| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Protein-glutamine gamma-glutamyltransferase 2 |
|---|
| Ligand | BDBM50241218 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1669694 (CHEMBL4019582) |
|---|
| Ki | 12900±n/a nM |
|---|
| Citation |  Akbar, A; McNeil, NMR; Albert, MR; Ta, V; Adhikary, G; Bourgeois, K; Eckert, RL; Keillor, JW Structure-Activity Relationships of Potent, Targeted Covalent Inhibitors That Abolish Both the Transamidation and GTP Binding Activities of Human Tissue Transglutaminase. J Med Chem60:7910-7927 (2017) [PubMed] Article Akbar, A; McNeil, NMR; Albert, MR; Ta, V; Adhikary, G; Bourgeois, K; Eckert, RL; Keillor, JW Structure-Activity Relationships of Potent, Targeted Covalent Inhibitors That Abolish Both the Transamidation and GTP Binding Activities of Human Tissue Transglutaminase. J Med Chem60:7910-7927 (2017) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Protein-glutamine gamma-glutamyltransferase 2 |
|---|
| Name: | Protein-glutamine gamma-glutamyltransferase 2 |
|---|
| Synonyms: | Protein-glutamine gamma-glutamyltransferase | Protein-glutamine gamma-glutamyltransferase 2 (TG2) | TGM2 | TGM2_HUMAN | Tissue transglutaminase | Tissue transglutaminase (TG2) | Transglutaminase 2 (TGM2) | Transglutaminase C | Transglutaminase-2 |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 77309.30 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P21980 |
|---|
| Residue: | 687 |
|---|
| Sequence: | MAEELVLERCDLELETNGRDHHTADLCREKLVVRRGQPFWLTLHFEGRNYEASVDSLTFS
VVTGPAPSQEAGTKARFPLRDAVEEGDWTATVVDQQDCTLSLQLTTPANAPIGLYRLSLE
ASTGYQGSSFVLGHFILLFNAWCPADAVYLDSEEERQEYVLTQQGFIYQGSAKFIKNIPW
NFGQFEDGILDICLILLDVNPKFLKNAGRDCSRRSSPVYVGRVVSGMVNCNDDQGVLLGR
WDNNYGDGVSPMSWIGSVDILRRWKNHGCQRVKYGQCWVFAAVACTVLRCLGIPTRVVTN
YNSAHDQNSNLLIEYFRNEFGEIQGDKSEMIWNFHCWVESWMTRPDLQPGYEGWQALDPT
PQEKSEGTYCCGPVPVRAIKEGDLSTKYDAPFVFAEVNADVVDWIQQDDGSVHKSINRSL
IVGLKISTKSVGRDEREDITHTYKYPEGSSEEREAFTRANHLNKLAEKEETGMAMRIRVG
QSMNMGSDFDVFAHITNNTAEEYVCRLLLCARTVSYNGILGPECGTKYLLNLNLEPFSEK
SVPLCILYEKYRDCLTESNLIKVRALLVEPVINSYLLAERDLYLENPEIKIRILGEPKQK
RKLVAEVSLQNPLPVALEGCTFTVEGAGLTEEQKTVEIPDPVEAGEEVKVRMDLLPLHMG
LHKLVVNFESDKLKAVKGFRNVIIGPA
|
|
|
|---|
| BDBM50241218 |
|---|
| n/a |
|---|
| Name | BDBM50241218 |
|---|
| Synonyms: | CHEMBL4103395 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C33H41N5O6S |
|---|
| Mol. Mass. | 635.774 |
|---|
| SMILES | CN(C)c1cccc2c(cccc12)S(=O)(=O)N1CCN(CC1)C(=O)[C@H](CCCCNC(=O)C=C)NC(=O)OCc1ccccc1 |r| |
|---|
| Structure | 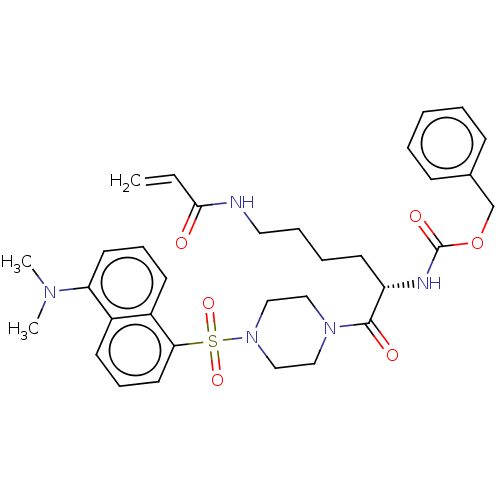
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Akbar, A; McNeil, NMR; Albert, MR; Ta, V; Adhikary, G; Bourgeois, K; Eckert, RL; Keillor, JW Structure-Activity Relationships of Potent, Targeted Covalent Inhibitors That Abolish Both the Transamidation and GTP Binding Activities of Human Tissue Transglutaminase. J Med Chem60:7910-7927 (2017) [PubMed] Article
Akbar, A; McNeil, NMR; Albert, MR; Ta, V; Adhikary, G; Bourgeois, K; Eckert, RL; Keillor, JW Structure-Activity Relationships of Potent, Targeted Covalent Inhibitors That Abolish Both the Transamidation and GTP Binding Activities of Human Tissue Transglutaminase. J Med Chem60:7910-7927 (2017) [PubMed] Article