| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Glycoprotein 42 |
|---|
| Ligand | BDBM48838 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | Dose Response Confirmation for Small Molecule Inhibitors of Epstein-Barr Virus |
|---|
| IC50 | 1580±n/a nM |
|---|
| Citation |  PubChem, PC Dose Response Confirmation for Small Molecule Inhibitors of Epstein-Barr Virus PubChem Bioassay(2008)[AID] PubChem, PC Dose Response Confirmation for Small Molecule Inhibitors of Epstein-Barr Virus PubChem Bioassay(2008)[AID] |
|---|
| More Info.: | Get all data from this article, Solution Info, Assay Method |
|---|
| |
| Glycoprotein 42 |
|---|
| Name: | Glycoprotein 42 |
|---|
| Synonyms: | BZLF2 | GP42_EBVA8 |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 25424.12 |
|---|
| Organism: | Human herpesvirus 4 type 2 |
|---|
| Description: | gi_139424501 |
|---|
| Residue: | 223 |
|---|
| Sequence: | MVSFKQVRVPLFTAIALVIVLLLAYFLPPRVRGGGRVSAAAITWVPKPNVEVWPVDPPPP
VNFNKTAEQEYGDKEIKLPHWTPTLHTFQVPKNYTKANCTYCNTREYTFSYKERCFYFTK
KKHTWNGCFQACAELYPCTYFYGPTPDILPVVTRNLNAIESLWVGVYRVGEGNWTSLDGG
TFKVYQIFGSHCTYVSKFSTVPVSHHECSFLKPCLCVSQRSNS
|
|
|
|---|
| BDBM48838 |
|---|
| n/a |
|---|
| Name | BDBM48838 |
|---|
| Synonyms: | (5Z)-2-(4-hydroxyanilino)-5-(2-ketoindolin-3-ylidene)-2-thiazolin-4-one | (5Z)-2-(4-hydroxyanilino)-5-(2-oxo-1H-indol-3-ylidene)-1,3-thiazol-4-one | (5Z)-2-(4-hydroxyanilino)-5-(2-oxo-1H-indol-3-ylidene)-4-thiazolone | (5Z)-2-[(4-hydroxyphenyl)amino]-5-(2-oxidanylidene-1H-indol-3-ylidene)-1,3-thiazol-4-one | 3-[2-[(4-hydroxyphenyl)amino]-4-oxo-1,3-thiazol-5(4H)-ylidene]-1,3-dihydro-2H-indol-2-one | MLS000676656 | SMR000298307 | cid_5398245 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C17H11N3O3S |
|---|
| Mol. Mass. | 337.353 |
|---|
| SMILES | Oc1ccc(NC2=NC(=O)C(S2)=C2C(=O)Nc3ccccc23)cc1 |w:12.22,t:6| |
|---|
| Structure | 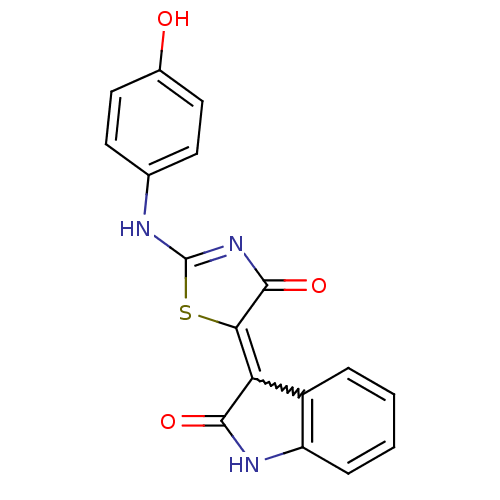
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 PubChem, PC Dose Response Confirmation for Small Molecule Inhibitors of Epstein-Barr Virus PubChem Bioassay(2008)[AID]
PubChem, PC Dose Response Confirmation for Small Molecule Inhibitors of Epstein-Barr Virus PubChem Bioassay(2008)[AID]