| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 2C19 |
|---|
| Ligand | BDBM562067 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | Inhibition of CYP Enzyme Activity In Vitro |
|---|
| IC50 | 12000±n/a nM |
|---|
| Citation |  Peterson, RT; Asnani, A; Liu, Y; Zheng, B; Wang, Y Tricyclic compounds as Cyp1 inhibitors US Patent US11396516 Publication Date 7/26/2022 Peterson, RT; Asnani, A; Liu, Y; Zheng, B; Wang, Y Tricyclic compounds as Cyp1 inhibitors US Patent US11396516 Publication Date 7/26/2022 |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 2C19 |
|---|
| Name: | Cytochrome P450 2C19 |
|---|
| Synonyms: | (R)-limonene 6-monooxygenase | (S)-limonene 6-monooxygenase | (S)-limonene 7-monooxygenase | CP2CJ_HUMAN | CYP2C19 | CYPIIC17 | CYPIIC19 | Cytochrome P450 2C19 (CYP2C19) | Cytochrome P450 2C19 [I331V] | Cytochrome P450-11A | Cytochrome P450-254C | Fenbendazole monooxygenase (4'-hydroxylating) | Mephenytoin 4-hydroxylase | P450-11A | P450-254C |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 55935.47 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P33261 |
|---|
| Residue: | 490 |
|---|
| Sequence: | MDPFVVLVLCLSCLLLLSIWRQSSGRGKLPPGPTPLPVIGNILQIDIKDVSKSLTNLSKI
YGPVFTLYFGLERMVVLHGYEVVKEALIDLGEEFSGRGHFPLAERANRGFGIVFSNGKRW
KEIRRFSLMTLRNFGMGKRSIEDRVQEEARCLVEELRKTKASPCDPTFILGCAPCNVICS
IIFQKRFDYKDQQFLNLMEKLNENIRIVSTPWIQICNNFPTIIDYFPGTHNKLLKNLAFM
ESDILEKVKEHQESMDINNPRDFIDCFLIKMEKEKQNQQSEFTIENLVITAADLLGAGTE
TTSTTLRYALLLLLKHPEVTAKVQEEIERVIGRNRSPCMQDRGHMPYTDAVVHEVQRYID
LIPTSLPHAVTCDVKFRNYLIPKGTTILTSLTSVLHDNKEFPNPEMFDPRHFLDEGGNFK
KSNYFMPFSAGKRICVGEGLARMELFLFLTFILQNFNLKSLIDPKDLDTTPVVNGFASVP
PFYQLCFIPV
|
|
|
|---|
| BDBM562067 |
|---|
| n/a |
|---|
| Name | BDBM562067 |
|---|
| Synonyms: | US11396516, Example 2 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C19H21NO6 |
|---|
| Mol. Mass. | 359.3731 |
|---|
| SMILES | CC(C)(C)OC(=O)NCCCOc1c2ccoc2cc2oc(=O)ccc12 |
|---|
| Structure | 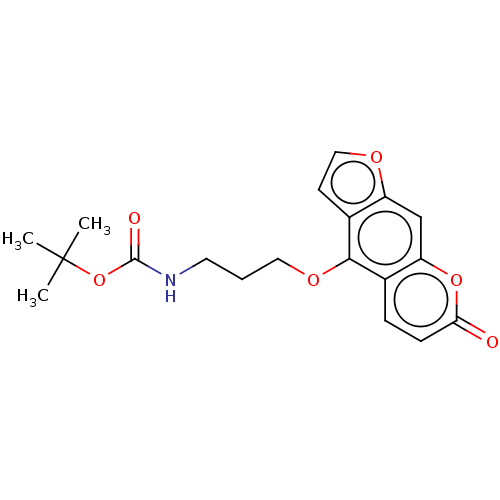
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Peterson, RT; Asnani, A; Liu, Y; Zheng, B; Wang, Y Tricyclic compounds as Cyp1 inhibitors US Patent US11396516 Publication Date 7/26/2022
Peterson, RT; Asnani, A; Liu, Y; Zheng, B; Wang, Y Tricyclic compounds as Cyp1 inhibitors US Patent US11396516 Publication Date 7/26/2022