-,45-,47-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | 35 | n/a | n/a | n/a | n/a | n/a |
University of Sherbrooke
Curated by ChEMBL
| Assay Description
Negative log of Kd for Vasopressin V1 receptor |
J Med Chem 35: 151-7 (1992)
BindingDB Entry DOI: 10.7270/Q2668DT9 |
More data for this
Ligand-Target Pair | |
Vasopressin V1a receptor
(RAT) | BDBM50008084
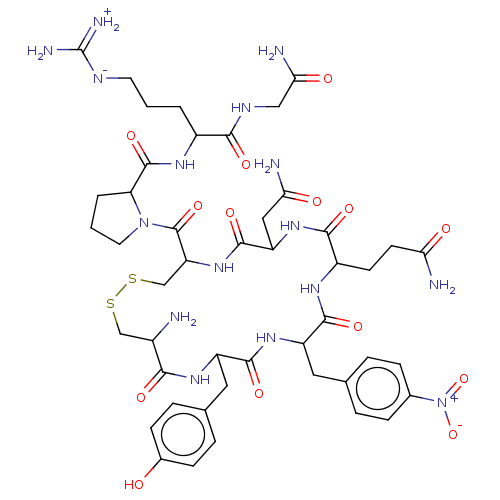
(1-[13-Benzyl-7-carbamoylmethyl-10-isopropyl-16-(4-...)Show SMILES [#7]-[#6@H]-1-[#6]-[#16]-[#16]-[#6]-[#6@H](-[#7]-[#6](=O)-[#6@H](-[#6]-[#6](-[#7])=O)-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6](-[#7])=O)-[#7]-[#6](=O)-[#6@H](-[#6]-c2ccc(cc2)-[#7+](-[#8-])=O)-[#7]-[#6](=O)-[#6@H](-[#6]-c2ccc(-[#8])cc2)-[#7]-[#6]-1=O)-[#6](=O)-[#7]-1-[#6]-[#6]-[#6]-[#6@H]-1-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](=O)-[#7]-[#6]-[#6](-[#7])=O |r| Show InChI InChI=1S/C46H64N16O14S2/c47-27-21-77-78-22-33(45(74)61-16-2-4-34(61)44(73)56-28(3-1-15-53-46(51)52)39(68)54-20-37(50)66)60-43(72)32(19-36(49)65)59-40(69)29(13-14-35(48)64)55-41(70)31(17-23-5-9-25(10-6-23)62(75)76)58-42(71)30(57-38(27)67)18-24-7-11-26(63)12-8-24/h5-12,27-34,63H,1-4,13-22,47H2,(H2,48,64)(H2,49,65)(H2,50,66)(H,54,68)(H,55,70)(H,56,73)(H,57,67)(H,58,71)(H,59,69)(H,60,72)(H4,51,52,53)/t27-,28-,29-,30-,31-,32-,33-,34-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | 41 | n/a | n/a | n/a | n/a | n/a |
University of Sherbrooke
Curated by ChEMBL
| Assay Description
Negative log of Kd for Vasopressin V1 receptor |
J Med Chem 35: 151-7 (1992)
BindingDB Entry DOI: 10.7270/Q2668DT9 |
More data for this
Ligand-Target Pair | |
Vasopressin V1a receptor
(RAT) | BDBM50008088

(CHEMBL2372416 | cyclo(Depa-Phe(N3)-Phe-Val-Asn-Cys...)Show SMILES CCC1(CC)CSSC[C@H](NC(=O)[C@H](CC(N)=O)NC(=O)[C@@H](NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@H](Cc2ccc(cc2)N=[N+]=[N-])NC1=O)C(C)C)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCCN)C(=O)N[C@H](Cc1ccc(O)cc1)C(N)=O Show InChI InChI=1S/C57H78N14O11S2/c1-5-57(6-2)32-84-83-31-44(55(81)71-26-12-16-45(71)53(79)62-39(15-10-11-25-58)49(75)63-40(48(60)74)27-36-19-23-38(72)24-20-36)66-51(77)43(30-46(59)73)65-54(80)47(33(3)4)68-52(78)41(28-34-13-8-7-9-14-34)64-50(76)42(67-56(57)82)29-35-17-21-37(22-18-35)69-70-61/h7-9,13-14,17-24,33,39-45,47,72H,5-6,10-12,15-16,25-32,58H2,1-4H3,(H2,59,73)(H2,60,74)(H,62,79)(H,63,75)(H,64,76)(H,65,80)(H,66,77)(H,67,82)(H,68,78)/t39-,40+,41-,42-,43-,44-,45-,47-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | 2.60 | n/a | n/a | n/a | n/a | n/a |
University of Sherbrooke
Curated by ChEMBL
| Assay Description
Negative log of Kd for Vasopressin V1 receptor |
J Med Chem 35: 151-7 (1992)
BindingDB Entry DOI: 10.7270/Q2668DT9 |
More data for this
Ligand-Target Pair | |
Vasopressin V1a receptor
(RAT) | BDBM50008097

