| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Nuclear receptor subfamily 4 group A member 1 |
|---|
| Ligand | BDBM42108 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | Fluorescence Polarization Dose Response Assay for TR3-Based Bcl-B Inhibitors |
|---|
| EC50 | 33360±66780 nM |
|---|
| Citation |  PubChem, PC Fluorescence Polarization Dose Response Assay for TR3-Based Bcl-B Inhibitors PubChem Bioassay(2008)[AID] PubChem, PC Fluorescence Polarization Dose Response Assay for TR3-Based Bcl-B Inhibitors PubChem Bioassay(2008)[AID] |
|---|
| More Info.: | Get all data from this article, Solution Info, Assay Method |
|---|
| |
| Nuclear receptor subfamily 4 group A member 1 |
|---|
| Name: | Nuclear receptor subfamily 4 group A member 1 |
|---|
| Synonyms: | GFRP1 | HMR | NAK1 | NR4A1 | NR4A1_HUMAN | nuclear receptor subfamily 4, group A, member 1 |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 64467.13 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | gi_27894344 |
|---|
| Residue: | 598 |
|---|
| Sequence: | MPCIQAQYGTPAPSPGPRDHLASDPLTPEFIKPTMDLASPEAAPAAPTALPSFSTFMDGY
TGEFDTFLYQLPGTVQPCSSASSSASSTSSSSATSPASASFKFEDFQVYGCYPGPLSGPV
DEALSSSGSDYYGSPCSAPSPSTPSFQPPQLSPWDGSFGHFSPSQTYEGLRAWTEQLPKA
SGPPQPPAFFSFSPPTGPSPSLAQSPLKLFPSQATHQLGEGESYSMPTAFPGLAPTSPHL
EGSGILDTPVTSTKARSGAPGGSEGRCAVCGDNASCQHYGVRTCEGCKGFFKRTVQKNAK
YICLANKDCPVDKRRRNRCQFCRFQKCLAVGMVKEVVRTDSLKGRRGRLPSKPKQPPDAS
PANLLTSLVRAHLDSGPSTAKLDYSKFQELVLPHFGKEDAGDVQQFYDLLSGSLEVIRKW
AEKIPGFAELSPADQDLLLESAFLELFILRLAYRSKPGEGKLIFCSGLVLHRLQCARGFG
DWIDSILAFSRSLHSLLVDVPAFACLSALVLITDRHGLQEPRRVEELQNRIASCLKEHVA
AVAGEPQPASCLSRLLGKLPELRTLCTQGLQRIFYLKLEDLVPPPPIIDKIFMDTLPF
|
|
|
|---|
| BDBM42108 |
|---|
| n/a |
|---|
| Name | BDBM42108 |
|---|
| Synonyms: | (4Z)-1-(3-chloranyl-4-fluoranyl-phenyl)-4-(furan-2-ylmethylidene)pyrazolidine-3,5-dione | (4Z)-1-(3-chloro-4-fluoro-phenyl)-4-(2-furfurylidene)pyrazolidine-3,5-quinone | (4Z)-1-(3-chloro-4-fluorophenyl)-4-(2-furanylmethylidene)pyrazolidine-3,5-dione | (4Z)-1-(3-chloro-4-fluorophenyl)-4-(furan-2-ylmethylidene)pyrazolidine-3,5-dione | BIM-0049357.P001 | cid_5343905 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C14H8ClFN2O3 |
|---|
| Mol. Mass. | 306.676 |
|---|
| SMILES | Fc1ccc(cc1Cl)N1NC(=O)\C(=C\c2ccco2)C1=O |
|---|
| Structure | 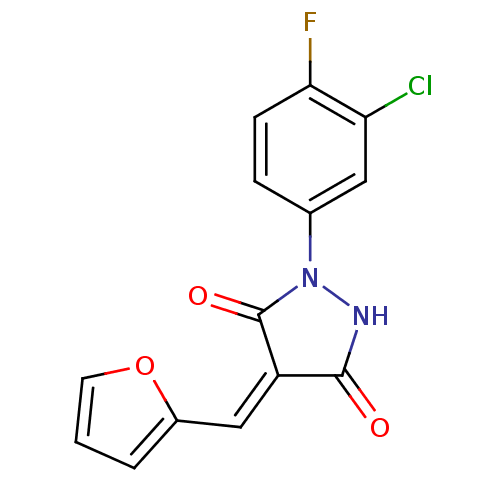
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 PubChem, PC Fluorescence Polarization Dose Response Assay for TR3-Based Bcl-B Inhibitors PubChem Bioassay(2008)[AID]
PubChem, PC Fluorescence Polarization Dose Response Assay for TR3-Based Bcl-B Inhibitors PubChem Bioassay(2008)[AID]