| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Orexin/Hypocretin receptor type 1 |
|---|
| Ligand | BDBM79912 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | Fluorescence-based cell-based high throughput dose response assay for antagonists of the orexin 1 receptor (OX1R; HCRTR1) |
|---|
| IC50 | 4281±n/a nM |
|---|
| Citation |  PubChem, PC Fluorescence-based cell-based high throughput dose response assay for antagonists of the orexin 1 receptor (OX1R; HCRTR1) PubChem Bioassay(2011)[AID] PubChem, PC Fluorescence-based cell-based high throughput dose response assay for antagonists of the orexin 1 receptor (OX1R; HCRTR1) PubChem Bioassay(2011)[AID] |
|---|
| More Info.: | Get all data from this article, Solution Info, Assay Method |
|---|
| |
| Orexin/Hypocretin receptor type 1 |
|---|
| Name: | Orexin/Hypocretin receptor type 1 |
|---|
| Synonyms: | HCRTR1 | Hypocretin receptor type 1 | OX1R_HUMAN | Orexin receptor type 1 | Orexin receptor type 1 (OR 1) | Orexin receptor type 1 (OR-1) | Orexin receptor type 1 (OX1) | Orexin receptor type 1 (OX1R) | Orexin receptor type 1 (OxR1) | Ox1r |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 47554.50 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | O43613 |
|---|
| Residue: | 425 |
|---|
| Sequence: | MEPSATPGAQMGVPPGSREPSPVPPDYEDEFLRYLWRDYLYPKQYEWVLIAAYVAVFVVA
LVGNTLVCLAVWRNHHMRTVTNYFIVNLSLADVLVTAICLPASLLVDITESWLFGHALCK
VIPYLQAVSVSVAVLTLSFIALDRWYAICHPLLFKSTARRARGSILGIWAVSLAIMVPQA
AVMECSSVLPELANRTRLFSVCDERWADDLYPKIYHSCFFIVTYLAPLGLMAMAYFQIFR
KLWGRQIPGTTSALVRNWKRPSDQLGDLEQGLSGEPQPRARAFLAEVKQMRARRKTAKML
MVVLLVFALCYLPISVLNVLKRVFGMFRQASDREAVYACFTFSHWLVYANSAANPIIYNF
LSGKFREQFKAAFSCCLPGLGPCGSLKAPSPRSSASHKSLSLQSRCSISKISEHVVLTSV
TTVLP
|
|
|
|---|
| BDBM79912 |
|---|
| n/a |
|---|
| Name | BDBM79912 |
|---|
| Synonyms: | (4aS,8R,8aS)-2-(3,4-dichlorobenzyl)-1-keto-N-phenyl-3,4,4a,7,8,8a-hexahydroisoquinoline-8-carboxamide | (4aS,8R,8aS)-2-[(3,4-dichlorophenyl)methyl]-1-oxidanylidene-N-phenyl-3,4,4a,7,8,8a-hexahydroisoquinoline-8-carboxamide | (4aS,8R,8aS)-2-[(3,4-dichlorophenyl)methyl]-1-oxo-N-phenyl-3,4,4a,7,8,8a-hexahydroisoquinoline-8-carboxamide | MLS000882858 | SMR000525469 | cid_24747377 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C23H22Cl2N2O2 |
|---|
| Mol. Mass. | 429.339 |
|---|
| SMILES | Clc1ccc(CN2CC[C@H]3C=CC[C@H]([C@H]3C2=O)C(=O)Nc2ccccc2)cc1Cl |c:10| |
|---|
| Structure | 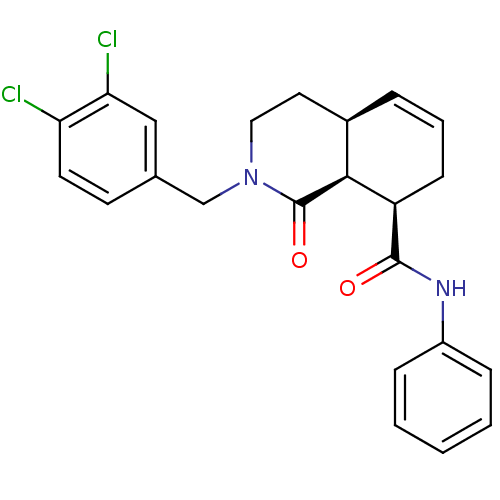
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 PubChem, PC Fluorescence-based cell-based high throughput dose response assay for antagonists of the orexin 1 receptor (OX1R; HCRTR1) PubChem Bioassay(2011)[AID]
PubChem, PC Fluorescence-based cell-based high throughput dose response assay for antagonists of the orexin 1 receptor (OX1R; HCRTR1) PubChem Bioassay(2011)[AID]