(CHEMBL2028983 | [2-({1-[19-Amino-13-(4-azido-benzy...)Show SMILES [#6]-[#6@H](-[#7](-[#6])-[#6](=O)-[#6@H]-1-[#6]-[#16]-[#16]-[#6]C2([#6]-[#6]-[#6]-[#6]2)[#6](=O)-[#7]-[#6@@H](-[#6]-c2ccc(-[#8])cc2)-[#6](=O)-[#7]-[#6@@H](-[#6]-c2ccccc2)-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6](-[#7])=O)-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#7])=O)-[#6](=O)-[#7]-1)-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-c1ccc(cc1)-[#7]=[N+]=[#7-])-[#6](-[#7])=O |r| Show InChI InChI=1S/C60H83N19O12S2/c1-34(51(84)71-41(14-10-28-69-59(65)66)52(85)70-40(50(64)83)13-6-9-27-68-49(63)37-17-19-38(20-18-37)77-78-67)79(2)57(90)46-32-92-93-33-60(25-7-8-26-60)58(91)76-44(30-36-15-21-39(80)22-16-36)55(88)73-43(29-35-11-4-3-5-12-35)54(87)72-42(23-24-47(61)81)53(86)74-45(31-48(62)82)56(89)75-46/h3-5,11-12,15-22,34,40-46,80H,6-10,13-14,23-33H2,1-2H3,(H2,61,81)(H2,62,82)(H2,63,68)(H2,64,83)(H,70,85)(H,71,84)(H,72,87)(H,73,88)(H,74,86)(H,75,89)(H,76,91)(H4,65,66,69)/t34-,40-,41-,42-,43-,44-,45-,46+/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | 0.330 | n/a | n/a | n/a | n/a | n/a |
University of Sherbrooke
Curated by ChEMBL
| Assay Description
Binding potency against Vasopressin V1 receptor in rat liver cells. |
J Med Chem 35: 151-7 (1992)
BindingDB Entry DOI: 10.7270/Q2668DT9 |
More data for this
Ligand-Target Pair | |
Vasopressin V1a receptor
(RAT) | BDBM50453149
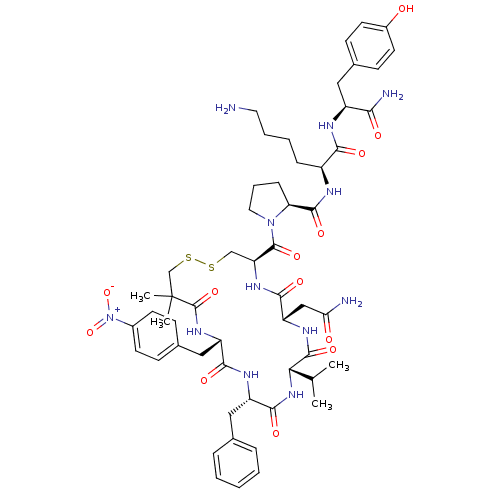
(CHEMBL2115368)Show SMILES CC(C)[C@@H]1NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@H](Cc2ccc(cc2)[N+]([O-])=O)NC(=O)C(C)(C)CSSC[C@H](NC(=O)[C@H](CC(N)=O)NC1=O)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](Cc1ccc(O)cc1)C(N)=O Show InChI InChI=1S/C55H74N12O13S2/c1-31(2)45-52(76)62-41(28-44(57)69)49(73)63-42(53(77)66-24-10-14-43(66)51(75)59-37(13-8-9-23-56)47(71)60-38(46(58)70)25-34-17-21-36(68)22-18-34)29-81-82-30-55(3,4)54(78)64-40(27-33-15-19-35(20-16-33)67(79)80)48(72)61-39(50(74)65-45)26-32-11-6-5-7-12-32/h5-7,11-12,15-22,31,37-43,45,68H,8-10,13-14,23-30,56H2,1-4H3,(H2,57,69)(H2,58,70)(H,59,75)(H,60,71)(H,61,72)(H,62,76)(H,63,73)(H,64,78)(H,65,74)/t37-,38-,39-,40-,41-,42-,43-,45-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | 1.70 | n/a | n/a | n/a | n/a | n/a |
University of Sherbrooke
Curated by ChEMBL
| Assay Description
Negative log of Kd for Vasopressin V1 receptor |
J Med Chem 35: 151-7 (1992)
BindingDB Entry DOI: 10.7270/Q2668DT9 |
More data for this
Ligand-Target Pair | |
Vasopressin V1a receptor
(RAT) | BDBM50453152

(CHEMBL2115365)Show SMILES CCC1(CC)CSSC[C@H](NC(=O)[C@H](CC(N)=O)NC(=O)[C@@H](NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@H](Cc2ccc(cc2)[N+]([O-])=O)NC1=O)C(C)C)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](Cc1ccc(O)cc1)C(N)=O Show InChI InChI=1S/C57H78N12O13S2/c1-5-57(6-2)32-84-83-31-44(55(79)68-26-12-16-45(68)53(77)61-39(15-10-11-25-58)49(73)62-40(48(60)72)27-36-19-23-38(70)24-20-36)65-51(75)43(30-46(59)71)64-54(78)47(33(3)4)67-52(76)41(28-34-13-8-7-9-14-34)63-50(74)42(66-56(57)80)29-35-17-21-37(22-18-35)69(81)82/h7-9,13-14,17-24,33,39-45,47,70H,5-6,10-12,15-16,25-32,58H2,1-4H3,(H2,59,71)(H2,60,72)(H,61,77)(H,62,73)(H,63,74)(H,64,78)(H,65,75)(H,66,80)(H,67,76)/t39-,40-,41-,42-,43-,44-,45-,47-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | 2.64 | n/a | n/a | n/a | n/a | n/a |
University of Sherbrooke
Curated by ChEMBL
| Assay Description
Binding potency against Vasopressin V1 receptor in rat liver cells. |
J Med Chem 35: 151-7 (1992)
BindingDB Entry DOI: 10.7270/Q2668DT9 |
More data for this
Ligand-Target Pair | |
Vasopressin V1a receptor
(RAT) | BDBM50008097

(CHEMBL2028983 | [2-({1-[19-Amino-13-(4-azido-benzy...)Show SMILES [#6]-[#6@H](-[#7](-[#6])-[#6](=O)-[#6@H]-1-[#6]-[#16]-[#16]-[#6]C2([#6]-[#6]-[#6]-[#6]2)[#6](=O)-[#7]-[#6@@H](-[#6]-c2ccc(-[#8])cc2)-[#6](=O)-[#7]-[#6@@H](-[#6]-c2ccccc2)-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6](-[#7])=O)-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#7])=O)-[#6](=O)-[#7]-1)-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-c1ccc(cc1)-[#7]=[N+]=[#7-])-[#6](-[#7])=O |r| Show InChI InChI=1S/C60H83N19O12S2/c1-34(51(84)71-41(14-10-28-69-59(65)66)52(85)70-40(50(64)83)13-6-9-27-68-49(63)37-17-19-38(20-18-37)77-78-67)79(2)57(90)46-32-92-93-33-60(25-7-8-26-60)58(91)76-44(30-36-15-21-39(80)22-16-36)55(88)73-43(29-35-11-4-3-5-12-35)54(87)72-42(23-24-47(61)81)53(86)74-45(31-48(62)82)56(89)75-46/h3-5,11-12,15-22,34,40-46,80H,6-10,13-14,23-33H2,1-2H3,(H2,61,81)(H2,62,82)(H2,63,68)(H2,64,83)(H,70,85)(H,71,84)(H,72,87)(H,73,88)(H,74,86)(H,75,89)(H,76,91)(H4,65,66,69)/t34-,40-,41-,42-,43-,44-,45-,46+/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | 0.331 | n/a | n/a | n/a | n/a | n/a |
University of Sherbrooke
Curated by ChEMBL
| Assay Description
Binding potency against Vasopressin V1 receptor in rat liver cells. |
J Med Chem 35: 151-7 (1992)
BindingDB Entry DOI: 10.7270/Q2668DT9 |
More data for this
Ligand-Target Pair | |
Vasopressin V1a receptor
(RAT) | BDBM35667

