

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Reaction Details | |||
|---|---|---|---|
 | Report a problem with these data | ||
| Target | Cytochrome P450 2C9 | ||
| Ligand | BDBM50250875 | ||
| Substrate/Competitor | n/a | ||
| Meas. Tech. | ChEBML_1682567 | ||
| IC50 | 11900±n/a nM | ||
| Citation |  Anumala, UR; Waaler, J; Nkizinkiko, Y; Ignatev, A; Lazarow, K; Lindemann, P; Olsen, PA; Murthy, S; Obaji, E; Majouga, AG; Leonov, S; von Kries, JP; Lehti�, L; Krauss, S; Nazar�, M Discovery of a Novel Series of Tankyrase Inhibitors by a Hybridization Approach. J Med Chem60:10013-10025 (2017) [PubMed] Article Anumala, UR; Waaler, J; Nkizinkiko, Y; Ignatev, A; Lazarow, K; Lindemann, P; Olsen, PA; Murthy, S; Obaji, E; Majouga, AG; Leonov, S; von Kries, JP; Lehti�, L; Krauss, S; Nazar�, M Discovery of a Novel Series of Tankyrase Inhibitors by a Hybridization Approach. J Med Chem60:10013-10025 (2017) [PubMed] Article | ||
| More Info.: | Get all data from this article, Assay Method | ||
| Cytochrome P450 2C9 | |||
| Name: | Cytochrome P450 2C9 | ||
| Synonyms: | (R)-limonene 6-monooxygenase | (S)-limonene 6-monooxygenase | CP2C9_HUMAN | CYP2C10 | CYP2C9 | CYPIIC9 | Cytochrome P450 2C9 (CYP2C9 ) | Cytochrome P450 2C9 (CYP2C9) | P-450MP | P450 MP-4/MP-8 | P450 PB-1 | S-mephenytoin 4-hydroxylase | ||
| Type: | Enzyme | ||
| Mol. Mass.: | 55636.33 | ||
| Organism: | Homo sapiens (Human) | ||
| Description: | P11712 | ||
| Residue: | 490 | ||
| Sequence: |
| ||
| BDBM50250875 | |||
| n/a | |||
| Name | BDBM50250875 | ||
| Synonyms: | CHEMBL4095003 | ||
| Type | Small organic molecule | ||
| Emp. Form. | C24H17ClN8O | ||
| Mol. Mass. | 468.898 | ||
| SMILES | Clc1ccccc1-n1c(nnc1-c1ccncn1)[C@H]1C[C@@H](C1)n1c2ccc(cc2[nH]c1=O)C#N |r,wU:20.25,wD:18.20,(25.39,-29.03,;24.05,-29.8,;24.05,-31.34,;22.72,-32.11,;21.39,-31.34,;21.39,-29.8,;22.72,-29.03,;22.72,-27.49,;23.97,-26.59,;23.49,-25.13,;21.95,-25.13,;21.48,-26.59,;20.01,-27.07,;19.69,-28.57,;18.22,-29.05,;17.08,-28.02,;17.4,-26.52,;18.87,-26.04,;25.43,-27.07,;26.13,-28.44,;27.5,-27.74,;26.8,-26.37,;28.97,-28.22,;29.44,-29.68,;28.67,-31.01,;29.44,-32.35,;30.98,-32.35,;31.75,-31.01,;30.98,-29.68,;31.46,-28.22,;30.21,-27.31,;30.21,-25.77,;31.75,-33.68,;32.52,-35.01,)| | ||
| Structure | 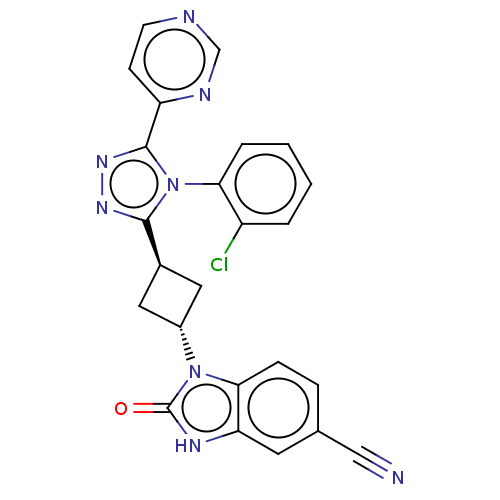
| ||