| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Neuropeptide Y receptor type 1 |
|---|
| Ligand | BDBM42532 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | Dose response counterscreen for neuropeptide Y receptor Y2 (NPY-Y2): Cell-based high throughput assay to measure NPY-Y1 antagonism |
|---|
| IC50 | 4079±n/a nM |
|---|
| Citation |  PubChem, PC Dose response counterscreen for neuropeptide Y receptor Y2 (NPY-Y2): Cell-based high throughput assay to measure NPY-Y1 antagonism PubChem Bioassay(2008)[AID] PubChem, PC Dose response counterscreen for neuropeptide Y receptor Y2 (NPY-Y2): Cell-based high throughput assay to measure NPY-Y1 antagonism PubChem Bioassay(2008)[AID] |
|---|
| More Info.: | Get all data from this article, Solution Info, Assay Method |
|---|
| |
| Neuropeptide Y receptor type 1 |
|---|
| Name: | Neuropeptide Y receptor type 1 |
|---|
| Synonyms: | NPY-Y1 | NPY1-R | NPY1R | NPY1R_HUMAN | NPYR | NPYY1 | neuropeptide Y receptor Y1 |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 44399.07 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | NPY-Y1 NPY1R HUMAN::P25929 |
|---|
| Residue: | 384 |
|---|
| Sequence: | MNSTLFSQVENHSVHSNFSEKNAQLLAFENDDCHLPLAMIFTLALAYGAVIILGVSGNLA
LIIIILKQKEMRNVTNILIVNLSFSDLLVAIMCLPFTFVYTLMDHWVFGEAMCKLNPFVQ
CVSITVSIFSLVLIAVERHQLIINPRGWRPNNRHAYVGIAVIWVLAVASSLPFLIYQVMT
DEPFQNVTLDAYKDKYVCFDQFPSDSHRLSYTTLLLVLQYFGPLCFIFICYFKIYIRLKR
RNNMMDKMRDNKYRSSETKRINIMLLSIVVAFAVCWLPLTIFNTVFDWNHQIIATCNHNL
LFLLCHLTAMISTCVNPIFYGFLNKNFQRDLQFFFNFCDFRSRDDDYETIAMSTMHTDVS
KTSLKQASPVAFKKINNNDDNEKI
|
|
|
|---|
| BDBM42532 |
|---|
| n/a |
|---|
| Name | BDBM42532 |
|---|
| Synonyms: | 2-[1-[(Z)-1-(4-chlorobenzoyl)-2-(2-furyl)vinyl]-2-pyridylidene]malononitrile | 2-[1-[(Z)-3-(4-chlorophenyl)-1-(2-furanyl)-3-oxoprop-1-en-2-yl]-2-pyridinylidene]propanedinitrile | 2-[1-[(Z)-3-(4-chlorophenyl)-1-(furan-2-yl)-3-oxidanylidene-prop-1-en-2-yl]pyridin-2-ylidene]propanedinitrile | 2-[1-[(Z)-3-(4-chlorophenyl)-1-(furan-2-yl)-3-oxoprop-1-en-2-yl]pyridin-2-ylidene]propanedinitrile | 2-{1-[(Z)-1-(4-Chloro-benzoyl)-2-furan-2-yl-vinyl]-1H-pyridin-2-ylidene}-malononitrile | MLS000589524 | SMR000213008 | cid_5806576 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C21H12ClN3O2 |
|---|
| Mol. Mass. | 373.792 |
|---|
| SMILES | Clc1ccc(cc1)C(=O)C(=C\c1ccco1)\N1C=CC=CC1=C(C#N)C#N |c:19,21| |
|---|
| Structure | 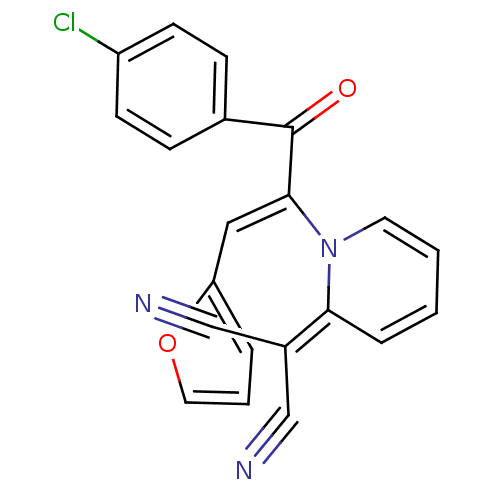
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 PubChem, PC Dose response counterscreen for neuropeptide Y receptor Y2 (NPY-Y2): Cell-based high throughput assay to measure NPY-Y1 antagonism PubChem Bioassay(2008)[AID]
PubChem, PC Dose response counterscreen for neuropeptide Y receptor Y2 (NPY-Y2): Cell-based high throughput assay to measure NPY-Y1 antagonism PubChem Bioassay(2008)[AID]