(AVP | CHEMBL373742 | US10131692, 44 (AVP) | [3H]Ar...)Show SMILES [#7]-[#6@H]-1-[#6]-[#16]-[#16]-[#6]-[#6@H](-[#7]-[#6](=O)-[#6@H](-[#6]-[#6](-[#7])=O)-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6](-[#7])=O)-[#7]-[#6](=O)-[#6@H](-[#6]-c2ccccc2)-[#7]-[#6](=O)-[#6@H](-[#6]-c2ccc(-[#8])cc2)-[#7]-[#6]-1=O)-[#6](=O)-[#7]-1-[#6]-[#6]-[#6]-[#6@H]-1-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6](=O)-[#7]-[#6]-[#6](-[#7])=O Show InChI InChI=1S/C46H65N15O12S2/c47-27-22-74-75-23-33(45(73)61-17-5-9-34(61)44(72)56-28(8-4-16-53-46(51)52)39(67)54-21-37(50)65)60-43(71)32(20-36(49)64)59-40(68)29(14-15-35(48)63)55-41(69)31(18-24-6-2-1-3-7-24)58-42(70)30(57-38(27)66)19-25-10-12-26(62)13-11-25/h1-3,6-7,10-13,27-34,62H,4-5,8-9,14-23,47H2,(H2,48,63)(H2,49,64)(H2,50,65)(H,54,67)(H,55,69)(H,56,72)(H,57,66)(H,58,70)(H,59,68)(H,60,71)(H4,51,52,53)/t27-,28-,29-,30-,31-,32-,33-,34-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MMDB
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | n/a | 0.640 | n/a | n/a | n/a | n/a | n/a |
University of Sherbrooke
Curated by ChEMBL
| Assay Description
Binding potency against V1 receptor in rat liver cells |
J Med Chem 35: 151-7 (1992)
BindingDB Entry DOI: 10.7270/Q2668DT9 |
More data for this
Ligand-Target Pair | |
Vasopressin V1a receptor
(RAT) | BDBM50008097

(CHEMBL2028983 | [2-({1-[19-Amino-13-(4-azido-benzy...)Show SMILES [#6]-[#6@H](-[#7](-[#6])-[#6](=O)-[#6@H]-1-[#6]-[#16]-[#16]-[#6]C2([#6]-[#6]-[#6]-[#6]2)[#6](=O)-[#7]-[#6@@H](-[#6]-c2ccc(-[#8])cc2)-[#6](=O)-[#7]-[#6@@H](-[#6]-c2ccccc2)-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6](-[#7])=O)-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#7])=O)-[#6](=O)-[#7]-1)-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-c1ccc(cc1)-[#7]=[N+]=[#7-])-[#6](-[#7])=O |r| Show InChI InChI=1S/C60H83N19O12S2/c1-34(51(84)71-41(14-10-28-69-59(65)66)52(85)70-40(50(64)83)13-6-9-27-68-49(63)37-17-19-38(20-18-37)77-78-67)79(2)57(90)46-32-92-93-33-60(25-7-8-26-60)58(91)76-44(30-36-15-21-39(80)22-16-36)55(88)73-43(29-35-11-4-3-5-12-35)54(87)72-42(23-24-47(61)81)53(86)74-45(31-48(62)82)56(89)75-46/h3-5,11-12,15-22,34,40-46,80H,6-10,13-14,23-33H2,1-2H3,(H2,61,81)(H2,62,82)(H2,63,68)(H2,64,83)(H,70,85)(H,71,84)(H,72,87)(H,73,88)(H,74,86)(H,75,89)(H,76,91)(H4,65,66,69)/t34-,40-,41-,42-,43-,44-,45-,46+/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | 0.331 | n/a | n/a | n/a | n/a | n/a |
University of Sherbrooke
Curated by ChEMBL
| Assay Description
Negative log of Kd for Vasopressin V1 receptor |
J Med Chem 35: 151-7 (1992)
BindingDB Entry DOI: 10.7270/Q2668DT9 |
More data for this
Ligand-Target Pair | |
Vasopressin V1a receptor
(RAT) | BDBM50406622

(CHEMBL2115364)Show SMILES CC(C)[C@@H]1NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@H](Cc2ccc(cc2)N=[N+]=[N-])NC(=O)C(C)(C)CSSC[C@H](NC(=O)[C@H](CC(N)=O)NC1=O)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](Cc1ccc(O)cc1)C(N)=O Show InChI InChI=1S/C55H74N14O11S2/c1-31(2)45-52(78)63-41(28-44(57)71)49(75)64-42(53(79)69-24-10-14-43(69)51(77)60-37(13-8-9-23-56)47(73)61-38(46(58)72)25-34-17-21-36(70)22-18-34)29-81-82-30-55(3,4)54(80)65-40(27-33-15-19-35(20-16-33)67-68-59)48(74)62-39(50(76)66-45)26-32-11-6-5-7-12-32/h5-7,11-12,15-22,31,37-43,45,70H,8-10,13-14,23-30,56H2,1-4H3,(H2,57,71)(H2,58,72)(H,60,77)(H,61,73)(H,62,74)(H,63,78)(H,64,75)(H,65,80)(H,66,76)/t37-,38-,39-,40-,41-,42-,43-,45-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | 0.480 | n/a | n/a | n/a | n/a | n/a |
University of Sherbrooke
Curated by ChEMBL
| Assay Description
Inhibition of the 3 nM VP stimulation of phospholipase-C in cultured WRK cells was used as a V1-mediated assay |
J Med Chem 35: 151-7 (1992)
BindingDB Entry DOI: 10.7270/Q2668DT9 |
More data for this
Ligand-Target Pair | |
Vasopressin V1a receptor
(RAT) | BDBM50406622

(CHEMBL2115364)Show SMILES CC(C)[C@@H]1NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@H](Cc2ccc(cc2)N=[N+]=[N-])NC(=O)C(C)(C)CSSC[C@H](NC(=O)[C@H](CC(N)=O)NC1=O)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](Cc1ccc(O)cc1)C(N)=O Show InChI InChI=1S/C55H74N14O11S2/c1-31(2)45-52(78)63-41(28-44(57)71)49(75)64-42(53(79)69-24-10-14-43(69)51(77)60-37(13-8-9-23-56)47(73)61-38(46(58)72)25-34-17-21-36(70)22-18-34)29-81-82-30-55(3,4)54(80)65-40(27-33-15-19-35(20-16-33)67-68-59)48(74)62-39(50(76)66-45)26-32-11-6-5-7-12-32/h5-7,11-12,15-22,31,37-43,45,70H,8-10,13-14,23-30,56H2,1-4H3,(H2,57,71)(H2,58,72)(H,60,77)(H,61,73)(H,62,74)(H,63,78)(H,64,75)(H,65,80)(H,66,76)/t37-,38-,39-,40-,41-,42-,43-,45-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | 0.479 | n/a | n/a | n/a | n/a | n/a |
University of Sherbrooke
Curated by ChEMBL
| Assay Description
Negative log of Kd. |
J Med Chem 35: 151-7 (1992)
BindingDB Entry DOI: 10.7270/Q2668DT9 |
More data for this
Ligand-Target Pair | |
Vasopressin V1a receptor
(RAT) | BDBM50453150

