| Reaction Details |
|---|
 | Report a problem with these data |
| Target | 5'-AMP-activated protein kinase catalytic subunit alpha-2 |
|---|
| Ligand | BDBM328265 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | Enzyme-Linked Immunosorbent Assay |
|---|
| EC50 | >10000±n/a nM |
|---|
| Citation |  Irving, M; Shaw, S AMPK-activating heterocyclic compounds and methods for using the same US Patent US9663496 Publication Date 5/30/2017 Irving, M; Shaw, S AMPK-activating heterocyclic compounds and methods for using the same US Patent US9663496 Publication Date 5/30/2017 |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| 5'-AMP-activated protein kinase catalytic subunit alpha-2 |
|---|
| Name: | 5'-AMP-activated protein kinase catalytic subunit alpha-2 |
|---|
| Synonyms: | AAPK2_HUMAN | ACACA kinase | AMP-activated protein kinase, alpha-2 subunit | AMPK | AMPK subunit alpha-2 | AMPK-alpha2 | AMPK2 | Acetyl-CoA carboxylase kinase | Hydroxymethylglutaryl-CoA reductase kinase | PRKAA2 |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 62331.02 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P54646 |
|---|
| Residue: | 552 |
|---|
| Sequence: | MAEKQKHDGRVKIGHYVLGDTLGVGTFGKVKIGEHQLTGHKVAVKILNRQKIRSLDVVGK
IKREIQNLKLFRHPHIIKLYQVISTPTDFFMVMEYVSGGELFDYICKHGRVEEMEARRLF
QQILSAVDYCHRHMVVHRDLKPENVLLDAHMNAKIADFGLSNMMSDGEFLRTSCGSPNYA
APEVISGRLYAGPEVDIWSCGVILYALLCGTLPFDDEHVPTLFKKIRGGVFYIPEYLNRS
VATLLMHMLQVDPLKRATIKDIREHEWFKQDLPSYLFPEDPSYDANVIDDEAVKEVCEKF
ECTESEVMNSLYSGDPQDQLAVAYHLIIDNRRIMNQASEFYLASSPPSGSFMDDSAMHIP
PGLKPHPERMPPLIADSPKARCPLDALNTTKPKSLAVKKAKWHLGIRSQSKPYDIMAEVY
RAMKQLDFEWKVVNAYHLRVRRKNPVTGNYVKMSLQLYLVDNRSYLLDFKSIDDEVVEQR
SGSSTPQRSCSAAGLHRPRSSFDSTTAESHSLSGSLTGSLTGSTLSSVSPRLGSHTMDFF
EMCASLITTLAR
|
|
|
|---|
| BDBM328265 |
|---|
| n/a |
|---|
| Name | BDBM328265 |
|---|
| Synonyms: | 5-(4-(4-methoxybenzoyl) piperidine-1-carbonyl)- N-(1-(4-(14-oxo-18- ((3aS,4S,6aR)-2- oxohexahydro-1H- thieno[3,4-d]imidazol-4- yl)-4,7,10-trioxa-13- azaoctadecanamido) benzyl)piperidin-4- yl)picolinamide | US10377742, Compound 403 | US9663496, Compound 403 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C51H68N8O10S |
|---|
| Mol. Mass. | 985.198 |
|---|
| SMILES | COc1ccc(cc1)C(=O)C1CCN(CC1)C(=O)c1ccc(nc1)C(=O)NC1CCN(Cc2ccc(NC(=O)CCOCCOCCOCCNC(=O)CCCC[C@@H]3SC[C@@H]4NC(=O)N[C@H]34)cc2)CC1 |r| |
|---|
| Structure | 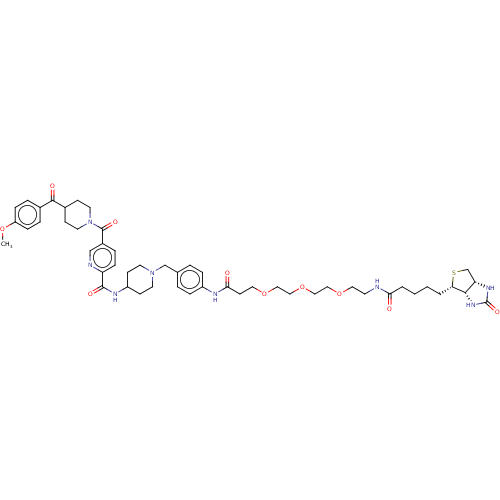
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Irving, M; Shaw, S AMPK-activating heterocyclic compounds and methods for using the same US Patent US9663496 Publication Date 5/30/2017
Irving, M; Shaw, S AMPK-activating heterocyclic compounds and methods for using the same US Patent US9663496 Publication Date 5/30/2017