| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Type-1 angiotensin II receptor |
|---|
| Ligand | BDBM54849 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | Dose Response screen for antagonists of Angiotensin II Receptor Type 1 to assess selectivity of uHTS small molecule antagonists hits of the APJ receptor |
|---|
| IC50 | 5897±1023 nM |
|---|
| Citation |  PubChem, PC Dose Response screen for antagonists of Angiotensin II Receptor Type 1 to assess selectivity of uHTS small molecule antagonists hits of the APJ receptor PubChem Bioassay(2010)[AID] PubChem, PC Dose Response screen for antagonists of Angiotensin II Receptor Type 1 to assess selectivity of uHTS small molecule antagonists hits of the APJ receptor PubChem Bioassay(2010)[AID] |
|---|
| More Info.: | Get all data from this article, Solution Info, Assay Method |
|---|
| |
| Type-1 angiotensin II receptor |
|---|
| Name: | Type-1 angiotensin II receptor |
|---|
| Synonyms: | AGTR1 | AGTR1A | AGTR1B | AGTR1_HUMAN | AT1 | AT1AR | AT1BR | AT2R1 | AT2R1B | Angiotensin II AT1 | Angiotensin II receptor | Angiotensin II type 1b (AT-1b) receptor | Type-1 angiotensin II receptor (AT1) |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 41080.75 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P30556 |
|---|
| Residue: | 359 |
|---|
| Sequence: | MILNSSTEDGIKRIQDDCPKAGRHNYIFVMIPTLYSIIFVVGIFGNSLVVIVIYFYMKLK
TVASVFLLNLALADLCFLLTLPLWAVYTAMEYRWPFGNYLCKIASASVSFNLYASVFLLT
CLSIDRYLAIVHPMKSRLRRTMLVAKVTCIIIWLLAGLASLPAIIHRNVFFIENTNITVC
AFHYESQNSTLPIGLGLTKNILGFLFPFLIILTSYTLIWKALKKAYEIQKNKPRNDDIFK
IIMAIVLFFFFSWIPHQIFTFLDVLIQLGIIRDCRIADIVDTAMPITICIAYFNNCLNPL
FYGFLGKKFKRYFLQLLKYIPPKAKSHSNLSTKMSTLSYRPSDNVSSSTKKPAPCFEVE
|
|
|
|---|
| BDBM54849 |
|---|
| n/a |
|---|
| Name | BDBM54849 |
|---|
| Synonyms: | 5-chloranyl-N-[4-(2-ethylpiperidin-1-yl)sulfonylphenyl]-2-methylsulfonyl-pyrimidine-4-carboxamide | 5-chloro-N-[4-(2-ethylpiperidin-1-yl)sulfonylphenyl]-2-methylsulfonylpyrimidine-4-carboxamide | 5-chloro-N-[4-(2-ethylpiperidino)sulfonylphenyl]-2-mesyl-pyrimidine-4-carboxamide | 5-chloro-N-[4-[(2-ethyl-1-piperidinyl)sulfonyl]phenyl]-2-methylsulfonyl-4-pyrimidinecarboxamide | BRD-A72341983-001-05-8 | cid_16437205 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C19H23ClN4O5S2 |
|---|
| Mol. Mass. | 486.993 |
|---|
| SMILES | CCC1CCCCN1S(=O)(=O)c1ccc(NC(=O)c2nc(ncc2Cl)S(C)(=O)=O)cc1 |
|---|
| Structure | 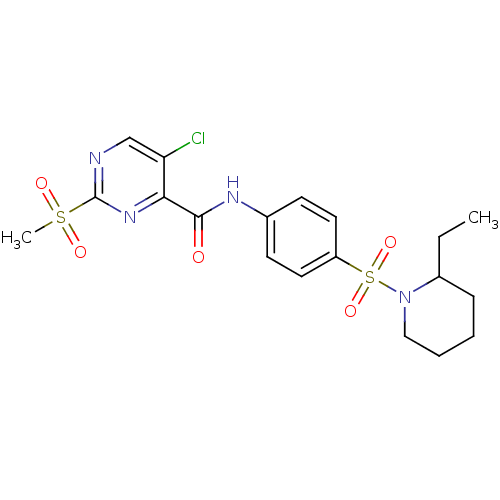
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 PubChem, PC Dose Response screen for antagonists of Angiotensin II Receptor Type 1 to assess selectivity of uHTS small molecule antagonists hits of the APJ receptor PubChem Bioassay(2010)[AID]
PubChem, PC Dose Response screen for antagonists of Angiotensin II Receptor Type 1 to assess selectivity of uHTS small molecule antagonists hits of the APJ receptor PubChem Bioassay(2010)[AID]