(CHEMBL2372420)Show SMILES CC(C)[C@@H]1NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@H](Cc2ccc(cc2)[N+]([O-])=O)NC(=O)CCSSC[C@H](NC(=O)[C@H](CC(N)=O)NC1=O)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCCN)C(=O)N[C@H](Cc1ccc(O)cc1)C(N)=O Show InChI InChI=1S/C53H70N12O13S2/c1-30(2)45-52(75)61-40(28-43(55)67)49(72)62-41(53(76)64-23-8-12-42(64)51(74)58-36(11-6-7-22-54)47(70)59-37(46(56)69)25-33-15-19-35(66)20-16-33)29-80-79-24-21-44(68)57-38(27-32-13-17-34(18-14-32)65(77)78)48(71)60-39(50(73)63-45)26-31-9-4-3-5-10-31/h3-5,9-10,13-20,30,36-42,45,66H,6-8,11-12,21-29,54H2,1-2H3,(H2,55,67)(H2,56,69)(H,57,68)(H,58,74)(H,59,70)(H,60,71)(H,61,75)(H,62,72)(H,63,73)/t36-,37+,38-,39-,40-,41-,42-,45-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | 27 | n/a | n/a | n/a | n/a | n/a |
University of Sherbrooke
Curated by ChEMBL
| Assay Description
Negative log of Kd. |
J Med Chem 35: 151-7 (1992)
BindingDB Entry DOI: 10.7270/Q2668DT9 |
More data for this
Ligand-Target Pair | |
Vasopressin V1a receptor
(RAT) | BDBM50453151
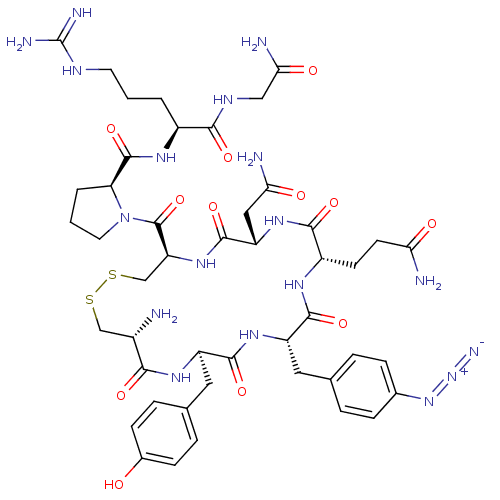
(CHEMBL2372421)Show SMILES [#7]-[#6@H]-1-[#6]-[#16]-[#16]-[#6]-[#6@H](-[#7]-[#6](=O)-[#6@H](-[#6]-[#6](-[#7])=O)-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6](-[#7])=O)-[#7]-[#6](=O)-[#6@H](-[#6]-c2ccc(cc2)-[#7]=[N+]=[#7-])-[#7]-[#6](=O)-[#6@@H](-[#6]-c2ccc(-[#8])cc2)-[#7]-[#6]-1=O)-[#6](=O)-[#7]-1-[#6]-[#6]-[#6]-[#6@H]-1-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](=O)-[#7]-[#6]-[#6](-[#7])=O Show InChI InChI=1S/C46H64N18O12S2/c47-27-21-77-78-22-33(45(76)64-16-2-4-34(64)44(75)57-28(3-1-15-54-46(51)52)39(70)55-20-37(50)68)61-43(74)32(19-36(49)67)60-40(71)29(13-14-35(48)66)56-41(72)31(17-23-5-9-25(10-6-23)62-63-53)59-42(73)30(58-38(27)69)18-24-7-11-26(65)12-8-24/h5-12,27-34,65H,1-4,13-22,47H2,(H2,48,66)(H2,49,67)(H2,50,68)(H,55,70)(H,56,72)(H,57,75)(H,58,69)(H,59,73)(H,60,71)(H,61,74)(H4,51,52,54)/t27-,28-,29-,30+,31-,32-,33-,34-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | 33 | n/a | n/a | n/a | n/a | n/a |
University of Sherbrooke
Curated by ChEMBL
| Assay Description
Binding potency against Vasopressin V1 receptor in rat liver cells. |
J Med Chem 35: 151-7 (1992)
BindingDB Entry DOI: 10.7270/Q2668DT9 |
More data for this
Ligand-Target Pair | |
Vasopressin V1a receptor
(RAT) | BDBM50008085

(CHEMBL2028984 | [2-({1-[19-Amino-13-(4-azido-benzy...)Show SMILES [#6]-[#6]-[#8]-c1ccc(-[#6]-[#6@@H](-[#7]-[#6](=O)-[#6]-c2ccncc2)-[#6](=O)-[#7]-[#6@@H](-[#6]-c2ccccc2)-[#6](=O)-[#7]-[#6@@H](-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#7])=O)-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6])-[#6](=O)-[#7]-2-[#6]-[#6]-[#6]-[#6@H]-2-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](-[#8])=O)cc1 |r| Show InChI InChI=1S/C57H82N16O12/c1-5-38(54(82)73-28-12-17-44(73)52(80)68-39(15-10-24-64-56(59)60)48(76)69-40(55(83)84)16-11-25-65-57(61)62)67-50(78)43(32-45(58)74)71-53(81)47(33(3)4)72-51(79)42(29-34-13-8-7-9-14-34)70-49(77)41(30-35-18-20-37(21-19-35)85-6-2)66-46(75)31-36-22-26-63-27-23-36/h7-9,13-14,18-23,26-27,33,38-44,47H,5-6,10-12,15-17,24-25,28-32H2,1-4H3,(H2,58,74)(H,66,75)(H,67,78)(H,68,80)(H,69,76)(H,70,77)(H,71,81)(H,72,79)(H,83,84)(H4,59,60,64)(H4,61,62,65)/t38-,39-,40-,41+,42-,43-,44-,47-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | 76 | n/a | n/a | n/a | n/a | n/a |
University of Sherbrooke
Curated by ChEMBL
| Assay Description
Negative log of Kd for Vasopressin V1 receptor |
J Med Chem 35: 151-7 (1992)
BindingDB Entry DOI: 10.7270/Q2668DT9 |
More data for this
Ligand-Target Pair | |
Vasopressin V1a receptor
(RAT) | BDBM50008097

