| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Bromodomain-containing protein 4 |
|---|
| Ligand | BDBM329756 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | Time-Resolved Fluorescence Resonance energy Transfer (TR-FRET) Assay |
|---|
| IC50 | 240±n/a nM |
|---|
| Citation |  Schmees, N; Kuhnke, J; Haendler, B; Neuhaus, R; Lejeune, P; Siegel, S; Krüger, M; Fernandez-Montalvan, AE; Künzer, H; Gallenkamp, D BET protein-inhibiting 5-aryltriazoleazepines US Patent US9663523 Publication Date 5/30/2017 Schmees, N; Kuhnke, J; Haendler, B; Neuhaus, R; Lejeune, P; Siegel, S; Krüger, M; Fernandez-Montalvan, AE; Künzer, H; Gallenkamp, D BET protein-inhibiting 5-aryltriazoleazepines US Patent US9663523 Publication Date 5/30/2017 |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Bromodomain-containing protein 4 |
|---|
| Name: | Bromodomain-containing protein 4 |
|---|
| Synonyms: | BRD4 | BRD4_HUMAN | Bromodomain-containing protein 4 (BRD4) | HUNK1 | Protein HUNK1 |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 152264.84 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | O60885 |
|---|
| Residue: | 1362 |
|---|
| Sequence: | MSAESGPGTRLRNLPVMGDGLETSQMSTTQAQAQPQPANAASTNPPPPETSNPNKPKRQT
NQLQYLLRVVLKTLWKHQFAWPFQQPVDAVKLNLPDYYKIIKTPMDMGTIKKRLENNYYW
NAQECIQDFNTMFTNCYIYNKPGDDIVLMAEALEKLFLQKINELPTEETEIMIVQAKGRG
RGRKETGTAKPGVSTVPNTTQASTPPQTQTPQPNPPPVQATPHPFPAVTPDLIVQTPVMT
VVPPQPLQTPPPVPPQPQPPPAPAPQPVQSHPPIIAATPQPVKTKKGVKRKADTTTPTTI
DPIHEPPSLPPEPKTTKLGQRRESSRPVKPPKKDVPDSQQHPAPEKSSKVSEQLKCCSGI
LKEMFAKKHAAYAWPFYKPVDVEALGLHDYCDIIKHPMDMSTIKSKLEAREYRDAQEFGA
DVRLMFSNCYKYNPPDHEVVAMARKLQDVFEMRFAKMPDEPEEPVVAVSSPAVPPPTKVV
APPSSSDSSSDSSSDSDSSTDDSEEERAQRLAELQEQLKAVHEQLAALSQPQQNKPKKKE
KDKKEKKKEKHKRKEEVEENKKSKAKEPPPKKTKKNNSSNSNVSKKEPAPMKSKPPPTYE
SEEEDKCKPMSYEEKRQLSLDINKLPGEKLGRVVHIIQSREPSLKNSNPDEIEIDFETLK
PSTLRELERYVTSCLRKKRKPQAEKVDVIAGSSKMKGFSSSESESSSESSSSDSEDSETE
MAPKSKKKGHPGREQKKHHHHHHQQMQQAPAPVPQQPPPPPQQPPPPPPPQQQQQPPPPP
PPPSMPQQAAPAMKSSPPPFIATQVPVLEPQLPGSVFDPIGHFTQPILHLPQPELPPHLP
QPPEHSTPPHLNQHAVVSPPALHNALPQQPSRPSNRAAALPPKPARPPAVSPALTQTPLL
PQPPMAQPPQVLLEDEEPPAPPLTSMQMQLYLQQLQKVQPPTPLLPSVKVQSQPPPPLPP
PPHPSVQQQLQQQPPPPPPPQPQPPPQQQHQPPPRPVHLQPMQFSTHIQQPPPPQGQQPP
HPPPGQQPPPPQPAKPQQVIQHHHSPRHHKSDPYSTGHLREAPSPLMIHSPQMSQFQSLT
HQSPPQQNVQPKKQELRAASVVQPQPLVVVKEEKIHSPIIRSEPFSPSLRPEPPKHPESI
KAPVHLPQRPEMKPVDVGRPVIRPPEQNAPPPGAPDKDKQKQEPKTPVAPKKDLKIKNMG
SWASLVQKHPTTPSSTAKSSSDSFEQFRRAAREKEEREKALKAQAEHAEKEKERLRQERM
RSREDEDALEQARRAHEEARRRQEQQQQQRQEQQQQQQQQAAAVAAAATPQAQSSQPQSM
LDQQRELARKREQERRRREAMAATIDMNFQSDLLSIFEENLF
|
|
|
|---|
| BDBM329756 |
|---|
| n/a |
|---|
| Name | BDBM329756 |
|---|
| Synonyms: | Preparation of 2-[6-(4-chlorophenyl)-1-methyl-4H-[1,2,4]triazolo[4,3-a][1]benzazepin-4-yl]-N-(2-oxo-2,3-dihydro-1H-indol-5-yl)acetamide | US9663523, Example 20 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C28H22ClN5O2 |
|---|
| Mol. Mass. | 495.96 |
|---|
| SMILES | Cc1nnc2C(CC(=O)Nc3ccc4NC(=O)Cc4c3)C=C(c3ccc(Cl)cc3)c3ccccc3-n12 |t:22| |
|---|
| Structure | 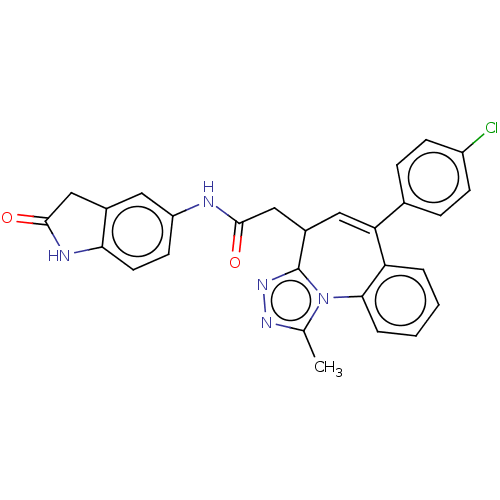
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Schmees, N; Kuhnke, J; Haendler, B; Neuhaus, R; Lejeune, P; Siegel, S; Krüger, M; Fernandez-Montalvan, AE; Künzer, H; Gallenkamp, D BET protein-inhibiting 5-aryltriazoleazepines US Patent US9663523 Publication Date 5/30/2017
Schmees, N; Kuhnke, J; Haendler, B; Neuhaus, R; Lejeune, P; Siegel, S; Krüger, M; Fernandez-Montalvan, AE; Künzer, H; Gallenkamp, D BET protein-inhibiting 5-aryltriazoleazepines US Patent US9663523 Publication Date 5/30/2017