| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 2A6 |
|---|
| Ligand | BDBM12371 |
|---|
| Substrate/Competitor | BDBM12342 |
|---|
| Meas. Tech. | CYP2A6 Inhibition Assay |
|---|
| Ki | 400±n/a nM |
|---|
| Citation |  Yano, JK; Denton, TT; Cerny, MA; Zhang, X; Johnson, EF; Cashman, JR Synthetic inhibitors of cytochrome P-450 2A6: inhibitory activity, difference spectra, mechanism of inhibition, and protein cocrystallization. J Med Chem49:6987-7001 (2006) [PubMed] Article Yano, JK; Denton, TT; Cerny, MA; Zhang, X; Johnson, EF; Cashman, JR Synthetic inhibitors of cytochrome P-450 2A6: inhibitory activity, difference spectra, mechanism of inhibition, and protein cocrystallization. J Med Chem49:6987-7001 (2006) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Solution Info, Assay Method |
|---|
| |
| Cytochrome P450 2A6 |
|---|
| Name: | Cytochrome P450 2A6 |
|---|
| Synonyms: | 1,4-cineole 2-exo-monooxygenase | 1.14.13.- | CP2A6_HUMAN | CYP2A3 | CYP2A6 | CYPIIA6 | Coumarin 7-hydroxylase | Cytochrome P450 2A6 | Cytochrome P450 IIA3 | Cytochrome P450(I) |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 56514.34 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P11509 |
|---|
| Residue: | 494 |
|---|
| Sequence: | MLASGMLLVALLVCLTVMVLMSVWQQRKSKGKLPPGPTPLPFIGNYLQLNTEQMYNSLMK
ISERYGPVFTIHLGPRRVVVLCGHDAVREALVDQAEEFSGRGEQATFDWVFKGYGVVFSN
GERAKQLRRFSIATLRDFGVGKRGIEERIQEEAGFLIDALRGTGGANIDPTFFLSRTVSN
VISSIVFGDRFDYKDKEFLSLLRMMLGIFQFTSTSTGQLYEMFSSVMKHLPGPQQQAFQL
LQGLEDFIAKKVEHNQRTLDPNSPRDFIDSFLIRMQEEEKNPNTEFYLKNLVMTTLNLFI
GGTETVSTTLRYGFLLLMKHPEVEAKVHEEIDRVIGKNRQPKFEDRAKMPYMEAVIHEIQ
RFGDVIPMSLARRVKKDTKFRDFFLPKGTEVYPMLGSVLRDPSFFSNPQDFNPQHFLNEK
GQFKKSDAFVPFSIGKRNCFGEGLARMELFLFFTTVMQNFRLKSSQSPKDIDVSPKHVGF
ATIPRNYTMSFLPR
|
|
|
|---|
| BDBM12371 |
|---|
| BDBM12342 |
|---|
| Name | BDBM12371 |
|---|
| Synonyms: | (3-(Pyridin-3-yl)isoxazol-5-yl)methanamine Dihydrochloride | [3-(3-pyridyl)isoxazol-5-yl]methylamine | [3-(pyridin-3-yl)-1,2-oxazol-5-yl]methanamine dihydrochloride | nicotine 3-heteroaromatic analogue 35 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C9H9N3O |
|---|
| Mol. Mass. | 175.1873 |
|---|
| SMILES | NCc1cc(no1)-c1cccnc1 |
|---|
| Structure | 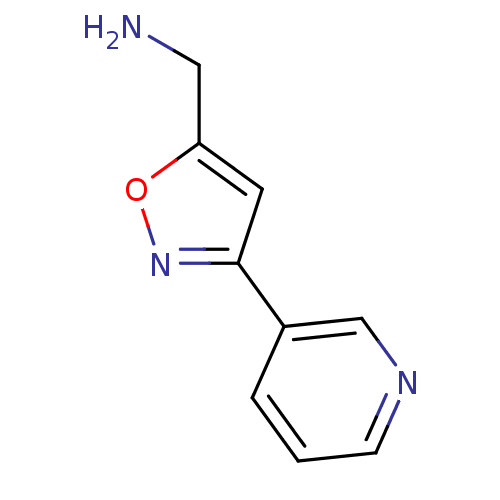
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Yano, JK; Denton, TT; Cerny, MA; Zhang, X; Johnson, EF; Cashman, JR Synthetic inhibitors of cytochrome P-450 2A6: inhibitory activity, difference spectra, mechanism of inhibition, and protein cocrystallization. J Med Chem49:6987-7001 (2006) [PubMed] Article
Yano, JK; Denton, TT; Cerny, MA; Zhang, X; Johnson, EF; Cashman, JR Synthetic inhibitors of cytochrome P-450 2A6: inhibitory activity, difference spectra, mechanism of inhibition, and protein cocrystallization. J Med Chem49:6987-7001 (2006) [PubMed] Article