(CHEMBL2028983 | [2-({1-[19-Amino-13-(4-azido-benzy...)Show SMILES [#6]-[#6@H](-[#7](-[#6])-[#6](=O)-[#6@H]-1-[#6]-[#16]-[#16]-[#6]C2([#6]-[#6]-[#6]-[#6]2)[#6](=O)-[#7]-[#6@@H](-[#6]-c2ccc(-[#8])cc2)-[#6](=O)-[#7]-[#6@@H](-[#6]-c2ccccc2)-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6](-[#7])=O)-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#7])=O)-[#6](=O)-[#7]-1)-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-c1ccc(cc1)-[#7]=[N+]=[#7-])-[#6](-[#7])=O |r| Show InChI InChI=1S/C60H83N19O12S2/c1-34(51(84)71-41(14-10-28-69-59(65)66)52(85)70-40(50(64)83)13-6-9-27-68-49(63)37-17-19-38(20-18-37)77-78-67)79(2)57(90)46-32-92-93-33-60(25-7-8-26-60)58(91)76-44(30-36-15-21-39(80)22-16-36)55(88)73-43(29-35-11-4-3-5-12-35)54(87)72-42(23-24-47(61)81)53(86)74-45(31-48(62)82)56(89)75-46/h3-5,11-12,15-22,34,40-46,80H,6-10,13-14,23-33H2,1-2H3,(H2,61,81)(H2,62,82)(H2,63,68)(H2,64,83)(H,70,85)(H,71,84)(H,72,87)(H,73,88)(H,74,86)(H,75,89)(H,76,91)(H4,65,66,69)/t34-,40-,41-,42-,43-,44-,45-,46+/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | 0.330 | n/a | n/a | n/a | n/a | n/a |
University of Sherbrooke
Curated by ChEMBL
| Assay Description
Binding potency against Vasopressin V1 receptor in rat liver cells. |
J Med Chem 35: 151-7 (1992)
BindingDB Entry DOI: 10.7270/Q2668DT9 |
More data for this
Ligand-Target Pair | |
Vasopressin V1a receptor
(RAT) | BDBM50406622

(CHEMBL2115364)Show SMILES CC(C)[C@@H]1NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@H](Cc2ccc(cc2)N=[N+]=[N-])NC(=O)C(C)(C)CSSC[C@H](NC(=O)[C@H](CC(N)=O)NC1=O)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](Cc1ccc(O)cc1)C(N)=O Show InChI InChI=1S/C55H74N14O11S2/c1-31(2)45-52(78)63-41(28-44(57)71)49(75)64-42(53(79)69-24-10-14-43(69)51(77)60-37(13-8-9-23-56)47(73)61-38(46(58)72)25-34-17-21-36(70)22-18-34)29-81-82-30-55(3,4)54(80)65-40(27-33-15-19-35(20-16-33)67-68-59)48(74)62-39(50(76)66-45)26-32-11-6-5-7-12-32/h5-7,11-12,15-22,31,37-43,45,70H,8-10,13-14,23-30,56H2,1-4H3,(H2,57,71)(H2,58,72)(H,60,77)(H,61,73)(H,62,74)(H,63,78)(H,64,75)(H,65,80)(H,66,76)/t37-,38-,39-,40-,41-,42-,43-,45-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | 0.600 | n/a | n/a | n/a | n/a | n/a |
University of Sherbrooke
Curated by ChEMBL
| Assay Description
Binding potency against Vasopressin V1 receptor in rat liver cells. |
J Med Chem 35: 151-7 (1992)
BindingDB Entry DOI: 10.7270/Q2668DT9 |
More data for this
Ligand-Target Pair | |
Vasopressin V2 receptor
(Sus scrofa) | BDBM50406623

(CHEMBL2115369)Show SMILES COc1ccc(C[C@@H]2NC(=O)C3(CCCC3)CSSC[C@H](NC(=O)[C@H](CC(N)=O)NC(=O)[C@@H](NC(=O)[C@H](Cc3ccccc3)NC2=O)C(C)C)C(=O)N2CCC[C@H]2C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](Cc2ccc(O)cc2)C(N)=O)cc1 Show InChI InChI=1S/C58H79N11O12S2/c1-34(2)48-55(78)65-44(31-47(60)71)52(75)66-45(56(79)69-27-11-15-46(69)54(77)62-40(14-7-10-26-59)50(73)63-41(49(61)72)28-36-16-20-38(70)21-17-36)32-82-83-33-58(24-8-9-25-58)57(80)67-43(30-37-18-22-39(81-3)23-19-37)51(74)64-42(53(76)68-48)29-35-12-5-4-6-13-35/h4-6,12-13,16-23,34,40-46,48,70H,7-11,14-15,24-33,59H2,1-3H3,(H2,60,71)(H2,61,72)(H,62,77)(H,63,73)(H,64,74)(H,65,78)(H,66,75)(H,67,80)(H,68,76)/t40-,41-,42-,43-,44-,45-,46-,48-/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | 100 | n/a | n/a | n/a | n/a | n/a |
University of Sherbrooke
Curated by ChEMBL
| Assay Description
Binding affinity towards Vasopressin V2 receptor in LLCPK cells |
J Med Chem 35: 151-7 (1992)
BindingDB Entry DOI: 10.7270/Q2668DT9 |
More data for this
Ligand-Target Pair | |
Vasopressin V1a receptor
(RAT) | BDBM50453149

(CHEMBL2115368)Show SMILES CC(C)[C@@H]1NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@H](Cc2ccc(cc2)[N+]([O-])=O)NC(=O)C(C)(C)CSSC[C@H](NC(=O)[C@H](CC(N)=O)NC1=O)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](Cc1ccc(O)cc1)C(N)=O Show InChI InChI=1S/C55H74N12O13S2/c1-31(2)45-52(76)62-41(28-44(57)69)49(73)63-42(53(77)66-24-10-14-43(66)51(75)59-37(13-8-9-23-56)47(71)60-38(46(58)70)25-34-17-21-36(68)22-18-34)29-81-82-30-55(3,4)54(78)64-40(27-33-15-19-35(20-16-33)67(79)80)48(72)61-39(50(74)65-45)26-32-11-6-5-7-12-32/h5-7,11-12,15-22,31,37-43,45,68H,8-10,13-14,23-30,56H2,1-4H3,(H2,57,69)(H2,58,70)(H,59,75)(H,60,71)(H,61,72)(H,62,76)(H,63,73)(H,64,78)(H,65,74)/t37-,38-,39-,40-,41-,42-,43-,45-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | 1.80 | n/a | n/a | n/a | n/a | n/a |
University of Sherbrooke
Curated by ChEMBL
| Assay Description
Negative log of Kd for Vasopressin V1 receptor |
J Med Chem 35: 151-7 (1992)
BindingDB Entry DOI: 10.7270/Q2668DT9 |
More data for this
Ligand-Target Pair | |
Vasopressin V1a receptor
(RAT) | BDBM50008083

