| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Membrane primary amine oxidase |
|---|
| Ligand | BDBM50268068 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1704403 (CHEMBL4055636) |
|---|
| IC50 | 31±n/a nM |
|---|
| Citation |  Yamaki, S; Koga, Y; Nagashima, A; Kondo, M; Shimada, Y; Kadono, K; Moritomo, A; Yoshihara, K Synthesis and pharmacological evaluation of glycine amide derivatives as novel vascular adhesion protein-1 inhibitors without CYP3A4 and CYP2C19 inhibition. Bioorg Med Chem25:4110-4122 (2017) [PubMed] Article Yamaki, S; Koga, Y; Nagashima, A; Kondo, M; Shimada, Y; Kadono, K; Moritomo, A; Yoshihara, K Synthesis and pharmacological evaluation of glycine amide derivatives as novel vascular adhesion protein-1 inhibitors without CYP3A4 and CYP2C19 inhibition. Bioorg Med Chem25:4110-4122 (2017) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Membrane primary amine oxidase |
|---|
| Name: | Membrane primary amine oxidase |
|---|
| Synonyms: | AOC3 | AOC3_HUMAN | Amine oxidase, copper containing | Semicarbazide-sensitive amine oxidase (SSAO) | Semicarbazide-sensitive amine oxidase/ Vascular adhesion protein 1 (SSAO/VAP-1) | VAP1 | Vascular adhesion protein 1 (VAP-1) |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 84620.86 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Q16853 |
|---|
| Residue: | 763 |
|---|
| Sequence: | MNQKTILVLLILAVITIFALVCVLLVGRGGDGGEPSQLPHCPSVSPSAQPWTHPGQSQLF
ADLSREELTAVMRFLTQRLGPGLVDAAQARPSDNCVFSVELQLPPKAAALAHLDRGSPPP
AREALAIVFFGRQPQPNVSELVVGPLPHPSYMRDVTVERHGGPLPYHRRPVLFQEYLDID
QMIFNRELPQASGLLHHCCFYKHRGRNLVTMTTAPRGLQSGDRATWFGLYYNISGAGFFL
HHVGLELLVNHKALDPARWTIQKVFYQGRYYDSLAQLEAQFEAGLVNVVLIPDNGTGGSW
SLKSPVPPGPAPPLQFYPQGPRFSVQGSRVASSLWTFSFGLGAFSGPRIFDVRFQGERLV
YEISLQEALAIYGGNSPAAMTTRYVDGGFGMGKYTTPLTRGVDCPYLATYVDWHFLLESQ
APKTIRDAFCVFEQNQGLPLRRHHSDLYSHYFGGLAETVLVVRSMSTLLNYDYVWDTVFH
PSGAIEIRFYATGYISSAFLFGATGKYGNQVSEHTLGTVHTHSAHFKVDLDVAGLENWVW
AEDMVFVPMAVPWSPEHQLQRLQVTRKLLEMEEQAAFLVGSATPRYLYLASNHSNKWGHP
RGYRIQMLSFAGEPLPQNSSMARGFSWERYQLAVTQRKEEEPSSSSVFNQNDPWAPTVDF
SDFINNETIAGKDLVAWVTAGFLHIPHAEDIPNTVTVGNGVGFFLRPYNFFDEDPSFYSA
DSIYFRGDQDAGACEVNPLACLPQAAACAPDLPAFSHGGFSHN
|
|
|
|---|
| BDBM50268068 |
|---|
| n/a |
|---|
| Name | BDBM50268068 |
|---|
| Synonyms: | CHEMBL4068336 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C22H29N5O8 |
|---|
| Mol. Mass. | 491.4944 |
|---|
| SMILES | O[C@H]([C@@H](O)C(O)=O)C(O)=O.CN(CC(N)=O)Cc1cccc(c1)-c1cnc(nc1)N1CCOCC1 |r| |
|---|
| Structure | 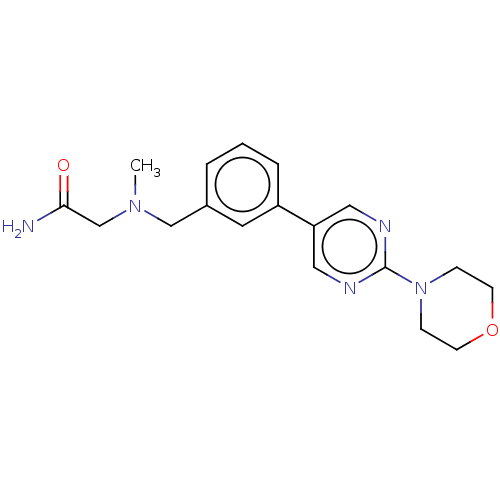
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Yamaki, S; Koga, Y; Nagashima, A; Kondo, M; Shimada, Y; Kadono, K; Moritomo, A; Yoshihara, K Synthesis and pharmacological evaluation of glycine amide derivatives as novel vascular adhesion protein-1 inhibitors without CYP3A4 and CYP2C19 inhibition. Bioorg Med Chem25:4110-4122 (2017) [PubMed] Article
Yamaki, S; Koga, Y; Nagashima, A; Kondo, M; Shimada, Y; Kadono, K; Moritomo, A; Yoshihara, K Synthesis and pharmacological evaluation of glycine amide derivatives as novel vascular adhesion protein-1 inhibitors without CYP3A4 and CYP2C19 inhibition. Bioorg Med Chem25:4110-4122 (2017) [PubMed] Article