| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Beta-lactamase |
|---|
| Ligand | BDBM51974 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | FRET-based counterscreen for selective VIM-2 inhibitors: dose response biochemical high throughput screening assay to identify inhibitors of IMP-1 metallo-beta-lactamase. |
|---|
| IC50 | 59640±n/a nM |
|---|
| Citation |  PubChem, PC FRET-based counterscreen for selective VIM-2 inhibitors: dose response biochemical high throughput screening assay to identify inhibitors of IMP-1 metallo-beta-lactamase. PubChem Bioassay(2009)[AID] PubChem, PC FRET-based counterscreen for selective VIM-2 inhibitors: dose response biochemical high throughput screening assay to identify inhibitors of IMP-1 metallo-beta-lactamase. PubChem Bioassay(2009)[AID] |
|---|
| More Info.: | Get all data from this article, Solution Info, Assay Method |
|---|
| |
| Beta-lactamase |
|---|
| Name: | Beta-lactamase |
|---|
| Synonyms: | Beta-lactamase | metallo-beta-lactamase IMP-1 |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 27125.88 |
|---|
| Organism: | Pseudomonas aeruginosa |
|---|
| Description: | gi_27368096 |
|---|
| Residue: | 246 |
|---|
| Sequence: | MSKLSVFFIFLFCSIATAAESLPDLKIEKLDEGVYVHTSFEEVNGWGVVPKHGLVVLVNA
EAYLIDTPFTAKDTEKLVTWFVERGYKIKGSISSHFHSDSTGGIEWLNSRSIPTYASELT
NELLKKDGKVQATNSFSGVNYWLVKNKIEVFYPGPGHTPDNVVVWLPERKILFGGCFIKP
YGLGNLGDANIEAWPKSAKLLKSKYGKAKLVVPSHSEVGDASLLKLTLEQAVKGLNESKK
PSKPSN
|
|
|
|---|
| BDBM51974 |
|---|
| n/a |
|---|
| Name | BDBM51974 |
|---|
| Synonyms: | 3-methyl-7-[(1-methyl-4-nitro-pyrazol-3-yl)carbonylamino]-8-oxidanylidene-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid | 3-methyl-7-[(1-methyl-4-nitropyrazole-3-carbonyl)amino]-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid | 3-methyl-7-[[(1-methyl-4-nitro-3-pyrazolyl)-oxomethyl]amino]-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid | 7-[({4-nitro-1-methyl-1H-pyrazol-3-yl}carbonyl)amino]-3-methyl-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid | 8-keto-3-methyl-7-[(1-methyl-4-nitro-pyrazole-3-carbonyl)amino]-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid | MLS000705418 | SMR000231345 | cid_4766021 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C13H13N5O6S |
|---|
| Mol. Mass. | 367.337 |
|---|
| SMILES | CC1=C(N2C(SC1)C(NC(=O)c1nn(C)cc1[N+]([O-])=O)C2=O)C(O)=O |t:1| |
|---|
| Structure | 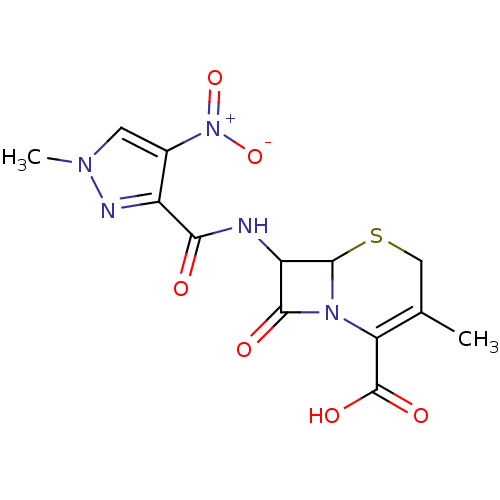
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 PubChem, PC FRET-based counterscreen for selective VIM-2 inhibitors: dose response biochemical high throughput screening assay to identify inhibitors of IMP-1 metallo-beta-lactamase. PubChem Bioassay(2009)[AID]
PubChem, PC FRET-based counterscreen for selective VIM-2 inhibitors: dose response biochemical high throughput screening assay to identify inhibitors of IMP-1 metallo-beta-lactamase. PubChem Bioassay(2009)[AID]