(CHEMBL2372418 | [2-({1-[19-Amino-10-(2-carbamoyl-e...)Show SMILES [#7]-[#6@H]-1-[#6]-[#16]-[#16]-[#6]-[#6@H](-[#7]-[#6](=O)-[#6@H](-[#6]-[#6](-[#7])=O)-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6](-[#7])=O)-[#7]-[#6](=O)-[#6@H](-[#6]-c2ccc(cc2)-[#7+](-[#8-])=O)-[#7]-[#6](=O)-[#6@@H](-[#6]-c2ccc(-[#8])cc2)-[#7]-[#6]-1=O)-[#6](=O)-[#7]-1-[#6]-[#6]-[#6]-[#6@H]-1-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](=O)-[#7]-[#6]-[#6](-[#7])=O Show InChI InChI=1S/C46H64N16O14S2/c47-27-21-77-78-22-33(45(74)61-16-2-4-34(61)44(73)56-28(3-1-15-53-46(51)52)39(68)54-20-37(50)66)60-43(72)32(19-36(49)65)59-40(69)29(13-14-35(48)64)55-41(70)31(17-23-5-9-25(10-6-23)62(75)76)58-42(71)30(57-38(27)67)18-24-7-11-26(63)12-8-24/h5-12,27-34,63H,1-4,13-22,47H2,(H2,48,64)(H2,49,65)(H2,50,66)(H,54,68)(H,55,70)(H,56,73)(H,57,67)(H,58,71)(H,59,69)(H,60,72)(H4,51,52,53)/t27-,28-,29-,30+,31-,32-,33-,34-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | 54 | n/a | n/a | n/a | n/a | n/a |
University of Sherbrooke
Curated by ChEMBL
| Assay Description
Inhibition of the 3 nM VP stimulation of phospholipase-C in cultured WRK cells was used as a V1-mediated assay (Vasopressin V1 receptor) |
J Med Chem 35: 151-7 (1992)
BindingDB Entry DOI: 10.7270/Q2668DT9 |
More data for this
Ligand-Target Pair | |
Vasopressin V1a receptor
(RAT) | BDBM50008092

(CHEMBL2372415 | cyclo(Ppa-Phe(NO2)-Phe-Val-Asn-Cys...)Show SMILES CC(C)[C@@H]1NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@H](Cc2ccc(cc2)[N+]([O-])=O)NC(=O)C2(CCCC2)CSSC[C@H](NC(=O)[C@H](CC(N)=O)NC1=O)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCCN)C(=O)N[C@H](Cc1ccc(O)cc1)C(N)=O Show InChI InChI=1S/C57H76N12O13S2/c1-33(2)47-54(78)64-43(30-46(59)71)51(75)65-44(55(79)68-26-10-14-45(68)53(77)61-39(13-6-9-25-58)49(73)62-40(48(60)72)27-36-17-21-38(70)22-18-36)31-83-84-32-57(23-7-8-24-57)56(80)66-42(29-35-15-19-37(20-16-35)69(81)82)50(74)63-41(52(76)67-47)28-34-11-4-3-5-12-34/h3-5,11-12,15-22,33,39-45,47,70H,6-10,13-14,23-32,58H2,1-2H3,(H2,59,71)(H2,60,72)(H,61,77)(H,62,73)(H,63,74)(H,64,78)(H,65,75)(H,66,80)(H,67,76)/t39-,40+,41-,42-,43-,44-,45-,47-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | 3.20 | n/a | n/a | n/a | n/a | n/a |
University of Sherbrooke
Curated by ChEMBL
| Assay Description
Negative log of Kd for Vasopressin V1 receptor |
J Med Chem 35: 151-7 (1992)
BindingDB Entry DOI: 10.7270/Q2668DT9 |
More data for this
Ligand-Target Pair | |
Vasopressin V1a receptor
(RAT) | BDBM50453148

(CHEMBL2372422)Show SMILES [#7]-[#6@H]-1-[#6]-[#16]-[#16]-[#6]-[#6@H](-[#7]-[#6](=O)-[#6@H](-[#6]-[#6](-[#7])=O)-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6](-[#7])=O)-[#7]-[#6](=O)-[#6@H](-[#6]-c2ccc(cc2)-[#7]=[N+]=[#7-])-[#7]-[#6](=O)-[#6@H](-[#6]-c2ccc(-[#8])cc2)-[#7]-[#6]-1=O)-[#6](=O)-[#7]-1-[#6]-[#6]-[#6]-[#6@H]-1-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](=O)-[#7]-[#6]-[#6](-[#7])=O |r| Show InChI InChI=1S/C46H64N18O12S2/c47-27-21-77-78-22-33(45(76)64-16-2-4-34(64)44(75)57-28(3-1-15-54-46(51)52)39(70)55-20-37(50)68)61-43(74)32(19-36(49)67)60-40(71)29(13-14-35(48)66)56-41(72)31(17-23-5-9-25(10-6-23)62-63-53)59-42(73)30(58-38(27)69)18-24-7-11-26(65)12-8-24/h5-12,27-34,65H,1-4,13-22,47H2,(H2,48,66)(H2,49,67)(H2,50,68)(H,55,70)(H,56,72)(H,57,75)(H,58,69)(H,59,73)(H,60,71)(H,61,74)(H4,51,52,54)/t27-,28-,29-,30-,31-,32-,33-,34-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | 16 | n/a | n/a | n/a | n/a | n/a |
University of Sherbrooke
Curated by ChEMBL
| Assay Description
Negative log of Kd for Vasopressin V1 receptor |
J Med Chem 35: 151-7 (1992)
BindingDB Entry DOI: 10.7270/Q2668DT9 |
More data for this
Ligand-Target Pair | |
Vasopressin V1a receptor
(RAT) | BDBM50453150

(CHEMBL2372420)Show SMILES CC(C)[C@@H]1NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@H](Cc2ccc(cc2)[N+]([O-])=O)NC(=O)CCSSC[C@H](NC(=O)[C@H](CC(N)=O)NC1=O)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCCN)C(=O)N[C@H](Cc1ccc(O)cc1)C(N)=O Show InChI InChI=1S/C53H70N12O13S2/c1-30(2)45-52(75)61-40(28-43(55)67)49(72)62-41(53(76)64-23-8-12-42(64)51(74)58-36(11-6-7-22-54)47(70)59-37(46(56)69)25-33-15-19-35(66)20-16-33)29-80-79-24-21-44(68)57-38(27-32-13-17-34(18-14-32)65(77)78)48(71)60-39(50(73)63-45)26-31-9-4-3-5-10-31/h3-5,9-10,13-20,30,36-42,45,66H,6-8,11-12,21-29,54H2,1-2H3,(H2,55,67)(H2,56,69)(H,57,68)(H,58,74)(H,59,70)(H,60,71)(H,61,75)(H,62,72)(H,63,73)/t36-,37+,38-,39-,40-,41-,42-,45-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | 27 | n/a | n/a | n/a | n/a | n/a |
University of Sherbrooke
Curated by ChEMBL
| Assay Description
Binding potency against Vasopressin V1 receptor in rat liver cells. |
J Med Chem 35: 151-7 (1992)
BindingDB Entry DOI: 10.7270/Q2668DT9 |
More data for this
Ligand-Target Pair | |
Vasopressin V1a receptor
(RAT) | BDBM50453151

(CHEMBL2372421)Show SMILES [#7]-[#6@H]-1-[#6]-[#16]-[#16]-[#6]-[#6@H](-[#7]-[#6](=O)-[#6@H](-[#6]-[#6](-[#7])=O)-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6](-[#7])=O)-[#7]-[#6](=O)-[#6@H](-[#6]-c2ccc(cc2)-[#7]=[N+]=[#7-])-[#7]-[#6](=O)-[#6@@H](-[#6]-c2ccc(-[#8])cc2)-[#7]-[#6]-1=O)-[#6](=O)-[#7]-1-[#6]-[#6]-[#6]-[#6@H]-1-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](=O)-[#7]-[#6]-[#6](-[#7])=O Show InChI InChI=1S/C46H64N18O12S2/c47-27-21-77-78-22-33(45(76)64-16-2-4-34(64)44(75)57-28(3-1-15-54-46(51)52)39(70)55-20-37(50)68)61-43(74)32(19-36(49)67)60-40(71)29(13-14-35(48)66)56-41(72)31(17-23-5-9-25(10-6-23)62-63-53)59-42(73)30(58-38(27)69)18-24-7-11-26(65)12-8-24/h5-12,27-34,65H,1-4,13-22,47H2,(H2,48,66)(H2,49,67)(H2,50,68)(H,55,70)(H,56,72)(H,57,75)(H,58,69)(H,59,73)(H,60,71)(H,61,74)(H4,51,52,54)/t27-,28-,29-,30+,31-,32-,33-,34-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | 33 | n/a | n/a | n/a | n/a | n/a |
University of Sherbrooke
Curated by ChEMBL
| Assay Description
Binding potency against Vasopressin V1 receptor in rat liver cells. |
J Med Chem 35: 151-7 (1992)
BindingDB Entry DOI: 10.7270/Q2668DT9 |
More data for this
Ligand-Target Pair | |
Vasopressin V1a receptor
(RAT) | BDBM50453149

(CHEMBL2115368)Show SMILES CC(C)[C@@H]1NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@H](Cc2ccc(cc2)[N+]([O-])=O)NC(=O)C(C)(C)CSSC[C@H](NC(=O)[C@H](CC(N)=O)NC1=O)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](Cc1ccc(O)cc1)C(N)=O Show InChI InChI=1S/C55H74N12O13S2/c1-31(2)45-52(76)62-41(28-44(57)69)49(73)63-42(53(77)66-24-10-14-43(66)51(75)59-37(13-8-9-23-56)47(71)60-38(46(58)70)25-34-17-21-36(68)22-18-34)29-81-82-30-55(3,4)54(78)64-40(27-33-15-19-35(20-16-33)67(79)80)48(72)61-39(50(74)65-45)26-32-11-6-5-7-12-32/h5-7,11-12,15-22,31,37-43,45,68H,8-10,13-14,23-30,56H2,1-4H3,(H2,57,69)(H2,58,70)(H,59,75)(H,60,71)(H,61,72)(H,62,76)(H,63,73)(H,64,78)(H,65,74)/t37-,38-,39-,40-,41-,42-,43-,45-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | 1.80 | n/a | n/a | n/a | n/a | n/a |
University of Sherbrooke
Curated by ChEMBL
| Assay Description
Negative log of Kd for Vasopressin V1 receptor |
J Med Chem 35: 151-7 (1992)
BindingDB Entry DOI: 10.7270/Q2668DT9 |
More data for this
Ligand-Target Pair | |
Vasopressin V1a receptor
(RAT) | BDBM50453149

(CHEMBL2115368)Show SMILES CC(C)[C@@H]1NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@H](Cc2ccc(cc2)[N+]([O-])=O)NC(=O)C(C)(C)CSSC[C@H](NC(=O)[C@H](CC(N)=O)NC1=O)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](Cc1ccc(O)cc1)C(N)=O Show InChI InChI=1S/C55H74N12O13S2/c1-31(2)45-52(76)62-41(28-44(57)69)49(73)63-42(53(77)66-24-10-14-43(66)51(75)59-37(13-8-9-23-56)47(71)60-38(46(58)70)25-34-17-21-36(68)22-18-34)29-81-82-30-55(3,4)54(78)64-40(27-33-15-19-35(20-16-33)67(79)80)48(72)61-39(50(74)65-45)26-32-11-6-5-7-12-32/h5-7,11-12,15-22,31,37-43,45,68H,8-10,13-14,23-30,56H2,1-4H3,(H2,57,69)(H2,58,70)(H,59,75)(H,60,71)(H,61,72)(H,62,76)(H,63,73)(H,64,78)(H,65,74)/t37-,38-,39-,40-,41-,42-,43-,45-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | 1.80 | n/a | n/a | n/a | n/a | n/a |
University of Sherbrooke
Curated by ChEMBL
| Assay Description
Binding potency against V1 receptor in rat liver cells |
J Med Chem 35: 151-7 (1992)
BindingDB Entry DOI: 10.7270/Q2668DT9 |
More data for this
Ligand-Target Pair | |
Vasopressin V1a receptor
(RAT) | BDBM50008084

(1-[13-Benzyl-7-carbamoylmethyl-10-isopropyl-16-(4-...)Show SMILES [#7]-[#6@H]-1-[#6]-[#16]-[#16]-[#6]-[#6@H](-[#7]-[#6](=O)-[#6@H](-[#6]-[#6](-[#7])=O)-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6](-[#7])=O)-[#7]-[#6](=O)-[#6@H](-[#6]-c2ccc(cc2)-[#7+](-[#8-])=O)-[#7]-[#6](=O)-[#6@H](-[#6]-c2ccc(-[#8])cc2)-[#7]-[#6]-1=O)-[#6](=O)-[#7]-1-[#6]-[#6]-[#6]-[#6@H]-1-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](=O)-[#7]-[#6]-[#6](-[#7])=O |r| Show InChI InChI=1S/C46H64N16O14S2/c47-27-21-77-78-22-33(45(74)61-16-2-4-34(61)44(73)56-28(3-1-15-53-46(51)52)39(68)54-20-37(50)66)60-43(72)32(19-36(49)65)59-40(69)29(13-14-35(48)64)55-41(70)31(17-23-5-9-25(10-6-23)62(75)76)58-42(71)30(57-38(27)67)18-24-7-11-26(63)12-8-24/h5-12,27-34,63H,1-4,13-22,47H2,(H2,48,64)(H2,49,65)(H2,50,66)(H,54,68)(H,55,70)(H,56,73)(H,57,67)(H,58,71)(H,59,69)(H,60,72)(H4,51,52,53)/t27-,28-,29-,30-,31-,32-,33-,34-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | 41 | n/a | n/a | n/a | n/a | n/a |
University of Sherbrooke
Curated by ChEMBL
| Assay Description
Binding potency against Vasopressin V1 receptor in rat liver cells. |
J Med Chem 35: 151-7 (1992)
BindingDB Entry DOI: 10.7270/Q2668DT9 |
More data for this
Ligand-Target Pair | |
Vasopressin V1a receptor
(RAT) | BDBM50008085

(CHEMBL2028984 | [2-({1-[19-Amino-13-(4-azido-benzy...)Show SMILES [#6]-[#6]-[#8]-c1ccc(-[#6]-[#6@@H](-[#7]-[#6](=O)-[#6]-c2ccncc2)-[#6](=O)-[#7]-[#6@@H](-[#6]-c2ccccc2)-[#6](=O)-[#7]-[#6@@H](-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#7])=O)-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6])-[#6](=O)-[#7]-2-[#6]-[#6]-[#6]-[#6@H]-2-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](-[#8])=O)cc1 |r| Show InChI InChI=1S/C57H82N16O12/c1-5-38(54(82)73-28-12-17-44(73)52(80)68-39(15-10-24-64-56(59)60)48(76)69-40(55(83)84)16-11-25-65-57(61)62)67-50(78)43(32-45(58)74)71-53(81)47(33(3)4)72-51(79)42(29-34-13-8-7-9-14-34)70-49(77)41(30-35-18-20-37(21-19-35)85-6-2)66-46(75)31-36-22-26-63-27-23-36/h7-9,13-14,18-23,26-27,33,38-44,47H,5-6,10-12,15-17,24-25,28-32H2,1-4H3,(H2,58,74)(H,66,75)(H,67,78)(H,68,80)(H,69,76)(H,70,77)(H,71,81)(H,72,79)(H,83,84)(H4,59,60,64)(H4,61,62,65)/t38-,39-,40-,41+,42-,43-,44-,47-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | 75 | n/a | n/a | n/a | n/a | n/a |
University of Sherbrooke
Curated by ChEMBL
| Assay Description
Binding potency against Vasopressin V1 receptor in rat liver cells. |
J Med Chem 35: 151-7 (1992)
BindingDB Entry DOI: 10.7270/Q2668DT9 |
More data for this
Ligand-Target Pair | |
Vasopressin V1a receptor
(RAT) | BDBM50453152

(CHEMBL2115365)Show SMILES CCC1(CC)CSSC[C@H](NC(=O)[C@H](CC(N)=O)NC(=O)[C@@H](NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@H](Cc2ccc(cc2)[N+]([O-])=O)NC1=O)C(C)C)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](Cc1ccc(O)cc1)C(N)=O Show InChI InChI=1S/C57H78N12O13S2/c1-5-57(6-2)32-84-83-31-44(55(79)68-26-12-16-45(68)53(77)61-39(15-10-11-25-58)49(73)62-40(48(60)72)27-36-19-23-38(70)24-20-36)65-51(75)43(30-46(59)71)64-54(78)47(33(3)4)67-52(76)41(28-34-13-8-7-9-14-34)63-50(74)42(66-56(57)80)29-35-17-21-37(22-18-35)69(81)82/h7-9,13-14,17-24,33,39-45,47,70H,5-6,10-12,15-16,25-32,58H2,1-4H3,(H2,59,71)(H2,60,72)(H,61,77)(H,62,73)(H,63,74)(H,64,78)(H,65,75)(H,66,80)(H,67,76)/t39-,40-,41-,42-,43-,44-,45-,47-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | 2.60 | n/a | n/a | n/a | n/a | n/a |
University of Sherbrooke
Curated by ChEMBL
| Assay Description
Negative log of Kd for Vasopressin V1 receptor |
J Med Chem 35: 151-7 (1992)
BindingDB Entry DOI: 10.7270/Q2668DT9 |
More data for this
Ligand-Target Pair | |
Vasopressin V1a receptor
(RAT) | BDBM50008091

(1-[16-(4-Azido-benzyl)-13-benzyl-7-carbamoylmethyl...)Show SMILES CC(C)[C@@H]1NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@H](Cc2ccc(cc2)N=[N+]=[N-])NC(=O)CCSSC[C@H](NC(=O)[C@H](CC(N)=O)NC1=O)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCCN)C(=O)N[C@H](Cc1ccc(O)cc1)C(N)=O Show InChI InChI=1S/C53H70N14O11S2/c1-30(2)45-52(77)62-40(28-43(55)69)49(74)63-41(53(78)67-23-8-12-42(67)51(76)59-36(11-6-7-22-54)47(72)60-37(46(56)71)25-33-15-19-35(68)20-16-33)29-80-79-24-21-44(70)58-38(27-32-13-17-34(18-14-32)65-66-57)48(73)61-39(50(75)64-45)26-31-9-4-3-5-10-31/h3-5,9-10,13-20,30,36-42,45,68H,6-8,11-12,21-29,54H2,1-2H3,(H2,55,69)(H2,56,71)(H,58,70)(H,59,76)(H,60,72)(H,61,73)(H,62,77)(H,63,74)(H,64,75)/t36-,37+,38-,39-,40-,41-,42-,45-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | 4.90 | n/a | n/a | n/a | n/a | n/a |
University of Sherbrooke
Curated by ChEMBL
| Assay Description
Binding potency against Vasopressin V1 receptor in rat liver cells. |
J Med Chem 35: 151-7 (1992)
BindingDB Entry DOI: 10.7270/Q2668DT9 |
More data for this
Ligand-Target Pair | |
Vasopressin V1a receptor
(RAT) | BDBM50453148

(CHEMBL2372422)Show SMILES [#7]-[#6@H]-1-[#6]-[#16]-[#16]-[#6]-[#6@H](-[#7]-[#6](=O)-[#6@H](-[#6]-[#6](-[#7])=O)-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6](-[#7])=O)-[#7]-[#6](=O)-[#6@H](-[#6]-c2ccc(cc2)-[#7]=[N+]=[#7-])-[#7]-[#6](=O)-[#6@H](-[#6]-c2ccc(-[#8])cc2)-[#7]-[#6]-1=O)-[#6](=O)-[#7]-1-[#6]-[#6]-[#6]-[#6@H]-1-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](=O)-[#7]-[#6]-[#6](-[#7])=O |r| Show InChI InChI=1S/C46H64N18O12S2/c47-27-21-77-78-22-33(45(76)64-16-2-4-34(64)44(75)57-28(3-1-15-54-46(51)52)39(70)55-20-37(50)68)61-43(74)32(19-36(49)67)60-40(71)29(13-14-35(48)66)56-41(72)31(17-23-5-9-25(10-6-23)62-63-53)59-42(73)30(58-38(27)69)18-24-7-11-26(65)12-8-24/h5-12,27-34,65H,1-4,13-22,47H2,(H2,48,66)(H2,49,67)(H2,50,68)(H,55,70)(H,56,72)(H,57,75)(H,58,69)(H,59,73)(H,60,71)(H,61,74)(H4,51,52,54)/t27-,28-,29-,30-,31-,32-,33-,34-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | 16 | n/a | n/a | n/a | n/a | n/a |
University of Sherbrooke
Curated by ChEMBL
| Assay Description
Binding potency against Vasopressin V1 receptor in rat liver cells. |
J Med Chem 35: 151-7 (1992)
BindingDB Entry DOI: 10.7270/Q2668DT9 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data













































